Showing all 2 results
When to Use Merrill Crowe Precipitation
Zinc dust cementation, or Merrill-Crowe precipitation is the most widely used method for gold and silver recovery. Because of its simple and efficient operation, the Merrill-Crowe process is currently used at the 10 largest gold producing mines in the free world, all of which are in South Africa. This process is attractive for use at new mines where the ore exhibits a high silver to gold ratio (ranging from 5:1 to 20:1). Ores with high silver values present significant problems to carbon adsorption circuits because the silver causes high carbon stripping requirements which may be economically prohibitive. Several of the existing operations using Merrill-Crowe precipitation actually experience higher silver recoveries than gold.
One other advantage to the zinc cementation system is that once dyamic equilibrium (steady state) is reached in the process plants, relatively few people are required for operation and maintainence (i.e., only simple valve operation and meter reading tasks are required).
The process does, however, have some disadvantages. The leach liquor must be treated (i.e., clarified and deareated) prior to precipitation. Indeed, as noted in a preceding section, inefficient pretreatment is detrimental to successful precipitation. Other disadvantages stem from the process being quite dependent on and sensitive to the pregnant liquor composition. Lower gold concentrations in the feed solution result in higher zinc consumption. This is because the zinc is mostly used to precipitate all of the other metals in solution before gold and silver are recovered. Also, interfering ions such as arsenic and antimony can be problematic, leading, in some cases, to the consideration of other recovery methods.
Zinc Precipitation
Portable Units/Shelf Units. Depending on the expected flow rate and the size of operation, Merrill-Crowe zinc precipitation units can come in a variety of sizes. These range from 20 gallons per minute (1.3 l/sec) in small self-contained units to 4,000 – 5,000 gpm (250 to 315 l/sec) throughput in
custom plants.
Smaller skid-mounted units are available from several suppliers. Each unit comes equipped with all the necessary pumps and piping. All the user must provide is a feed line to the unit, an effluent (barren) line out, and electrical supply. These units are very useful for ranges of flows from 50 to 250 gpm (3 to 16 l/sec). They are easily assembled or dismantled and often are mounted on trailers for easy transportation. However, beyond a certain point, it becomes apparent that the filtration supplied with these units is insufficient for optimum operation. The operator may spend more time cleaning the clarifier filters than actually treating solution. It is for this reason that operations are custom designed for handling large daily volumes of pregnant solution.
Custom Designed Units. Here the choice of size and volume throughput is not limited. Typically, the designs are based on an expected volume of requirements, with a built-in over-design included. Stationary pressure filters, or the leaf-vacuum type, are incorporated as well as vacuum deareators. Plate and frame presses are often a standard. Larger throughputs of these plants justify instrumentation such as turbidimeters or continuous flow (in-line) oxygen probe detectors to facilitate the operation and improve efficiency.
Disadvantages are inherent with the size of the plants. They are usually permanent installations and are expensive to relocate.
Merrill Crowe Process Description
Chemistry of Process. The chemistry of zinc precipitation depends mainly on the fact that gold and silver metals are more noble than zinc. This means that they are more apt to reduce to their native states (i.e., Au° and Ag°) than remain in a gold or silver-cyanide complex. A generalized cementation reaction may be written as follows:
2Au (CN)²- + Zn = 2Au = Zn (CN)4²-
As the gold is precipitated, the zinc combines with the cyanide to form a zinc-cyanide complex. High pH (10.0+) is required to precipitate precious metals and to minimize precipitation of copper which is at times present in solution.
Detailed fundamental studies of the kinetics and mechanisms of gold precipitation were published. Their paper is an excellent reference for information concerning precipitation of precious metals by zinc dust.
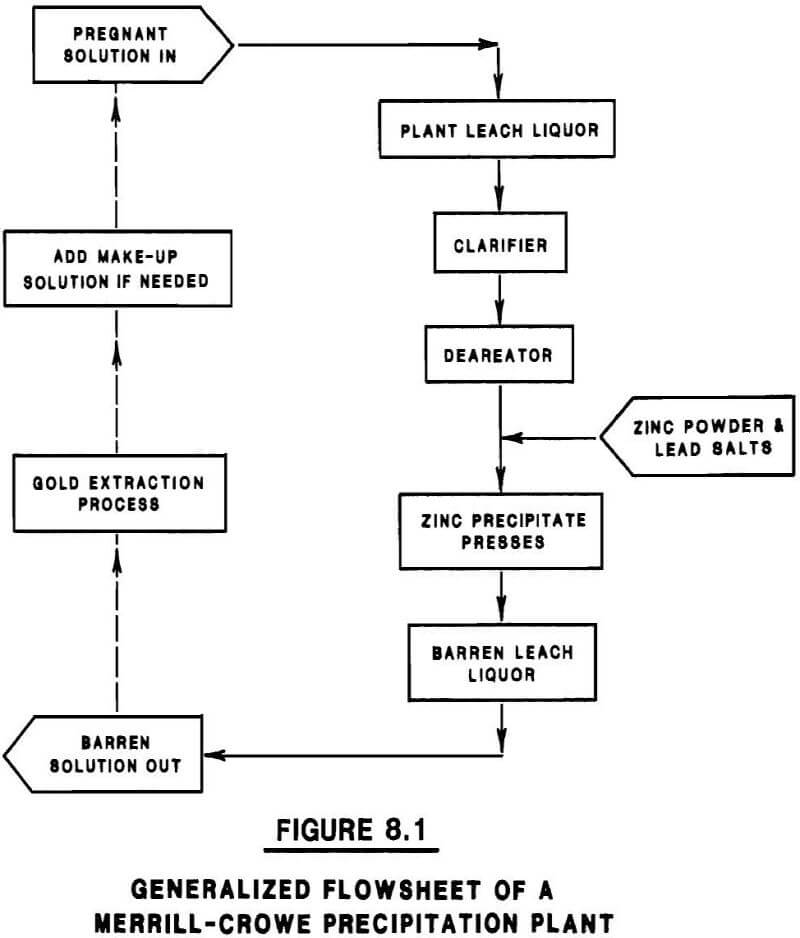
Flowsheet. Merrill-Crowe zinc precipitation is the most commonly used cementation process for gold recovery, and consists of four basic steps (Figure 8.1):
- Clarification of the leach liquor;
- Deareation;
- Addition of zinc (and lead salts); and
- Recovery of Zn/Au precipitate.
The precipitate is then smelted to recover the precious metals.
Solution Characteristics for Efficient Precipitation. Three types of solution constituents (or potential constituents) can have a major effect on the efficiency of the zinc precipitation process: suspended solids, dissolved oxygen, and interfering metallic ions.
Suspended solids can drastically reduce process efficiency, thus making clarification (i.e., the process of removing suspended material from a solution) the most important single factor in obtaining efficient precipitation. Through long experience, pre-coat pressure clarification is know to provide the best operational results. When this method of clarification is followed, partial removal of dissolved oxygen has already been achieved when the solution enters the Crowe receiver (or vacuum tower), where the deoxygenation is completed. By clarifying and deaerating simultaneously, available vacuum is used more efficiently.
Efficient and complete precipitation of metals from cyanide leach liquors requires that the solution, after clarification, be further conditioned by the removal of dissolved oxygen. Even minute traces of dissolved oxygen have a deleterious effect on complete precipitation because of passivation of the zinc surface. Vacuum deareation removes dissolved oxygen from solution and appears to be the best process because it also removes carbon dioxide. Carbon dioxide can react to form calcium carbonate and blind precipitate filters.
When metallic zinc dissolves in alkaline cyanide solutions, hydrogen is formed. Provided the dissolved oxygen has been substantially removed (generally through vacuum dearation, as discussed above), the hydrogen will combine with and nullify the effect of any oxygen remaining in solution. Zinc and cyanide consumption then decreases and the formation of troublesome byproducts is minimized.
Several metallic ions are known to have varying degrees of detrimental (i.e., interfering) effects on zinc cementation. The most troublesome ions are those of antimony and arsenic. Concentrations of these ions as low as 1 ppm can reduce the rate of cementation by 20 percent. High values of these ions may be reduced by treating the mill solution with sodium sulfide and filtering the resulting insoluble antimony and arsenic sulfides. However, all silver in the mill solution is lost when this process is used.
The presence of copper in cyanide solutions usually causes high cyanide consumption, and also decreases precipitation efficiency by passivation of the zinc, if allowed to accumulate beyond certain limits. However, with the use of powdered zinc, fresh surface is exposed on a continuous basis, and hence gold
and silver precipitation is usually complete. Maintaining higher free cyanide concentration in the precipitation circuit normally keeps the copper from precipitating, thereby reducing passivation of the zinc surface.
The presence of small amounts of lead in solution has been found to be beneficial to precipitation. However, if lead concentrations are too high (i.e., greater than 20 ppm), subsequent consumption of zinc is increased.
| Summary Of Advantages And Disadvantages Of Carbon Adsorption And Merrill-Crowe Precipitation | |
| MERRILL CROWE | |
| ADVANTAGES | DISADVANTAGES |
|
|
|
|
|
|





