- To participate in the 911Metallurgist Forums, be sure to JOIN & LOGIN
- Use Add New Topic to ask a New Question/Discussion about Hydrometallurgy.
- OR Select a Topic that Interests you.
- Use Add Reply = to Reply/Participate in a Topic/Discussion (most frequent).
Using Add Reply allows you to Attach Images or PDF files and provide a more complete input. - Use Add Comment = to comment on someone else’s Reply in an already active Topic/Discussion.
Cyanide Consumers in Gold Leaching Process (4 replies and 1 comment)
Hello Nathan, for a laboratory to do mineralogical analysis and gold metallurgy testing you can contact, for example, brad AT basemetlabs.com
I will venture here and I hope others do join me.
A 6% to may 16% recovery drop can pay for a lot of testing and equipment to "fix" or reduce the impact on your recovery and NaCN costs.
Yes, sulphides will act as cyanicides and consume cyanide in your CIL. Does you feasibility studies indicate how much you were expected to deal with? What species? What size/tonnage is your plant? What are the Au g/t feed grade?
Do you have greater than say 25% GRG gravity recoverable gold in your ore? If so, do you have a gravity circuit? Maybe you'd be lucky and you have GRG in your sulphides. This may hold the recovery but not solve the cyanide dosage problem.
If this is the future of you mine's ore, you should consider the insertion of a flotation circuit prior to CIL.
You may be able to direct sell that flotation concentrate or roast and leach or regrind/leach ... whatever the testwork indicates.
Here is an example of sulphide rich treatment methods https://www.911metallurgist.com/blog/gold-ore-processing-methods-gold-extraction
It may result in https://www.911metallurgist.com/blog/flash-flotation-whole-ore-cyanidation-leaching
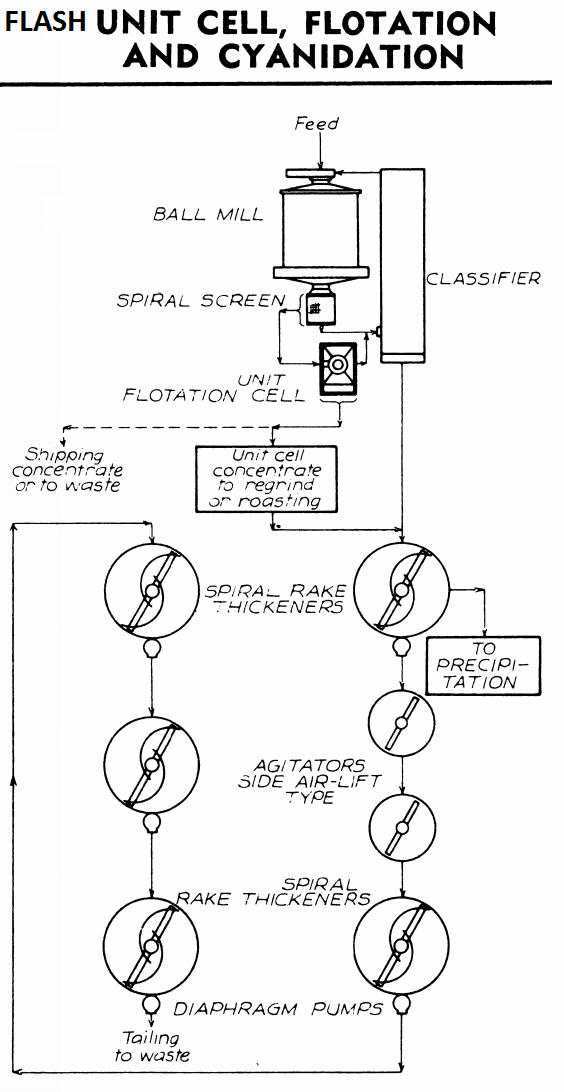
OR https://www.911metallurgist.com/blog/cyanide-processing-equipment-plant-configuration
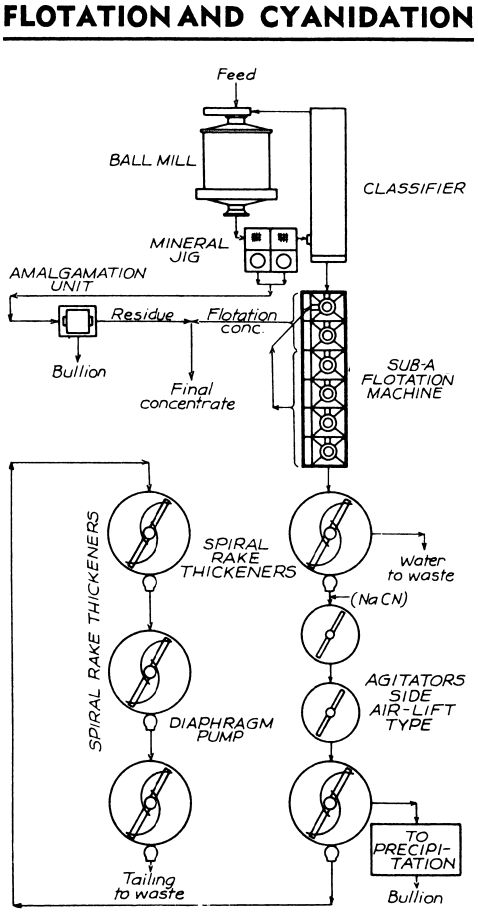
In any case, test and you'll see.
Use the Social Share Bar on the Left. Tell everyone you can about https://www.911metallurgist.com/metallurgy/ It's FREE & GOOD.
Hi Nathan
The short answer to part 1 of your query - yep, afraid sulfides will do exactly this.
Most metal sulfides will decompose quite happily in the typical cyanide leach environment to form metal ions, metal oxides or metal cyanide complexes and a variety of sulfur compounds like thiocyanates and thiosulfates. Pyrite is a big culprit here - if you follow the stoichiometry of the reaction, an ore with 1% pyrite will consume 32.6 kg/t of NaCN (and 2.7 kg/t O2, by the way).
2MS + 2(x+1)CN- + O2 + 2H2O 2M(CN)x(x-2)- + 2SCN- + 4OH-
You might have more copper or other cyanides in the ore too - which leads on to the next question. You need mineralogy to figure out what the heck is going on here - and the answer to part 2 of your question depends very much on where in the world you are located? I think there are a few mineralogists active on these forums who might be able to help with recommendations.
Nathan, review Cyanidation and concentration of gold and silver ores, by John Dorr
Usually, a high consumption of cyanide and low gold extractions are related to the presence of oxide copper minerals because they consume cyanide. Secondary copper sulphides can be dissolved by cyanide. If the total copper content is higher than expected, it is important to consider sequential copper analysis to assess the presence of the different copper minerals. The presence of other minerals have an impact on the metallurgical performance.
Arsenic and antimony are present as arsenopyrite, realgar, orpiment and stibnite. In many gold deposits, if gold is associated to arsenopyrite, there is a serious problem when gold is finely disseminated in the crystalline structure and the liberation of gold particles could be difficult. The presence of stibnite, realgar and orpiment is a problem due to react with cyanide at pH values higher than 10. These minerals are oxygen and cyanide consumers and the products formed during the reaction with cyanide may passivate the surface of gold and retard its dissolution.
Carbon can be present as carbonate mineral and inorganic material. The last one ranges from graphite, which is a poison from a treatment consideration, to the named carbonaceous matter that has near of eighty percent of carbon and consists of active element carbon, hydrocarbons and organic acids. This type of carbon is problem when the ore must be treated by cyanidation because they cause the called effect of pre-robbing. This phenomenon consists in absorbs the free cyanide and gold cyanide complex from cyanide leach solutions.
The presence of arsenopyrite and pyrrhotite can be an issue if the process selected is flotation and cyanidation due to the high consumption of oxygen. In this case special pre-treatments before cyanidation can be included in order to minimize this problem. The presence of pyrite tend to be a problem.
Clay minerals are troublesome in some gold ores causing high consumption of cyanide, also high viscosity slurries that are difficult to settle. These problems occur when some organic compounds are present and it is necessary a pre-treatment or identify its presence to avoid problems.
It is important to get information from ICP analysis and compare the information used during the engineering studies. In this it is possible to study the possible differences in the being treated in the CIL circuit. Obviously, the mineralogical information is important. Try to perform QemScan or MLA studies to get information on the gold bearing minerals, gold particle size, gold associations, and gangue minerals. Some operations add oxygen to increase the gold extraction because the ore contents certain content of sulphides. Evaluate the effect of oxygen on gold extraction should be part of a metallurgical testing program
Where is the best location to add oxygen (from hydrogen peroxide)? Can it be added at the head of leach tanks? Or into leach tanks?
Please join and login to participate and leave a comment.

Hello All, I work in a CIL plant, processing oxide ore with more and more intrusion of sulphides (fresh ore body). The plant is designed for oxide ore but we noticed that at a certain blending, the transitional ore could be processed. It impacted on the gold recovery (going from 83-84% for pure oxide to around 76-78% for Oxide-transition blending) but this was within an acceptable range. The problem is that going deeper (more transition and less oxide), the recovery has dropped up to 65%, the parameters are still almost the same (CN-200 ppm, pH-10.5 and DO-15ppm) but the cyanide consumption has drastically increased (from 0.45 kg/t to 0.75kg/t)