Gases, Drying and Absorbing.—For this purpose U-tubes, potash bulbs, calcium chloride tubes and calcium chloride jars are used. U-tubes, Fig. 103, are made in a number of forms and are suited to drying gases by the use of calcium chloride, anhydrous copper sulphate, sulphuric acid, etc. Figs, a 103 and b 103 are good forms where the gas has merely to be dried or purified and the form shown in Fig c 103, with glass-stoppers, is to be preferred where the gas has to be caught and weighed. The other forms can also be used for this purpose. In this case, the tubes should be capped when they are not connected in the train
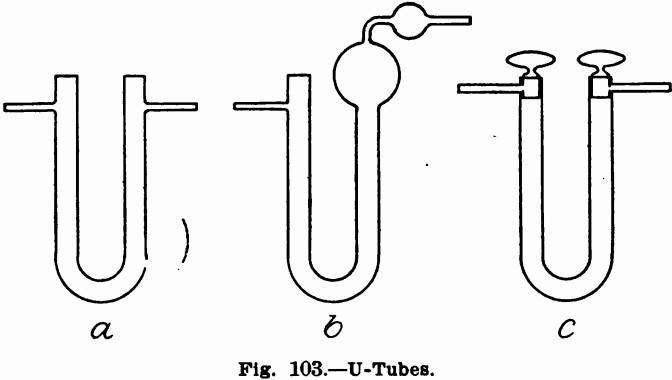
by means of small pieces of thin walled rubber tubing, one end of which are closed by bits of glass rod or, better still, capillary glass tubing. To suspend tubes to be weighed, use fine platinum wire. U-tubes, in which liquids are to be used, should be filled with glass beads or cracked up pumice-stone, and should contain only sufficient liquid to cover the bend. Where dry substances are used, plugs of loose absorbent cotton should be placed on top of this, in both branches of the tube, to prevent fine particles from being carried out of the tube. Where tubes are stoppered with corks the ends containing these should be dipped in melted paraffin or wax, so that all the cork and some of the end is covered to prevent escape of the gas.
Calcium chloride tubes are shown in a and b, Fig. 104. They may be obtained either with one or two bulbs. The form shown in b, Fig. 104, has a small tube extending into the bulb and is intended to catch some moisture (as water) by condensation in the first bulb, and so save the calcium chloride. These tubes are seldom weighed and are usually used as guard tubes. The calcium chloride jar is shown in Fig. 104 c. It is never weighed and is usually used only to purify or dry air or gases. A small pad of asbestos fibre or cotton is placed at the constricted point just above the tubulature and the purifying substance on this. They may also be used for liquids by filling the upper portion with glass leads and drenching these with the liquid. They may be used for both liquids and solids by having the lower part of the tube filled with liquid nearly to the tubulature and having the tube by which
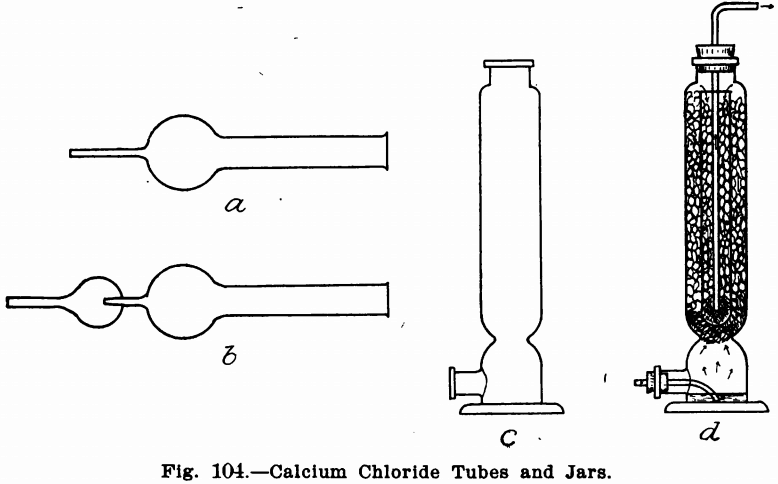
the gas enters bent so that the latter will have to bubble through this. Fig. 104, d, shows a very powerful purifying apparatus made from a calcium chloride jar and a long narrow test tube. The lower part is filled with sulphuric acid (caustic potash for CO2) and the upper part and test tube with calcium chloride (soda lime for CO2). The test tube rests on a plug of loose cotton and the gas is led out of the apparatus by means of the tube as shown. In this apparatus, the gas has to travel the entire length of the jar and also the test tube, as shown by the arrows.
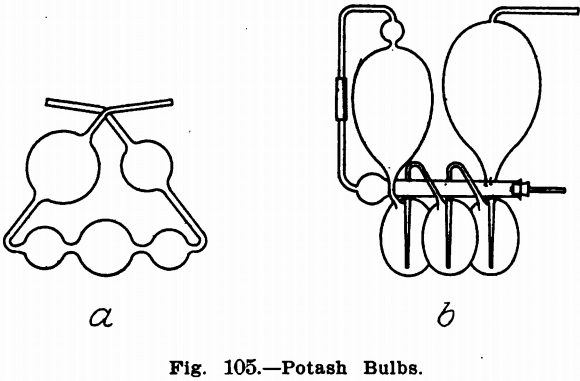
Potash bulbs are for use with liquids only, though there is usually attached to them when they are to be weighed a tube containing calcium chloride or soda lime. The most common forms are shown in Fig. 105. Where the bulb is to be weighed the form marked b is best suited, or the form a may be weighed together with a U-tube for a guard. When not connected up in the train they should be capped as directed for U-tubes. To fill the bulbs, attach a short piece of rubber tubing to one end and dip the other in the solution held in a small shallow dish. Apply suction to the rubber tubing with the mouth until the bulbs are filled to the proper height. Then wipe the tube dry inside and out with pieces of filter paper or a soft cloth.
Gas may also be purified by allowing it to bubble through solutions held in 4-ounce bottles.