Recovery Of Precious Metals From Electronic Scrap
De-Silverizing Bullion

The de-silverizing of base bullion is carried out in accordance with the principles of the well-known Parkes process. The base bullion produced by the blast furnace department carries (apart from silver values) impurities, the chief of which are copper, antimony, and arsenic. In order to more clearly indicate the grade of the base bullion, the […]
How to Make Gold
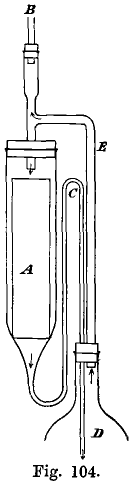
The purest gold obtainable is required for use as standards or check pieces in the assay of gold bullion. The following method of preparing it is now in use at the Mint. Gold assay cornets from the purest gold which can be obtained are dissolved in nitrohydrochloric acid, and the excess of nitric acid expelled […]
Gold Inquartation Process

The bead of silver and gold obtained by cupellation is squeezed between pliers, or flattened by a hammer on a clean anvil, to loosen the bone ash adhering to its lower surface, and is then cleaned by a brush of wires or stiff bristles. It is then weighed, the silver removed by solution in nitric […]
Cupellation Furnaces for Precious Metal Assaying

Cupellation is conducted in a muffle furnace, the construction of which is shown in Figs. 62-63. The fire is lighted, a little bone-ash is sprinkled on the floor of the muffle to prevent its corrosion by litharge in case of the upsetting of a cupel, and the cupels introduced as soon as a bright red […]
Precious Metals Refinery Security

“Highgrading” a polite word for stealing gold and silver for your refinery, has been a way of life since the metals in native form have been mined or produced. Unfortunately, in the past, highgraders were not always severely prosecuted or even looked upon unfavorably in their communities. Recent high metal prices with attendant world wide […]
Fire Assaying; a Miniature Smelting Process

Fire assaying, in essence, is a miniature or small scale smelting process which recovers and reports the total gold content of the assay sample, including gold combined with other elements or locked in the ore particles. Because of this, a assay may report values that cannot be recovered by placer methods and it cannot be […]
Gold Refining Process by Aqua Regia
The Gold Refining Process by Aqua Regia was introduced at the Pretoria Mint after the Miller process had been tried and abandoned owing to the alleged difficulty of treating the gold bullion extracted by the cyanide process. In the aqua regia process the gold is dissolved and precipitated. It is made very difficult if the […]
Purification of Gold by Electrolysis
The Moebius Process of Purification of Gold by Electrolysis is now in successful operation and is said to be specially suitable for refining copper bullion containing large proportions of silver and gold with small quantities of lead, platinum, and other metals, but is chiefly used in parting dore silver containing not more than 20 per 1,000 […]
Gold Refining Machine

The guard pot, with the clay pot in it containing 2 or 3 ozs. of fused borax, is placed in the furnace, and is heated gradually until the bottom of the clay pot is dull red. The ingots (of which the larger are slipper-shaped) to be refined, amounting in all to 650 to 720 ozs. […]
