FORGE 4-TAP HYDRATION STATION – Portable Water Dispenser
An Industrial Hydration Station for Remote Mining Operations Ensuring reliable access to clean, cold drinking water is a critical safety requirement on modern mining sites. The FORGE 4-TAP HYDRATION STATION from Forge Water Systems is a purpose-built hydration unit engineered specifically for industrial, remote, and high-demand applications where durability, sanitation, and uptime matter most. Constructed from […]
Different Crusher Alloys
Motor Crusher
Are you looking for a motor crusher or a crusher motor? This difference is significant. The first is if you want to crush motors, while the second is in case you are looking for a motor to power up a crusher. Here is the answer to either option: For crusher motor, you can go to […]
Can I use Seawater in my Greenhouse
How do Seawater Greenhouses Work? A seawater greenhouse is a type of greenhouse technology that utilizes seawater to grow crops in arid or desert regions. It combines two main elements: the greenhouse structure and the seawater desalination process. Here’s how a seawater greenhouse typically works: 1. Greenhouse Structure: The greenhouse is designed to provide a […]
What is the Role of Water in Lithium Mining
Balancing Resource Extraction and Water Sustainability Lithium, a key component in batteries that power electric vehicles and store renewable energy, has become increasingly vital in our pursuit of a sustainable future. As demand for lithium rises, so does the importance of understanding and addressing the environmental impact of its extraction. One critical aspect of lithium […]
Direct Lithium Extraction (DLE)
Direct Lithium Extraction (DLE) is an innovative method of extracting lithium from brine or other lithium-rich sources without the need for traditional evaporation ponds. Unlike conventional lithium extraction processes, which rely on the natural evaporation of water to concentrate lithium, DLE techniques offer a more efficient and environmentally friendly approach. The traditional lithium extraction process […]
Diamond Drilling
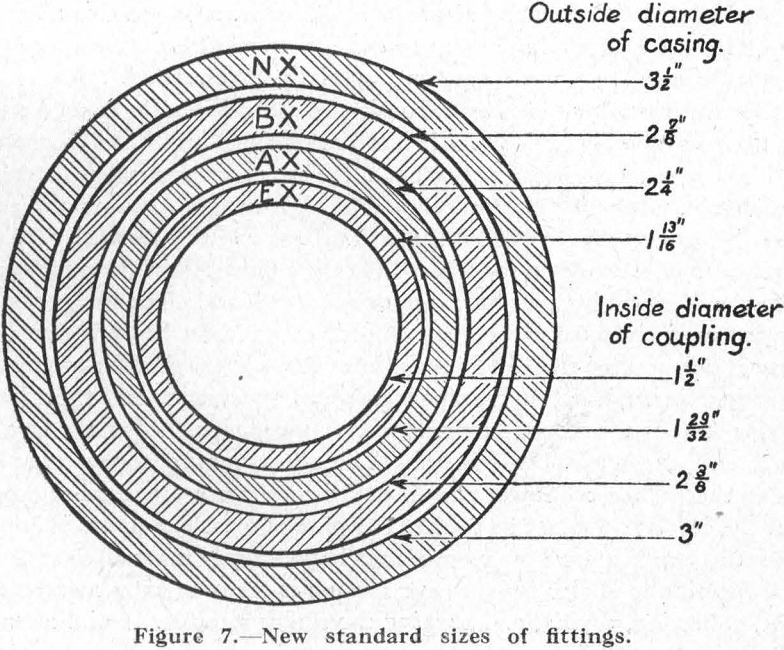
In approaching the subject of diamond-drilling practice, a brief history of the origin and development of the diamond drill may be of interest. The use of black diamonds for boring holes in rock was discovered in 1863 by Rudolph Leschot, a French engineer. In 1864, he employed a diamond bit to bore holes for blasting […]
Geology
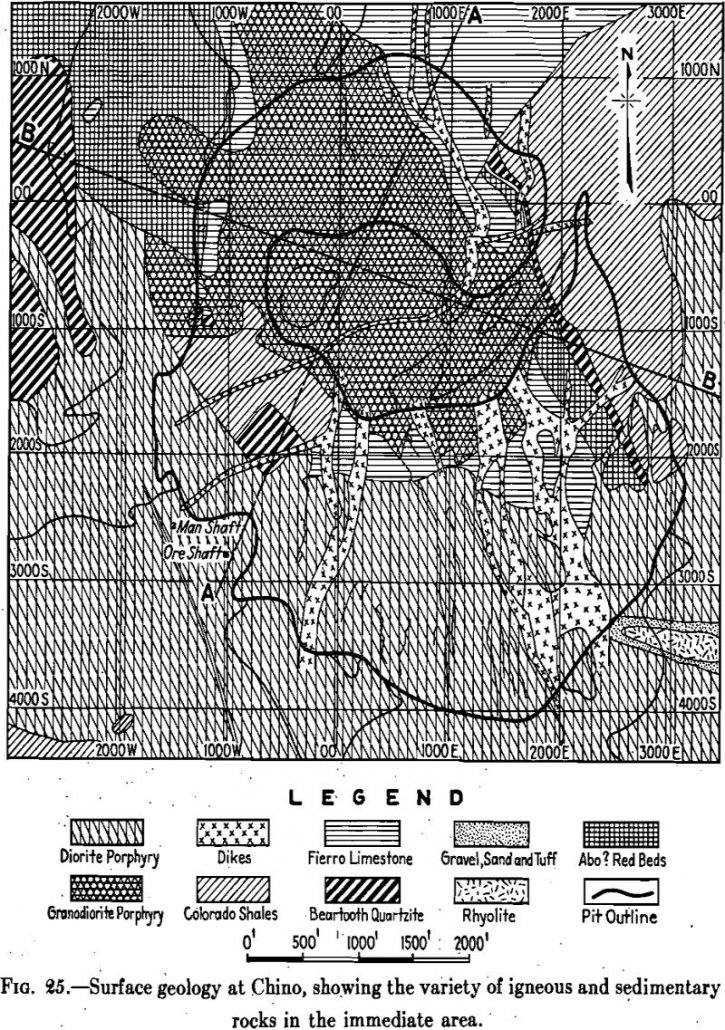
Frequently the careful engineer puts the word porphyry in quotation marks or precedes it with “so-called,” when he writes of the Porphyry Copper mines. This is done by way of serving notice that he is aware that the term is not a precise one, though no one who is informed on the subject would in […]
Metallurgy Problems
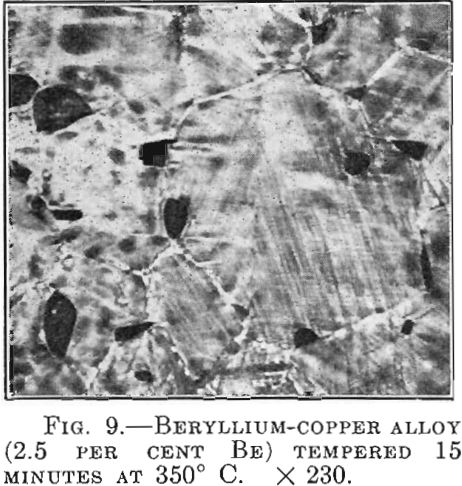
While reflecting on the subject of my present paper, I soon realized that it would be very difficult to say something really new about any special aspects of metallurgy. In consideration of the excellent scientific work that has been done in the United States in connection with almost all the fundamental questions that are of […]
Hydraulic Riffles VS Hungarian Riffles
In-depth research on gold recovery in the Yukon conducted in 1990 by Randy Clarkson concluded that the “reliability, inexpensiveness and operating simplicity of sluice-boxes are hard to beat”. The same still applies, although sluice design has progressed since. Author: Jonas Boehnke Shop for a Gold Wash Plant with Hydraulic Riffles here Water injection into the […]
