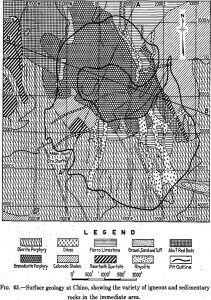
Geology
Frequently the careful engineer puts the word porphyry in quotation marks or precedes it with “so-called,” when he writes of the Porphyry Copper mines. This

Frequently the careful engineer puts the word porphyry in quotation marks or precedes it with “so-called,” when he writes of the Porphyry Copper mines. This

While reflecting on the subject of my present paper, I soon realized that it would be very difficult to say something really new about any
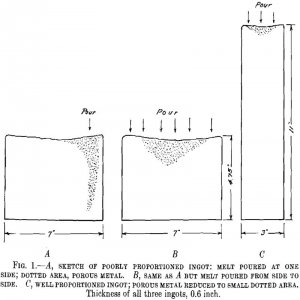
The problem of scrap is probably of greater importance in the production of gold, silver and other precious metal alloys than is the case for
In-depth research on gold recovery in the Yukon conducted in 1990 by Randy Clarkson concluded that the “reliability, inexpensiveness and operating simplicity of sluice-boxes are
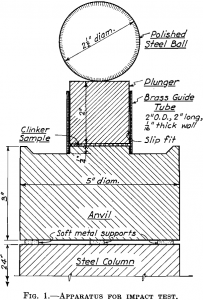
Grinding costs are an important item in cement manufacture, and the cost of power is one of the large items in grinding costs. Even where

The Freeport Sulphur Co. built a portable screw crusher in 1944 that solved a problem that none of the crusher manufacturing companies contacted were able
Safety Lift Table unit using minimum 1.5 tonnes lifting equipment Use marked balanced lifting points Ensure drive is isolated before guard removal Ensure unit is

Here is my list of the Top 15 largest and biggest mines in the world: They are economically big and physically large, taking a
Jaw crushers include Superior, Type “B” Blake, Fine-Reduction, and Dodge — sizes, 4 by 6 to 84 by 66 inches. A reciprocating machine, the crushes
As it is malleable, no crusher can crush gold but, all rock crushers can crush rock with gold minerals in it. Small gold prospectors are