Zinc Electrowinning & Zinc Cathodes
Commercial production of zinc by electrolysis began in 1881 following a patent by Letrange of France. The process has come down to the present day essentially unchanged and consists of three basic steps: Leaching of roasted zinc concentrates in dilute H2SO4; for example, ZnO + H2SO4 → ZnSO4 + H2O. Purification of the resulting zinc sulfate […]
Platinum Electrocatalysts for Oxygen Reduction
This report describes possible material application as oxygen electrode catalysts. The ultimate objective was to assess the potential of abundant, less costly materials as substitutes for platinum, or to increase the efficiency of platinum when used as an oxygen electrocatalyst. Presently, imports account for essentially all of the U.S. requirement for primary platinum metals. The […]
Zinc Electrolysis
Zinc Electrolysis was studied to help maintain an adequate domestic minerals base, the Bureau of Mines is investigating an aqueous chlorine-oxygen leaching procedure to produce ZnCl2 from sulfide concentrate and is also studying a fused-salt electrolytic technique to produce zinc metal from ZnCl2. Several chlorine leach/electrolysis methods for treating zinc ores and concentrates have been […]
Electroslag & Electric Arc Furnace Processes
Demand for pressure vessel steels for use in power generation and energy transport applications is increasing in the United States. Coupled with the increased demand is the requirement for higher quality steels that are better able to withstand temperature extremes and mechanical stresses, and that possess greater tensile ductility, impact strengths, and isotropy. One important […]
Electrorefining Copper
This report discusses Electrorefining Copper research conducted to increase the rate, deposit purity, and overall efficiency in industrial processes used to electrodeposit and purify metals by increasing substantially the operating current density range and optimizing the deposition process. Traditionally, copper refineries operate electrolytic cells at a current density of 20 amp/ft². At this current density, the […]
Recovery of Molybdenum and Rhenium from Mo Concentrate
As part of the goal to maximize minerals and metals recovery from primary domestic resources, design factors associated with minimizing current leakage in bipolar cell configurations were studied by the Bureau of Mines as a means of improving the efficiency of bipolar electrooxidation cells. Initial studies that were conducted in a small bipolar cell operating […]
Copper Electrolytic Refining Process Explained
Experiments on a Porphyry Copper Ore: This research was done partly in the non-ferrous laboratory of the Department of Metallurgy of Columbia University, under the direction of Dr. Edward F. Kern, and completed elsewhere. Acknowledgment is due to Prof. Arthur L. Walker, Dr. Edward F. Kern and Dr. William Campbell of the Department of Metallurgy, for their […]
Electricity with Copper and Zinc Sulphate: Daniell Cells
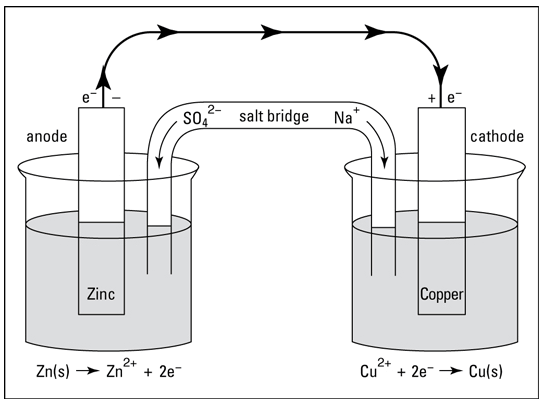
https://www.dummies.com/education/science/chemistry/electrochemical-cells-the-daniell-cell/
Copper Precipitation Methods

In a leaching process, having obtained the copper in solution, the choice of the precipitation method is influenced by the following factors: Availability of precipitant. Adaptability to the leaching process. Final product desired. Regeneration of leaching solutions. Fouling of solutions. There are three general classes of copper precipitation methods: 1. The copper precipitation by use […]
Precipitating Gold & Silver Using Metallic Minerals

While the reducing action of organic matter, of ferrous sulphate, and of hydrogen sulphide has frequently been invoked to account for the deposition of native gold and silver from ore-forming solutions, the high efficiency in this respect of certain of the metallic minerals which form the ore itself has not been sufficiently recognized. If a […]
