Radioisotopes Applications
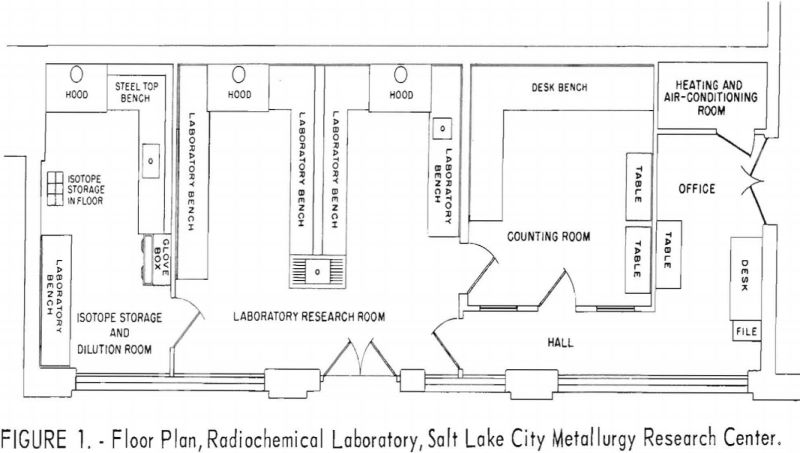
Many factors contributed to the decision to build a radiochemical laboratory at the Salt Lake City Metallurgy Research Center to investigate the application of radioactive isotopes and chemical compounds in extractive metallurgy. These reasons included the notable success achieved in biological sciences with radioisotopes, the development of reliable instruments and methods for identifying the isotopes […]
CUTTING MILL
Our line of cutting mills includes models which reliably process: soft, medium-hard, elastic, tough and, fibrous materials. All models are based on the same grinding geometry and feature. Depending on which model, our cutting mills may feature: Variable speed and adjustable in a range from 700- 3000 revolutions per minute A fixed rotational speed of 1500 […]
Mercury Detector
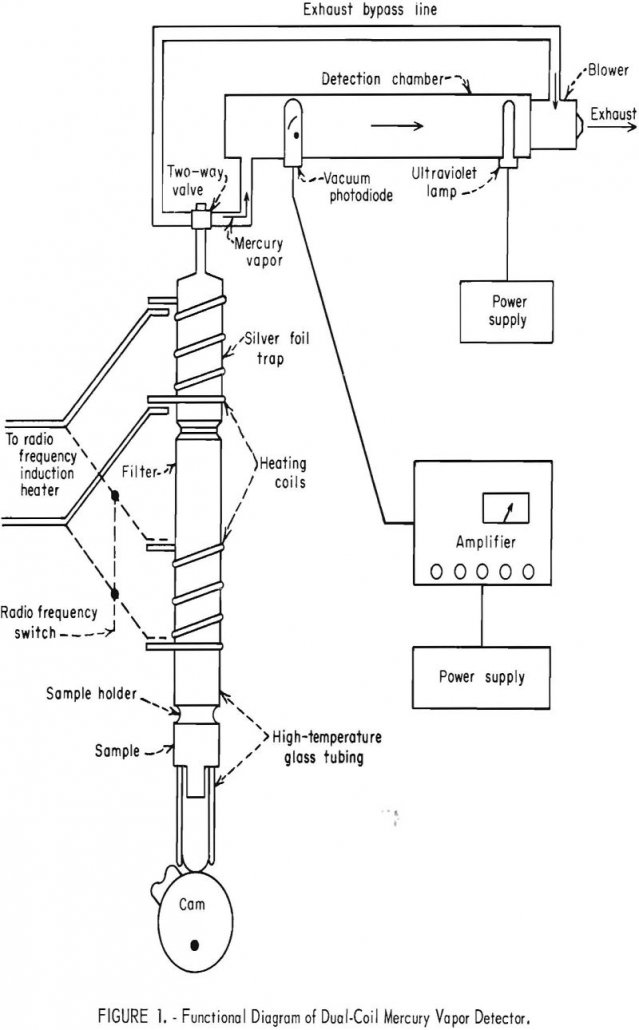
The Bureau of Mines recognized the need for a high-sensitivity mercury detector years ago and undertook the development of such an instrument at the Denver research facility. Although the detector has been used primarily in mining applications, the instrument could readily be adapted for use in pollution measurement. The amplifier circuit is designed for use […]
Drop Weight Impact Tester
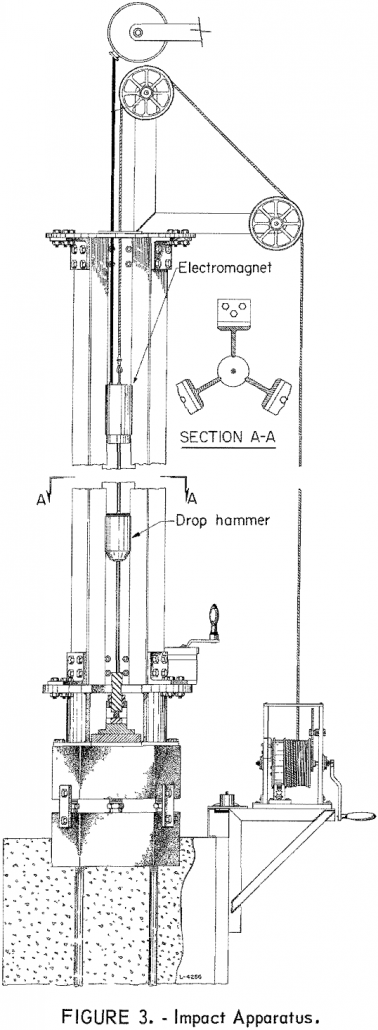
For many years, the impact test has been one of several techniques used to determine explosive sensitivity. This test has provided a useful index having practical value; however, it provides only an ordering of the sensitivities of a series of materials and, because of a lack of standardization in apparatus design and test conditions, comparison […]
Permanent Magnets

The magnetic properties of rare earth-cobalt intermetallic compounds have been studied by various investigators. It was not until Strnat and Hoffer investigated single crystals of YCo5 and reported an extremely high magnetocrystalline anisotropy that systematic studies of the magnetic properties of rare earth-cobalt alloys were undertaken. These studies determined that RCo5 compounds (R = one […]
Chemiluminescence Detector
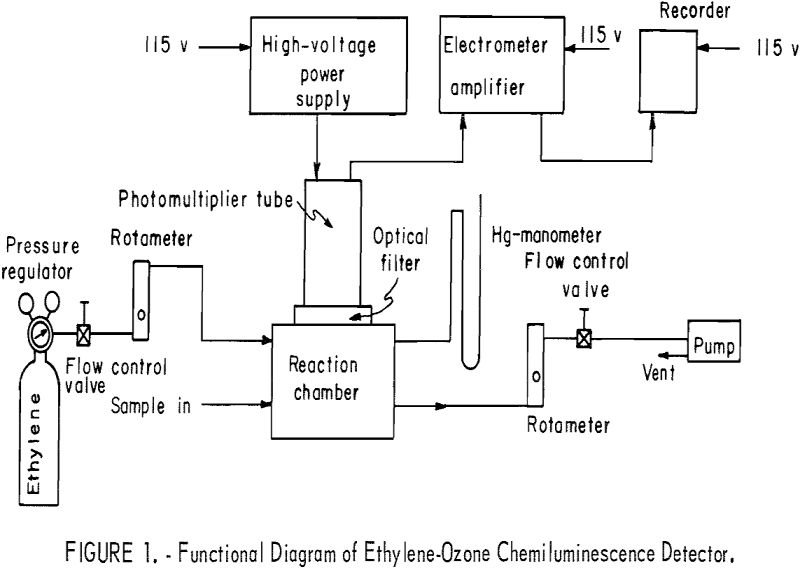
Instruments currently in use for routine measurement of ozone in the atmosphere are based on ultraviolet spectrometry, coulometry, or colorimetry. Studies have shown that none of these techniques is completely satisfactory for routine ozone measurement, either because the instruments require an inordinate amount of attention or because they lack the requisite specificity to ozone or […]
Gold Redox ORP Probes used in Flotation Circuit

Three gold sensing electrodes were made from 3 mm gold rod. The rod was cut with a diamond wafering saw into 3 mm lengths, soldered to insulated copper wire, placed inside 9 cm lengths of 1 cm (outside diameter) Plexiglas tubing, and cast inside the tubing with epoxy. After the epoxy was set, the gold […]
Cryogenic Plant at Mining Metallurgical Processing Plants
In the 1940’s a top blown water-cooled oxygen lance was developed to inject oxygen into liquid iron. This process, the LD or BOF process technique, today is used in 65% of the world’s steel production, and represents the largest oxygen market for temperature enhancement in steel and non-ferrous smelting applications. There are three major approaches […]
Zirconium Crucibles
Zirconium crucibles are rapidly becoming a standard piece of equipment, particularly in laboratories where fusions using strongly alkaline fluxes are common. This Center has found no better material to use for the thousands of sodium peroxide fusions that are done here every year. Zirconium crucibles usually arrive from their suppliers in a polished, silvery state. […]
Bruker Handheld XRF
This is a general safety information regarding Bruker’s handheld X-ray fluorescence XRF analyzers. Proper training is vital for compliance, safe operation and understanding of the operator’s responsibilities associated with handheld XRF analyzer use. The term radiation is used to describe forms of energy emitted as particles or electromagnetic waves. There are two types of radiation: […]
