ASSAYING CHLORINE BY PRECIPITATION WITH SILVER NITRATE
When we consider the ordinary gravimetric precipitations, it is evident that if the exact strength (as regards precipitation) of the precipitating reagent Apparatus, Reagents.—Apparatus as usual. For the preparation of the standard solution, pure silver nitrate (preferably triple crystallized) is required. For practice in this analysis the student may first estimate the chlorine in pure […]
Electrolytic Analysis
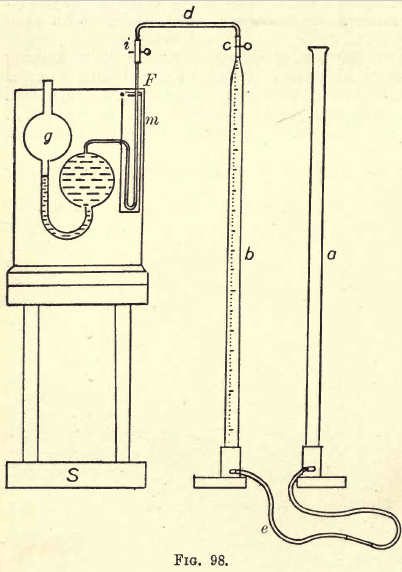
The theory of electrolysis cannot be taken up at any length here, and the student desiring fuller information on this subject is referred to the works of Ostwald, Nernst, James Walker, and others. Briefly, it may be stated that on passing a current of electricity through a solution of a salt in water the salt […]
What the Effect of Varying Furnace Temperatures

The Wind or Melting Furnace Principle.—According to the equation 2PbO + C = Pb2 + CO2, 446 grains of litharge fused with excess of charcoal should yield 414 grains of metallic lead. If the temperature be too high, PbO volatilises, and this loss, which varies with the temperature and air present, can be checked by […]
Assaying Lead Ores

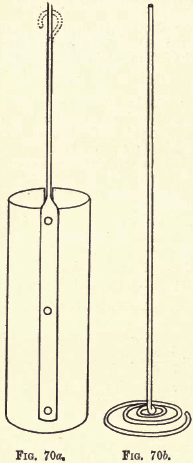
To determine by fire-assay the percentage of lead in: (a) The sulphide. Galena (PbS). (b) An oxidised ore containing Cerussite (PbCO3), Anglesite (PbSO4), etc. (a) The sulphide, Galena Methods employed. Regarding the methods employed authorities vary widely ; here two methods are given for sulphide ores, the Soda and Argol (or Nail) method, and the Cyanide method. The […]
ESTIMATE SILVER IN COINAGE
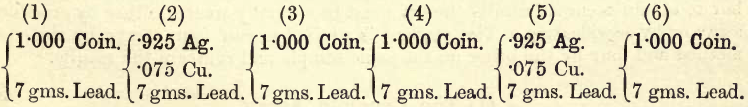
For practice, a convenient material to operate on is an English silver coin, which contains 925 parts of silver to 75 parts of copper by weight. The student, therefore, knows the composition of the alloy. When dealing with alloys of unknown composition the process is more complicated (see note appended). Method.—A given weight of […]
Fire Assay of Copper

This assay is presented here, not with the purpose of giving the student an accurate and handy means of estimating copper in its ores, but to give him some practice in manipulation, which, on the small scale, somewhat resembles metallurgical methods. In all modern mining fields the fire-assay of copper is replaced by the Volumetric […]
Water Analysis Methods

In this section eight chapters are given, each dealing with one or more technical methods of analysis. The typical technical method should be quick, accurate, and inexpensive. Accuracy may to some extent be sacrificed in favour of speed when the quantity and effect of such inaccuracy are approximately known, yet it should always be the […]
Chimney / Furnace Gase Analysis
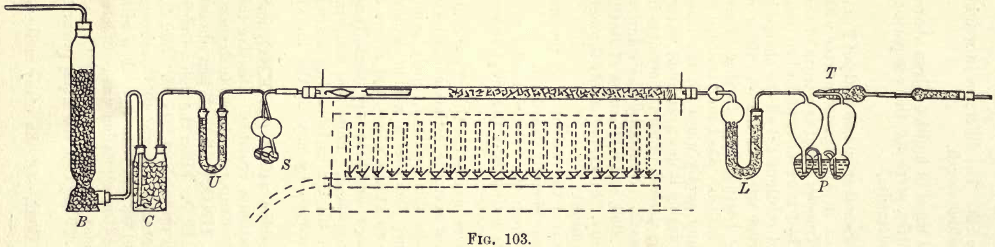
In chimney or furnace gases the estimations usually made are the percentages by volume of oxygen, nitrogen, carbon-monoxide, and carbon-dioxide. The more refined methods of gas analysis are too slow for technical requirements, and are replaced by the Elliott, Hempel, or other methods. The method here to be described is that of Hempel, and differs chiefly from the Elliott and others […]
Coal and Coke Analysis
For technical purposes the following estimations may be required: Moisture, Volatile Matter, Ash, and Fixed Carbon; Sulphur, and Phosphorus. Carbon and Hydrogen by ultimate organic analysis. The calorific (or heating) power of the coal. Besides the chemical analysis of cokes, there are required the “Crushing resistance,” “Porosity,” and “Specific Gravity.” For information concerning these tests, and […]
Chemical Composition of Cements

A suitable material for the student to operate on is the well known brand —Portland Cement. As the student has already examined in detail a silicate, the following notes are given somewhat briefly. A good cement should consist chiefly of SiO2 and CaO, with a little Al2O3 and Fe2O3, and less than 2% MgO, and […]
