Action of certain strains of bacteria has been implicated as a significant factor in the oxidation of ore deposits. Other bacteria are thought to be significant in the formation of syngenetic sedimentary ores and in supergene enrichment processes.
A normal soil/ground-water regime provides all of the necessary pH and Eh conditions for several groups of bacteria which influence the sulfur cycle in nature. Limiting concentrations of nutrients such as sulfate ions can determine whether a particular microbial species is active at a given place.
Sulfide producing bacteria (presumably sulfate users) were found in water from about 50% of wells sampled in the mineralized area and in about 30% of samples from the barren area. These bacteria ore readily detected and may provide a useful tool for regional geochemical reconnaissance. Bacterially produced sulfide ions can remove metals such as copper and zinc from solution thus complicating the interpretation of hydrogeochemical prospecting.
In our everyday experience we think of living forms (other than photosynthetic) as oxidising organic materials such as searches or proteins to produce primarily CO2 plus H2O and the energy necessary to get the creature about its daily tasks. Typically the energy available in this type of metabolism is substantial. Many possible reactions in nature produce too little energy to be utilized by higher forms of life such as man. For almost every potential source of energy, some form of bacterium has evolved that can release this energy and use it to sustain life.

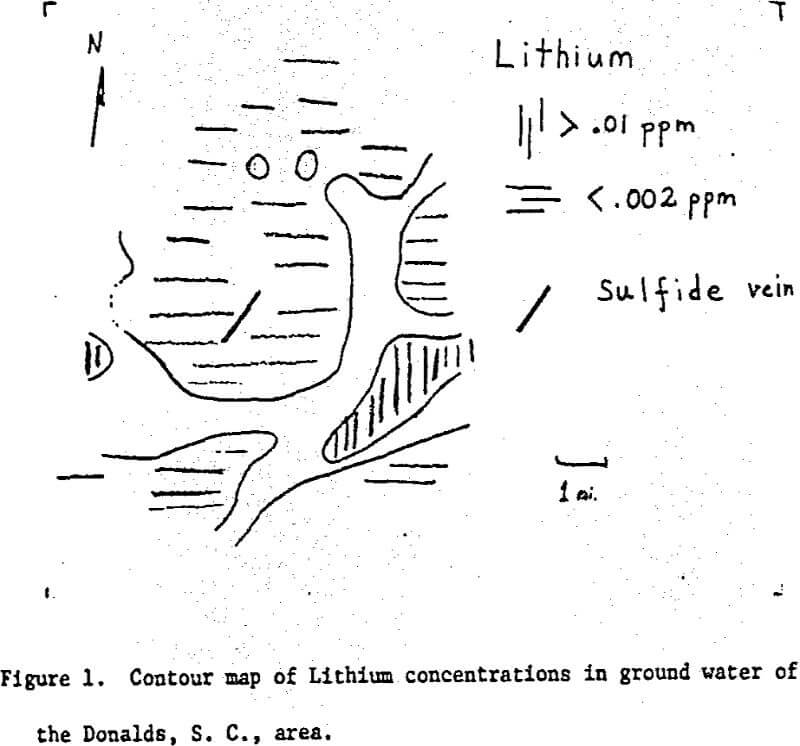
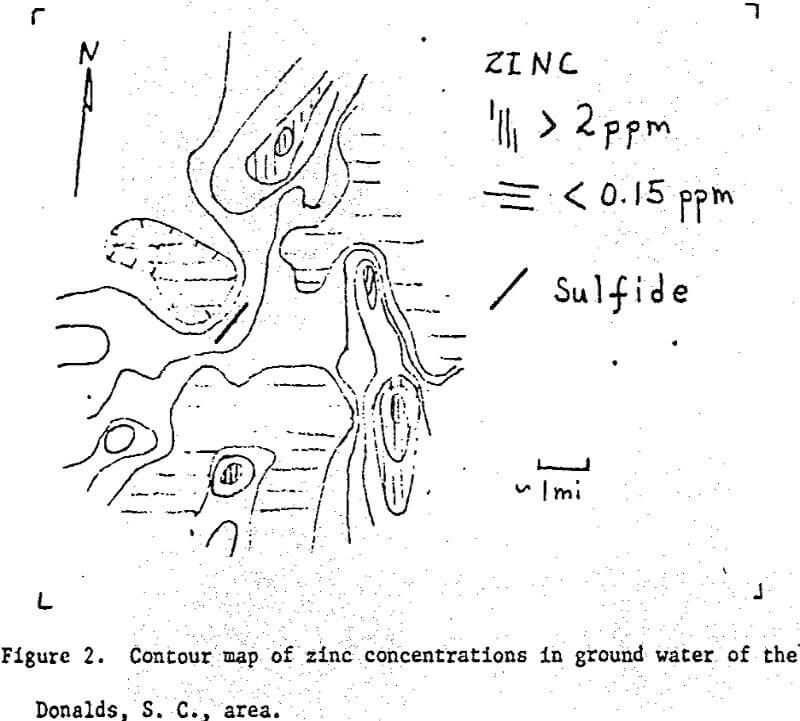
Over one hundred samples of well water were collected within approximately a five-mile radius of Donalds, South Carolina. These samples were analysed for Li, Na, K, Mg, Ca, Sr, Zn, Fe, and Cu by atomic absorption spectrophotometry. A crude geologic map was made as the samples were collected.
The zone of lithium-bearing ground water coincides with areas in which granitic rocks and pegmatites were noted in the field. No obvious relation between zinc highs and the sulfide mineralization were noted. It appears that the sulfide vein is surrounded by areas of low zinc concentrations.
Distribution cf copper shows less regularity than that of zinc. An attempt to use factor analysis to reveal obscure correlations in the analytical data indicates that the distribution of copper is essentially random, or is not closely related to the distribution of any other element studied.