Silica flotation with amine salts has been a commercial process for years in certain non-sulfide mineral systems. Phosphate rock and feldspar deposits are notable examples. Generally, the quartz is clean and coarse. Primary amines and imidazolines are used, usually in a mole weight range 250-300, and often fairly impure grades of collectors are satisfactory.
The synthesis of ether amines involves two basic chemical reactions. The first is the cyanoethylation of a long chain alkyl primary alcohol to yield an alkyl oxypropylnitrile adduct.
![]()
Hydrogenation of the nitrile during the second reaction reduces the nitrile to a primary amine.
![]()
An alkyl oxy-n-propyl 1,1,3-diamine (R-O-CH2CH2CH2NHCH2CH2NH2) is synthesized by reacting acrylonitrile with the above primary amine and repeating the hydrogenation step. This molecule is difunctional in that it contains one secondary amine group and a terminal primary amine group.
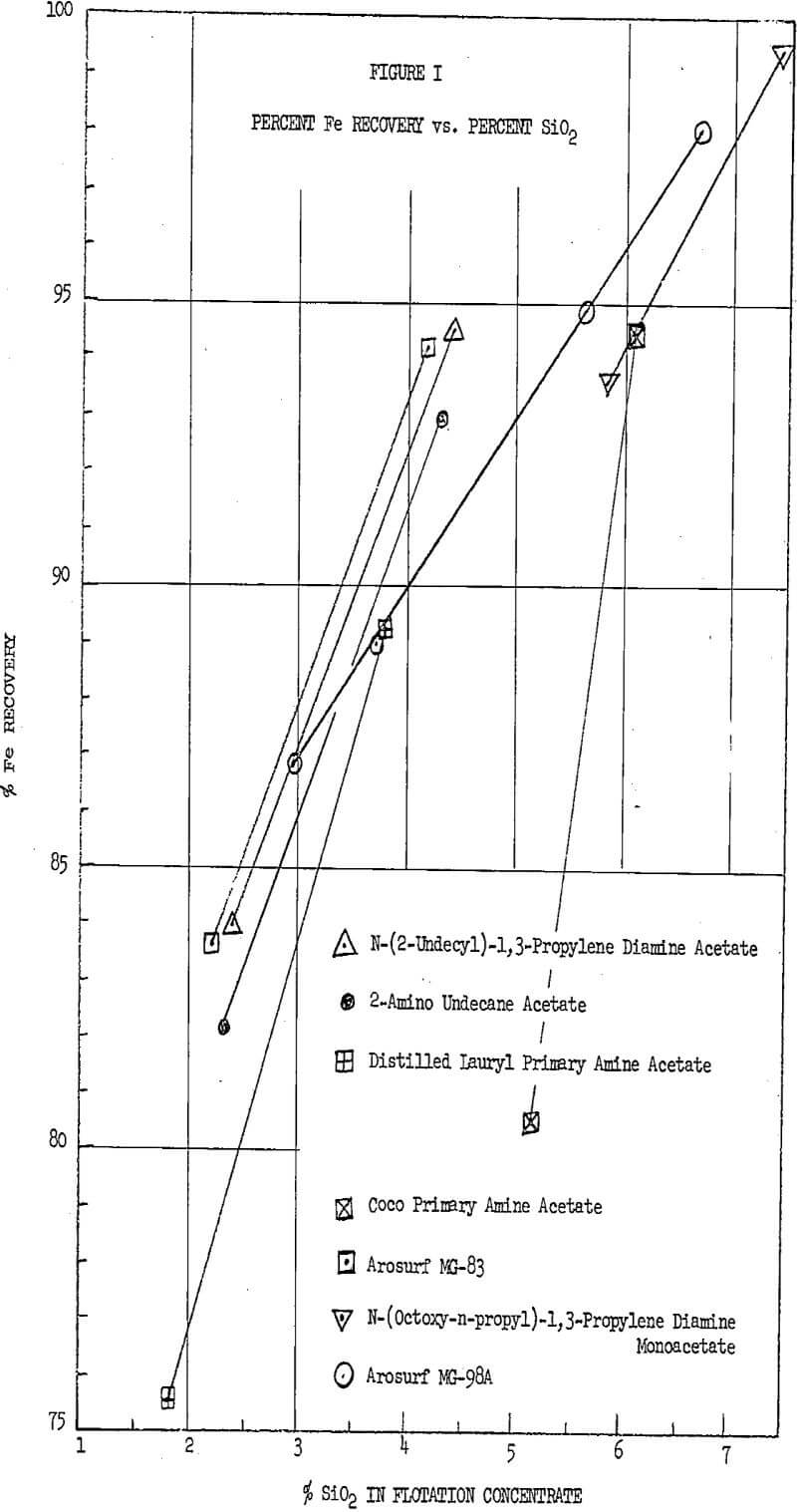
A series of flotation tests were run on a magnetic iron concentrate to compare the performance of ether amines against conventional aliphatic primary amines and beta amines. The beta amines, which are also relative new-comers as flotation reagents, were included because of their good reputation as selective silica collectors. Table I gives a detailed description of the reagents tests.


The bench scale flotation studies were run on a magnetic concentrate assaying 65.9% Fe with 96.3% passing through 325 mesh. The tests were conducted under neutral pH (7-8) conditions on 560 gram ore samples in a Denver laboratory cell. The ore was conditioned at about 25% solids in the flotation cell for about five seconds with the indicated amount of collector and 0.04 lb/LT of frother.
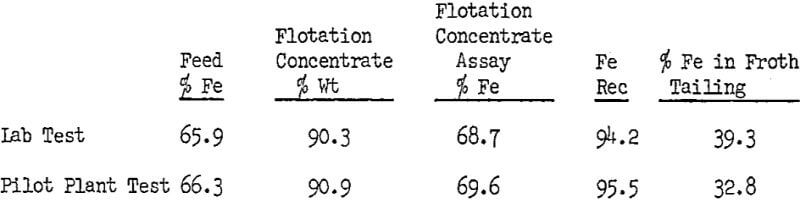
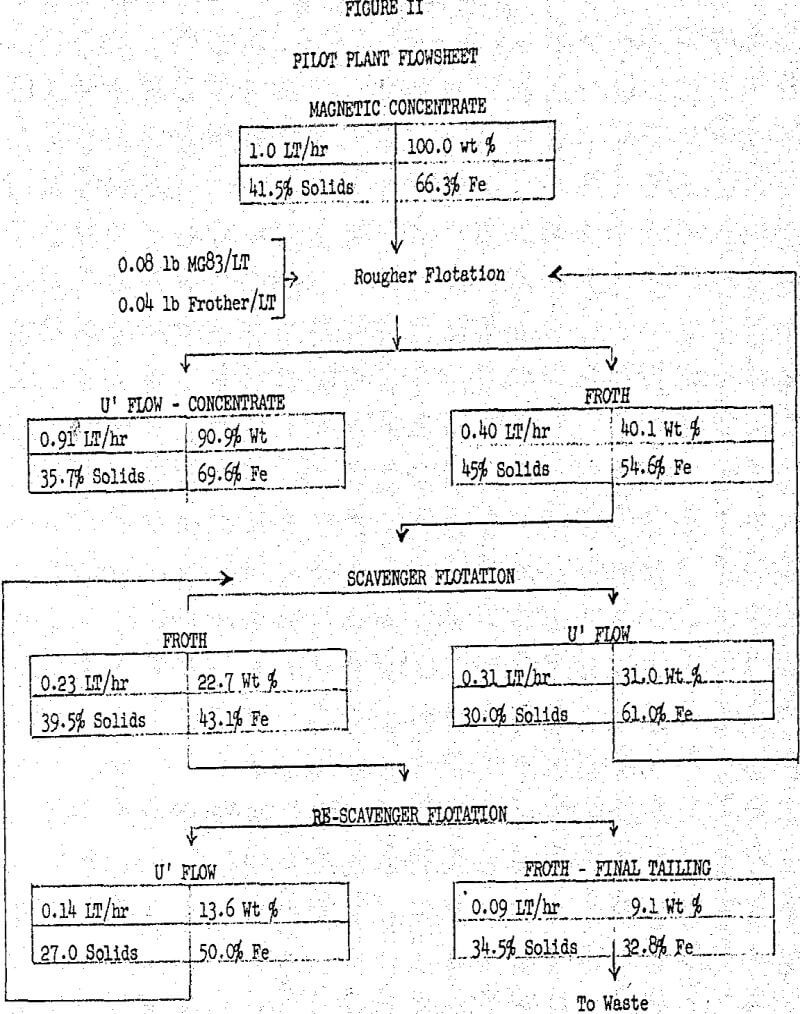
The above results indicate that Arosurf MG-83 was more efficient in the plant type flotation circuit than in the laboratory tests. Most significant was the sharp drop in the iron content of the tailings, while concentrate grade and recovery improved.