The determination of the quantity of a cyanide is made by finding how much silver nitrate is required to convert the whole of the cyanide into potassium silver cyanide or one of the allied compounds. It will be seen from the equation that 170 parts by weight of silver nitrate are required for 130 parts by weight of potassium cyanide. As already explained it is customary to report the cyanide-strength in terms of potassium cyanide, even when only the sodium salt is present. One gram of potassium cyanide will require 1.3076 gram of silver nitrate. The standard solution of silver nitrate is made by dissolving 13.076 grams of silver nitrate in distilled water and diluting to 1 litre; 100 c.c. of such a solution are equivalent to 1 gram of potassium cyanide.
The titration is performed in the usual way, running the standard solution of silver nitrate into a solution containing a known weight or volume of the material containing the cyanide. The finishing point is determined in one of two ways, both of which are largely used. In the first place, as long as there remains any free cyanide in the solution the silver nitrate will combine with it forming the double cyanide and yielding a clear solution ; but as soon as all the free cyanide is used up the silver nitrate will react with the double cyanide forming silver cyanide, which separates as a white precipitate and renders the solution turbid. But, in the second place, if potassium iodide is present in the solution the excess of silver nitrate will react with it, rather than with the double cyanide; and silver iodide will separate as a yellowish turbidity which is easily recognised.
In working with pure solutions, the two finishing points give the same results; and this is true even when there is much difference in the degree of dilution. The finishing point with the iodide, however, has an advantage in precision. Moreover, it is but little affected by variations in alkalinity, which render the other finishing point quite useless. The great difference between the two is shown when zinc is present in the solution. In this case, when working without the iodide, the first appearance of a turbidity is less distinct; the turbidity increases on standing and as a finishing point is unsatisfactory. It can be determined with precision only by very systematic working and after some experience. The turbidity is due to the separation of an insoluble zinc compound. A most important point (to which reference has already been made) is that less silver nitrate is required to give this turbidity and, consequently, a lower strength in cyanide is reported. On the other hand, as much silver nitrate is required to give the yellow turbidity due to silver iodide as would be required if no zinc were present.
Unfortunately the difference in the two titrations does not depend merely on the quantity of zinc present; as it is also influenced by the extent of dilution, the degree of alkalinity of the solution, and the quantity of cyanide present. In an experiment with .055 gram of zinc sulphate and .1 gram of potassium cyanide the difference in the two finishing points was only .1 c.c.; whereas with .4 gram of potassium cyanide, the other conditions being the same, the difference was 1.5 c.c. of standard silver nitrate. On the assumption that all the zinc was present as potassium zinc cyanide (K2ZnCy4) the difference should have been 5 c.c. in each case. Again, repeating the experiment with .4 gram of potassium cyanide, but with .11 gram of crystallised zinc sulphate, the difference was 6.5 c.c.: that is, merely doubling the quantity of zinc increased the difference by more than four times. Hence it would appear better to use the method with the iodide and make a separate assay for the zinc. But since the student may be called on to use the other method, he is advised to practice it also.
The assay without iodide.—The standard solution of silver nitrate is placed in a small burette divided into tenths of a c.c. Ten c.c. of the cyanide solution to be assayed is transferred to a small flask and diluted with water to about 70 c.c. The silver solution is then run in from the burette (with constant shaking of the flask), a little at a time but somewhat rapidly, until a permanent turbidity appears. Since 1 c.c. of the silver nitrate solution corresponds to .01 gram of potassium cyanide, it also corresponds to .1 per cent, of this salt counted on the 10 c.c. of cyanide solution taken. The titration should be performed in a fairly good uniform light. The learner should practice on a fairly pure solution of potassium cyanide at first, and this may conveniently have a strength of about 1 per cent. For practice with solutions containing zinc make a solution containing 1.1 gram of crystallised zinc sulphate in 100 c.c. and slowly add measured quantities of from 1 to 5 c.c. of this to the 10 c.c. of cyanide liquor before diluting for the titration.
If a cyanide solution blackens on the addition of the silver nitrate it contains sulphide. In this case, shake up a considerable bulk of the liquor with a few grams of lead carbonate, allow to settle and make the assay on 10 c.c. of the clear liquor.
If the cyanide liquor be suspected to contain free prussic acid, take 10 c.c. for the assay as usual; but, before titrating, add .1 or .2 gram of sodium carbonate. On no condition must caustic soda or ammonia be added. The difference between the results, with and without the addition of carbonate of soda, is supposed to measure the quantity of free prussic acid. If this has to be reported it is best done as “ prussic acid equivalent to per cent, of potassium cyanide.” Suppose, for example, the difference in the two titrations equals 1 c.c. of standard silver nitrate; the prussic acid found would be equivalent to. 1 per cent, of potassium cyanide.
The assay with iodide.—The standard solution of silver nitrate is placed in a burette divided into tenths of a c.c. Take 10 c.c. of the cyanide liquor, which should previously have been treated with white lead for the removal of sulphides if these happened to be present. Transfer to a small flask, add 3 or 4 drops of a solution of potassium iodide and 2 or 3 c.c. of a solution of sodium hydrate; dilute to 60 or 70 c.c. with water. If much zinc is present the soda may be increased to 20 or 30 c.c. with advantage. The standard solution should be run in somewhat rapidly, but a little at a time, so that the precipitate at first formed shall be small and have only a momentary existence. The titration is continued until there is a permanent yellowish turbidity. The most satisfactory and exact finish is got by ignoring any faint suspicion of a turbidity and accepting the unmistakable turbidity which the next drop of silver nitrate is sure to produce. This finishing point gives results which are exactly proportional to the quantity of cyanide present; and it can be recognised with more than ordinary precision even in solutions which are not otherwise perfectly clear.
Each c.c. of the standard silver nitrate solution corresponds to .01 gram .of potassium cyanide; and if 10 c.c. of the liquor are taken for assay this corresponds to .1 per cent, or 2 lbs. to the short ton or 2.24 lbs. to the long ton. As already explained the result should be reported as 44 cyanide equivalent to so much per cent, of potassium cyanide.”
The following experimental results were obtained with a solution of potassium cyanide made up to contain about 1.2 per cent, of the salt.
Effect of varying cyanide. The bulk before titration was in each case 60 c.c. ; 2 c.c. of soda and 3 drops of potassium iodide were used in each case.
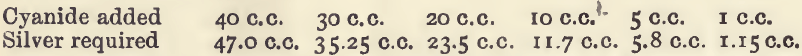
Accepting the result for 40 c.c. as correct, the others are in very satisfactory agreement.
Effect of varying dilution. The conditions were those of the 40 c.c. experiment in the last series ; but varying amounts of water were used in diluting.

Very considerable dilution therefore has no effect.
Effect of varying soda. The conditions were those of the 40 c.c. experiment in the first series, except that varying amounts of soda solution were used.

This alkali therefore has no prejudicial effect.
Effect of ammonia. Soda causes turbidity in some cyanide liquors ; with these it should be replaced by 2 or 3 c.c. of dilute ammonia with a gram or so of ammonium chloride. The following experiments with dilute ammonia show that larger quantities of this reagent must be avoided.

CYANIDES
The cyanides ought perhaps to be considered along with chlorides; bromides and iodides in Chapter XV. But they are treated here because they owe their importance to their use in the extraction of gold and because their determination has become a part of the ordinary work of an assayer of gold ores.
Formerly, the cyanide most easily obtained in commerce was potassium cyanide; and it was generally sold in cakes which might contain as little as 40 per cent, or as much as 95 per cent, of the pure salt. It became customary to express the quality of a sample of commercial cyanide by saying it contained so much per cent, of potassium cyanide. The commercial product now made by improved methods of manufacture is actually sodium cyanide, but is called “potassium cyanide” (probably with the words “ double salt ” on the label); it contains cyanide equivalent to something over 100 per cent, of potassium cyanide in addition to a large proportion of sodium carbonate and other impurities. What is wanted in most cases is merely a soluble cyanide, and it is a matter of indifference whether the base be sodium or potassium. But since 49 parts of sodium cyanide (NaCN = 49) are equivalent to 65 parts of potassium cyanide (KCN = 65) it is evident that a pure sample of sodium cyanide would contain cyanide equivalent to little less than 133 per cent, of potassium cyanide. Therefore a sample of cyanide reported on in this way may be rich in cyanide, and yet have much impurity.
The commonest impurity in commercial cyanide is carbonate of sodium or potassium. This may be tested for by dissolving, say, 2 grams in a little water and adding barium chloride. There may be formed a white precipitate of barium carbonate, which if filtered off, washed and treated with acid, will dissolve with effervescence. Cyanate may be tested for in the solution from which the barium carbonate has been filtered by adding a little soda and boiling; if cyanates are present they decompose, giving off ammonia (which may be tested for in the steam) and yielding a further precipitate of barium carbonate. If the soda alone gave a further precipitate of barium carbonate, this may, perhaps, be due to the presence of bicarbonates. Alkaline sulphides may be present in small quantity in commercial cyanide. Their presence is shown at once when the sample is being tested for its strength in cyanide, inasmuch as the first few drops of silver nitrate solution produce at once a darkening of the liquor. A special test for sulphide may be made by adding a drop or two of solution of acetate of lead to four or five c.c. of soda solution and adding this to a clear solution of the suspected cyanide. This will cause a black precipitate or colour, if any sulphide is present.
The cyanides of the heavier metals combine with the alkaline cyanides to form double cyanides. Some of these, ferrocyanide and ferricyanide of potassium for example, have such characteristic properties that the fact that they are cyanides maybe overlooked. Others, such as potassium zinc cyanide (K2ZnCy4), have much less distinctiveness: they behave more or less as a mixture of two cyanides and are moreover, so easily decomposed that it may be doubted if they can exist in dilute alkaline solutions. In reporting the cyanide strength of a cyanide liquor as equivalent to so much per cent, of potassium cyanide, there is a question as to whether the cyanide present in the form of any of these double cyanides should be taken into account. It must be remembered that the object of the assay is not to learn how much of the cyanide exists in the solution as actual potassium cyanide; reporting the strength in terms of this salt is a mere matter of convenience; what is really desired is to know how much of the cyanide present in the liquor is “free” or “available” for the purposes of dissolving gold. Every one is agreed as to the exclusion of such cyanides as the following: potassium ferrocyanide (K4FeCy6), potassium ferricyanide (K3FeCy6), potassium silver cyanide (KAgCy2), and potassium aurocyanide (KAuCy2); and the double cyanides with copper or nickel. But with cyanide liquors containing zinc the position is less satisfactory. One method of assay gives a lower proportion of cyanide when this metal is present; and the loss of available cyanide thus reported depends, though in a fitful and uncertain way, upon the quantity of zinc present. The other method of assay reports as full a strength in cyanide as if no zinc were present. Unfortunately, using both methods and accepting the difference in the results as a measure of the quantity of zinc present, or at any rate of the zinc present as cyanide, is not satisfactory. It appears best to use the method which ignores the zinc; and to determine the amount of zinc by a special assay of the liquor for this metal.
The cyanide present as hydrogen cyanide or prussic acid (HCy) is practically useless as a gold solvent. Hence any report on the strength of a cyanide liquor which assigned to this the same value as its equivalent of alkaline cyanide would be misleading. On the other hand, it is “available cyanide” inasmuch as a proper addition of sodium hydrate would restore its value. The question of the presence or absence of free prussic acid is involved in the larger one as to whether the cyanide solution has the right degree of alkalinity. The assay for “cyanide” should include the hydrogen cyanide with the rest.
A rough test of the power of a cyanide liquor for dissolving gold may be made by floating a gold leaf on its surface and noting the time required for its solution. This test might, perhaps, be improved by taking, say, 20 c.c. of the liquor and adding three or four gold leaves so that the gold shall always be in considerable excess. The liquor should not be diluted as this will affect the result. It should be allowed to stand for a definite time, say at least two or three hours, or better, that corresponding to the time the liquor is left in contact with the ore in actual practice. The liquor should then be filtered off and, with the washings, be evaporated in a lead dish as in the assay of cyanide liquors for gold (p. 141). The gold obtained on cupelling, less any gold and silver originally present in the liquor, would be the measure of the gold dissolving power.
Effect of sodium bicarbonate. In this experiment 1 gram of bicarbonate of soda was used instead of the soda or ammonia of the other experiments. The silver nitrate required was only 46.45 c.c. instead of the 47.0 c.c. which is the normal result.
This is probably due to the liberation of prussic acid and shows the importance of having the solution alkaline.
Effect of zinc. In each experiment 40 c.c. of the cyanide solution and .5 gram of zinc sulphate crystals were used and the bulk was made up to 100 c.c. before titrating.
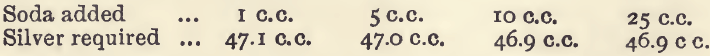
The work was easier with the more alkaline solutions. The titration in the presence of zinc is comparatively easy, but, in learning it, it is well to have a burette with cyanide so that if a titration be overdone it can be brought back by the addition of 1 or 2 c.c. more cyanide and the finish repeated; a quarter of an hour’s work in this way will ensure confidence in the method.
Effect of other substances.—It was found that an alkaline cyanate, sulphocyanate, ferrocyanide, nitrite, borate, silicate or carbonate has no effect. The ferricyanide had a small influence and, as might be expected, hyposulphite is fatal to the assay. The addition of salts of lead and cadmium was without effect. On the other hand, nickel produces its full effect; and the quantity of nickel added can be calculated with accuracy from the extent of its interference with the titration.
Assay of commercial cyanide of potassium.—Break off 20 or 30 grams of the cyanide in clean fresh pieces, weigh accurately to the nearest centigram. Dissolve in water containing a little sodium hydroxide; transfer to a 2-litre flask: dilute to 2 litres; add a few grams of white lead; shake up and allow to settle. Run 50 c.c. of the clear liquor from a burette into an 8 oz. flask; add 2 or 3 c.c. of soda solution and 3 drops of potassium iodide. Titrate with the standard solution of silver nitrate. The percentage may be calculated by multiplying the number of c.c. used by 40 (50 c.c. is one fortieth of the 2 litres) and dividing by the weight of commercial cyanide originally taken.
Alkalinity of commercial potassium cyanide and of cyanide solutions.—Hydrocyanic acid like carbonic acid has no action on methyl-orange; hence the alkaline cyanides may be titrated with “ normal acid ” as easily as the carbonates or hydrates. 100 c.c. of normal acid will neutralise 6.5 grams of pure potassium cyanide. A solution of commercial cyanide prepared as for the assay last described, but best without the addition of white lead, may be used for the test. Take 50 c.c. of it; tint faintly yellow with methyl-orange and titrate with normal acid till the liquor acquires a permanent reddish tint. In the case of the purer samples of cyanide the quantity of acid used will correspond exactly with that required to neutralise the actual quantity of cyanide present as determined by the assay with nitrate of silver. The less pure samples will show an excess of alkalinity because of the presence of sodium carbonate or of potassium carbonate.
In comparing the alkalinity and cyanide strength of a solution the simplest plan is to take 65 c.c. of the solution and titrate with normal acid; for in this case each c.c. of normal acid corresponds to .1 per cent, of potassium cyanide. In systematic assays of this kind, the alkalinity would no doubt be generally in excess of that required by the cyanide present: there would be no inconvenience in recording such excess in terms of potassium cyanide.
Determination of the acidity of an ore
Most ores have the power of destroying more or less of the alkalinity of a cyanide solution and in a proportionate degree of damaging its efficiency. An assay is needed to determine how much lime or soda must be added for each ton of ore in order to counteract this. Whether this acidity should be reported in terms of the lime or of the soda required to neutralise it will depend on which of these reagents is to be used in the actual practice. Again, if the ore is washed with water before treating with cyanide on the large scale, then the assay should be made of the acidity of the ore after a similar washing.
The standard solutions of acid and alkali used for this determination, may be one-fifth normal. 200 c.c. of the normal solution should be diluted to 1 litre in each case. 1 c.c. of the resulting solutions would be equivalent to 8 milligrams of soda (NaHO) or 5.6 milligrams of lime, CaO. It must be remembered this refers to the pure bases in each case. Suppose it is desired to report as so many lbs. of lime to the short ton (2000 lbs.) of ore. Since 1 c.c. of the standard solution is equivalent to 5.6 milligrams of lime, if we take 2000 times this weight of ore (i.e. 11,200 milligrams or 11.2 grams) for the assay, each c.c. of standard solution will be equivalent to 1 lb. of lime to the short ton.
Total acidity.—Weigh out 11.2 grams of the ore, place them in a four-inch evaporating dish and measure on to it from a burette 10 or 20 c.c. of the standard solution of soda. Stir the soda solution into the ore and allow to stand for 15 or 20 minutes with occasional stirring. Stir up with 30 or 40 c.c. of water, float a piece of litmus paper on the liquid and titrate with the standard solution of acid. If the ore is strictly neutral the quantity of “acid” required to redden the litmus will be the same as the quantity of “ soda ” originally used. If the ore is acid, less acid will be used. For example, if 10 c.c. of soda were used and only 7 c.c. of acid were required, the ore will have done the work of the remaining 3 c.c. of acid. And the ton of ore will require 3 lbs. of lime to neutralise its acidity.
Acidity after washing.—Take 11.2 grams of the ore; wash thoroughly with water and immediately treat the residue, without drying, exactly as just described.
Examination of cyanide solutions for metals
Take a measured quantity of the solution, say 20 c.c. and evaporate in a small dish with, say, half a c.c. of strong sulphuric acid. Evaporate at first, on a water-bath in a well ventilated place, but finish off with a naked Bunsen flame, using a high temperature at the end in order to completely decompose the more refractory double cyanides. Allow to cool; moisten with strong hydrochloric acid; warm with a little water and test for the metals in the solution by the ordinary methods. Since the quantities of the metals likely to be present may be given in milligrams the work must be carefully performed. It may be worth while to determine the proportions of lime and magnesia as well as those of the metals proper.
Or the 20 c.c. of cyanide liquor may be evaporated with 5 c.c. of strong nitric acid to dryness and gently ignited and the residue taken up with 2 or 3 c.c. of strong hydrochloric acid.
Copper, iron, and zinc can be rapidly determined in such a solution, as follows. Dilute with water to 10 or 15 c.c., add an excess of ammonia, and filter. The precipitate will contain the iron as ferric hydrate; dissolve it in a little hot dilute sulphuric acid : reduce with sulphuretted hydrogen; boil off the excess of gas, cool and titrate with standard potassium permanganate (p. 236). Determine the copper in the filtrate colorimetrically (p. 203); but avoid further dilution. Then add dilute hydrochloric acid, so as to have an excess of 4 or 5 c.c. after neutralising the ammonia; add some clean strips of lead foil, and boil until the solution has for some time become colourless. Titrate with standard potassium ferrocyanide (p. 263) without further dilution, and bearing in mind that at most only one or two c.c. will be required.
Examination of an ore for “ cyanicides”
Place 100 grams of the ore with 200 c.c. of a cyanide solution of known strength (say .1 or .2 per cent.) in a bottle and agitate for a definite time, such as one or two days. Filter off some of the liquor and assay for cyanide, using say 20 c.c. Calculate how much cyanide has been destroyed in the operation. Evaporate 20 c.c. with sulphuric or nitric acid and examine for metal. Test another portion for sulphides, &c.
The student who has mastered the methods of assaying can greatly improve himself by working out such problems as the above.
