The production of gold bullion from gravity or flotation concentrates is often an important economic consideration for an isolated gold mining operation. It is assumed in this case that the coarse free gold has been recovered by the Mineral Jig in the grinding circuit and that the jig concentrate has been amalgamated. This treatment produces a portion of the gold (25 to 40 percent of the total) as amalgam, but the balance of the recoverable gold is present as non-amalgamable flotation concentrate together with the amalgamation residue. The problem is therefore, to develop a batch cyanidation procedure that will convert the gold contained in these products into bullion. A batch procedure is desired because of the small quantity of total concentrate (5 to 6 tons) produced daily and the greater flexibility and simplicity afforded by a batch type process.
The Flowsheet
Gravity and flotation concentrates that contain a portion of gold closely associated with sulphides may require fine grinding (—200 mesh) followed by pre-aeration and/or pre-agitation with lime prior to cyanidation in order to minimize the chemical consumption. Refractory gold concentrates may require roasting, but this step is avoided if possible due to the greater cost involved.
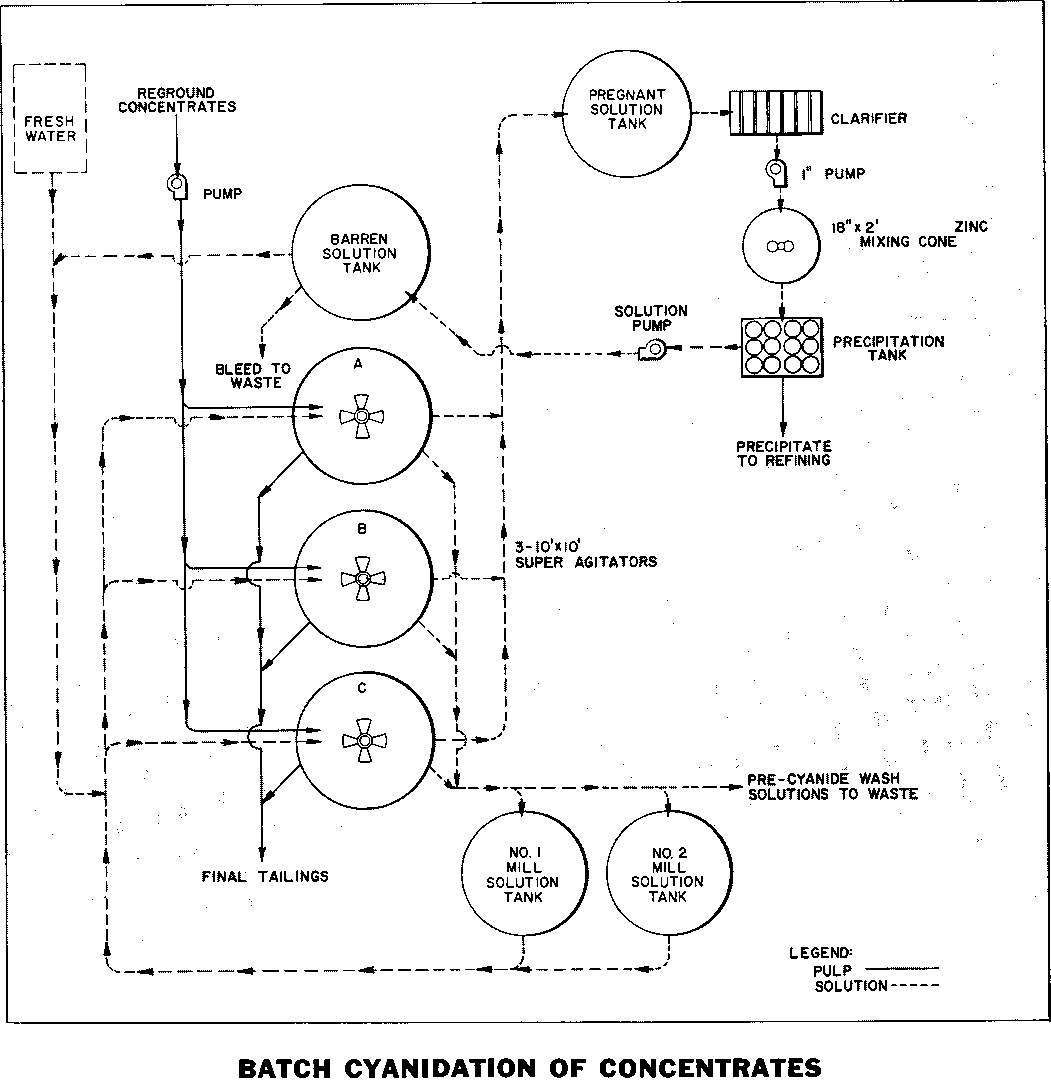
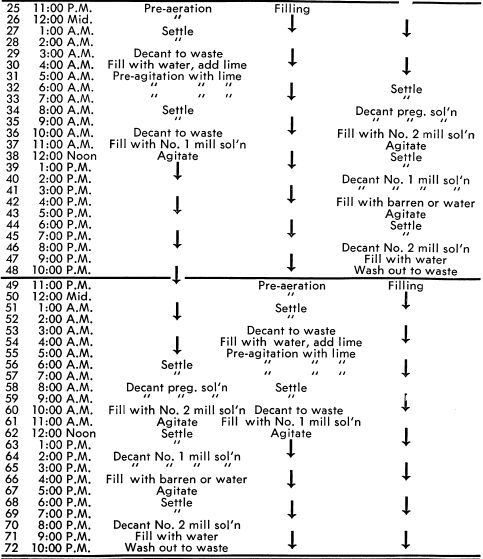
BATCH CYANIDATION CYCLES—As shown in the flowsheet, the reground concentrates are alternately pumped to one of three 10′ x 10′ Super Agitators equipped with turbine-type propellers. These agitators produce intense mixing and aeration for optimum dissolution of the gold. The Super Agitator with its hood protected propeller permits the solids to settle in the bottom of the tank and then be repulped by simply starting the motor. The agitators are operated as batch units, that is, one agitator is filling while the next agitator is mixing and the third agitator is settling and decanting.
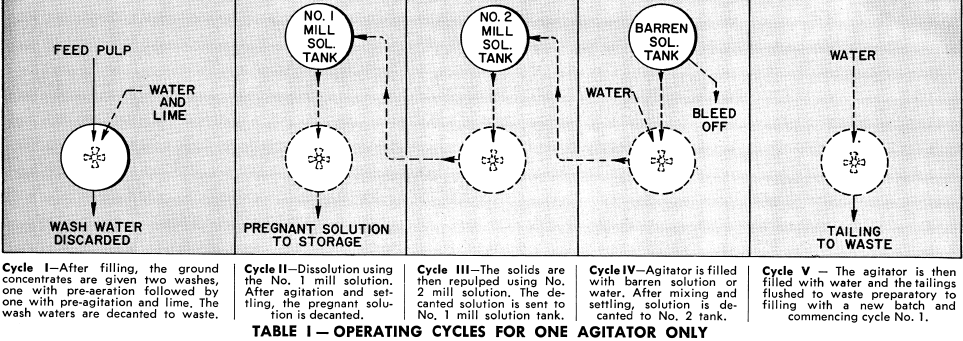
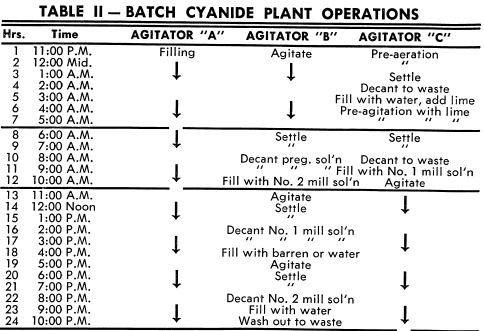
This can best be understood by following the functions of one agitator through all its operating cycles during a 72-hour period. Referring to Tables I and II, Agitator “A” is filled with reground concentrates and given two washes, one with water during pre-aeration followed by one with pre-agitation in the presence of lime. These wash waters are decanted to waste.
Following pre-agitation with lime and decanting the solution to waste, agitator “A” is filled with Number 1 mill solution. After agitation for dissolution of the gold and settling, the pregnant solution is decanted to the pregnant solution tank.
Agitator “A” is then filled with Number 2 mill solution and after agitation and settling, the solution is decanted to Number 1 mill solution tank. The agitator is then filled with barren solution or water and after agitation and settling, the solution is decanted to the Number 2 mill solution tank. The agitator is filled with water and the tailings washed to waste preparatory to filling with a new batch. This completes the entire cycle of operations for agitator “A”. The simultaneous cycles of the other agitators are shown in Table II. The time intervals shown for each cycle can be varied depending on the physical and metallurgical characteristics of the feed.
PRECIPITATION—The pregnant solution passes from the storage tank to a Zinc Dust Precipitation Unit for recovery of the gold precipitate. The barren solution, as needed, is returned to the three agitators for washing. A portion of the barren solution is also bled off and discarded to prevent the build-up of sulpho-cyanates.
CHEMICAL CONSUMPTION — Inasmuch as the concentrates from any ore will usually represent an enirchment of the base metals and other sulphides contained in the crude ore, a relatively high chemical consumption can be expected during cyanidation. However, the chemical consumption in terms of pounds per ton of concentrate when related back to the crude ore basis will usually not be excessive. The exception would be a concentrate containing appreciable copper which, of course, would preclude the use of cyanidation.
Remarks
The gold extraction will be in the order of 85% depending on the operating conditions. The batch cycle cited in this study include the pre-aeration and pre-agitation (with lime) steps to demonstrate their necessity in some instances. If the concentrates do not contain a prevalent amount of sulphides or if care is taken to protect concentrates from exposure to air and oxidation, the pre-aeration and pre-agitation steps may not be required. In this instance this part of the batch cycle can be utilized for additional dissolution or washing time and the concentrate regrinding can be performed in cyanide solution.
AERATING AGITATOR AIDS GOLD DISSOLUTION AND REDUCES CYANIDE CONSUMPTION
Increased dissolution rates for gold and many other elements as well as substantial reductions in chemical consumption are obtained by a maximum effective concentration of oxygen.
Large volumes of uncontrolled air are not as affective in saturating a solution with oxygen as the controlled distribution of fine bubbles throughout the slurry.
It can be calculated that one cubic foot of air will create 3,300 bubbles of 1″ diameter. These 3,300 bubbles have a combined surface area of 10,360 square inches. If these bubbles are reduced in size to 1/10″ diameter the number increases to 3,300,000 bubbles with a surface area of 103,600 square inches. The larger surface area is many times more effective in putting oxygen into solution.
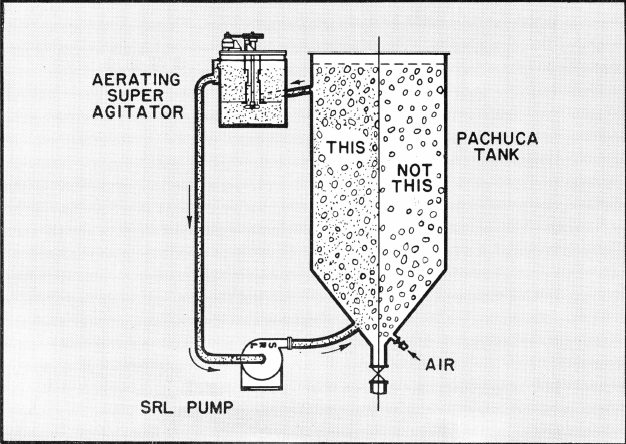
A small Aerating Agitator used in conjunction with a Pachuca or other large tank aids materially in gold dissolution and cyanide reduction by saturating the pulp with large quantities of ultra-fine air bubbles.
A portion of the pulp from the large tank is bled to a small Aerating Agitator. The controlled recirculation of the pulp in the Agitator forces air into the slurry. Overflow from the Agitator is pumped by a SRL Pump into the bottom of the large tank. By keeping 10% of the total pulp highly aerated the balance will be fully saturated with oxygen.