Beryllium Processing & Extraction Problem
Beryl, the principal Beryllium mineral, is found generally in pegmatites associated with mica, feldspar, and quartz. Its specific gravity is almost identical to quartz and feldspar, thus ruling out the possibility of gravity separation methods. Beryl is harder than quartz. Most of the production of this mineral in the past has been by hand sorting and cobbing of high grade crystals from pegmatite mining operations. The crystals are hexagonal and may range up to several inches across.
Beryllium Extraction Process Flowsheet
Flotation offers the best means for recovering beryl in concentrate form. Much of the flotation technique developed in separation of feldspar from quartz and mica is applicable. In general, beryl will concentrate with the feldspar flotation product. Depressing the feldspar and reflotation of the beryl into a high grade marketable concentrate (at least 8.0% BeO) is the usual procedure.
GRINDING AND CLASSIFICATION
For the grinding of beryl and other non-metallic ores a Rod Mill is used to minimize sliming. A more granular product is desirable since for subsequent treatment slimes must be classified out and sent to waste. In most non-metallics a size reduction to at least 20 mesh is necessary, and in case of beryl ores, 35 mesh or finer may be required to liberate the beryl crystals from the feldspar and quartz. In view of the finer grind a Rake or Spiral Classifier is used to close the grinding circuit. A Spiral Screen on the mill discharge will remove tramp oversize and coarse flakes of mica delaminated by the rod mill action.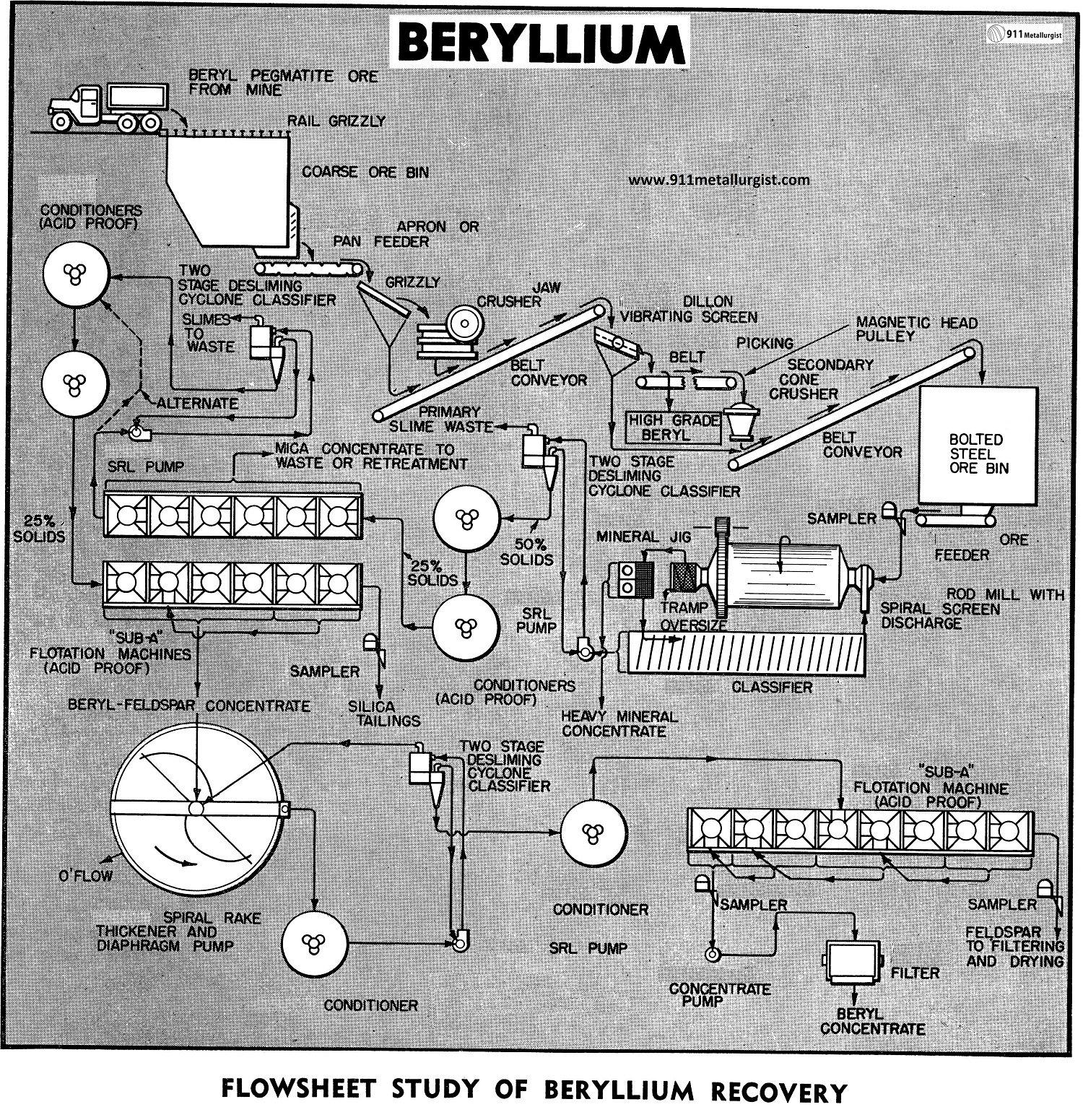
Many of the pegmatite ores contain variable amounts of heavy minerals such as Cassiterite, Columbite, Tantalite, and Microlite. It is therefore advisable to incorporate a Mineral Jig in the grinding circuit for recovery of the heavy minerals when present. The heavy mineral concentrate can be cleaned up and separated on a batch basis with high intensity magnetic and/or electrostatic separators.
Desliming of the rod mill discharge may be done through a Cyclone Classifier, preferably of two stages, to minimize sand losses and assure efficient removal of colloidial slimes. Generally it is necessary to discard the minus 325 mesh fraction containing the slimes which would otherwise affect the flotation separation. The classification also thickens the deslimed sand for subsequent conditioning at 50% solids or higher. Beryl is a rather tough mineral and does not slime readily.
MICA FLOTATION
The thickened deslimed pulp at 50% solids is conditioned with sulphuric acid for about 3 minutes at a pH of 1.5 to 2.0. This usually requires from 1 to 5 lbs. of sulphuric acid per ton of ore. After the short acid conditioning step the balance of the mica flotation reagents are added to the second conditioner tank. The reagents are usually 0.10 to 0.20 lbs. of tallow amine acetate and a small amount (0.01-0.05 lbs.) of an alcohol frother such as methyl amyl alcohol. The reagentized pulp, as it passes to the flotation cells, is diluted with fresh water to 25% solids and the mica is floated. If the mica is a good variety of muscovite, it may be cleaned several times by flotation and saved; otherwise, it goes to waste. It is, however, important to remove the mica from the pulp prior to subsequent recovery of the feldspar and beryl.
BULK FELDSPAR-BERYL FLOTATION
The tailing from the mica flotation cells at approximately 25% solids is conditioned about 5 minutes with 1 to 3 lbs. hydrofluoric acid per ton of solids. pH is kept in the range of 2.0 to 2.5. In some cases it is necessary to thicken ahead of the hydrofluoric acid conditioning step. This can be done by the cyclone arrangement as shown in the flowsheet. Thickening also aids in removing any additional slimes which may have formed or carried over from the initial desliming circuit.
The bulk feldspar and beryl concentrate is floated with amine acetate collector and an alcohol frother, usually the same as used in the mica float but in larger quantities, up to 0.5 lbs. amine acetate and 0.05 lbs. of frother per ton of solids.
BERYL FLOTATION
The bulk feldspar-beryl concentrate is thickened in a Thickener. In addition to removing the reagents and excess water the thickener provides storage capacity so the beryl flotation section can be operated under more carefully controlled conditions and therefore not be subject to fluctuations in the previous processing and treatment steps.
The thickened bulk concentrate at 50% solids is conditioned with 0.5 to 2.0 lbs. of calcium hypochlorite per ton of original ore treated for about 5 minutes. Next it is necessary to wash the pulp to remove all reagents. This is done in the Cyclone arrangements as shown in the flowsheet. Fresh water should be added to the pump feeding the cyclone. SRL Pumps are recommended for cyclone service because their flat pumping curve makes close pressure control possible and the soft rubber lining resists abrasives. The washed solids are conditioned at 50% solids for 3 minutes with 1 to 4 lbs. of sulphuric acid and 0.5-3.0 lbs. of petroleum sulfonate and then diluted to 20 to 25% solids for flotation of the beryl. The feldspar fraction is rejected into the flotation tailing in this part of the treatment. The froth containing the beryl may be cleaned two or more times to raise the grade up to the desired beryllium oxide content, usually 10% BeO. A small amount of alcohol frother may be necessary to stabilize the froth and assure carry-over of the beryl in the cleaning stages.
The froth containing the beryl is pumped to a Rotary Filter for dewatering. For small tonnages of final beryl concentrate it may be advantageous to use a Pan Filter. Feldspar in the beryl flotation tailing fraction, if low in iron, and of acceptable quality, can be dewatered, filtered and dried.
DIRECT FLOTATION OF BERYL
The procedure previously outlined applies to pegmatite ores containing appreciable feldspar along with the beryl and mica. This procedure is desirable if the recovery of the feldspar is required. In case beryl only is the important mineral the bulk beryl-feldspar flotation steps may be modified or eliminated. This would also be applicable on beryl ores with low feldspar content.
In the direct flotation of beryl the mica is removed as previously described. The mica free pulp is then conditioned with calcium hypochlorite. After washing to remove reagents, the beryl is floated with petroleum sulfonate in a sulphuric acid circuit.
LABORATORY TESTING
Laboratory testing is definitely recommended to establish the flotation characteristics of a specific pegmatite beryl deposit.
Analysis of beryllium ore products is rather difficult and tedious. This can be accomplished spectographically or chemically, the later preferable by fluorometric methods. BeO analyses currently cost $15.00 per determination so laboratory work on the recovery of this mineral is still rather expensive.
Facilities and experience of the Ore Testing Division are available to work on beryl concentration problems.
Beryllium Facts
- Atomic number (number of protons in the nucleus): 4
- Atomic symbol (on the Periodic Table of the Elements): Be
- Atomic weight (average mass of the atom): 9.012182
- Density: 1.85 grams per cubic centimeter
- Phase at room temperature: Solid
- Melting point: 2,348.6 degrees Fahrenheit (1,287 degrees Celsius)
- Boiling point: 4,479.8 F (2,471 C)
- Number of isotopes (atoms of the same element with a different number of neutrons): 12, including one stable isotope.
- Most common isotopes: 9Be (Natural abundance: 100 percent)
- Beryllium discovery and use
Beryllium Geology
Beryl, a silicate of aluminum and beryllium, is found along with other beryllium minerals in pegmatite dikes. Large quantities have been taken out of some feldspar mines. The metal beryllium is much like aluminum, but is about a third lighter, and is as hard as steel. It is also about four times as elastic as aluminum, and 25% more elastic than steel. Unlike aluminum, it resists corrosion by salt water, etc. Its rate of expansion with heat is the same as that of cast iron. It alloys easily with aluminum. An alloy of beryllium 70% and aluminum 30%, is one-fifth lighter than aluminum, and resists corrosion much better. In tensile strength it greatly exceeds duralumin.
Minerals of the Rare Earths
There is a group of minerals known as rare earths minerals, called by that name because they contain uncommon oxides of an earthy nature; they are found mainly in granite, and particularly in pegmatite dikes. Since they are hard, heavy substances, they become concentrated in the sand and gravel formed by the weathering of these rocks, and they may be noticed in panning for gold. A heavy tail of red, yellow, orange, black, or white, color should be tested for these minerals.
Source: This article is a reproduction of an excerpt of “In the Public Domain” documents held in 911Metallurgy Corp’s private library.
