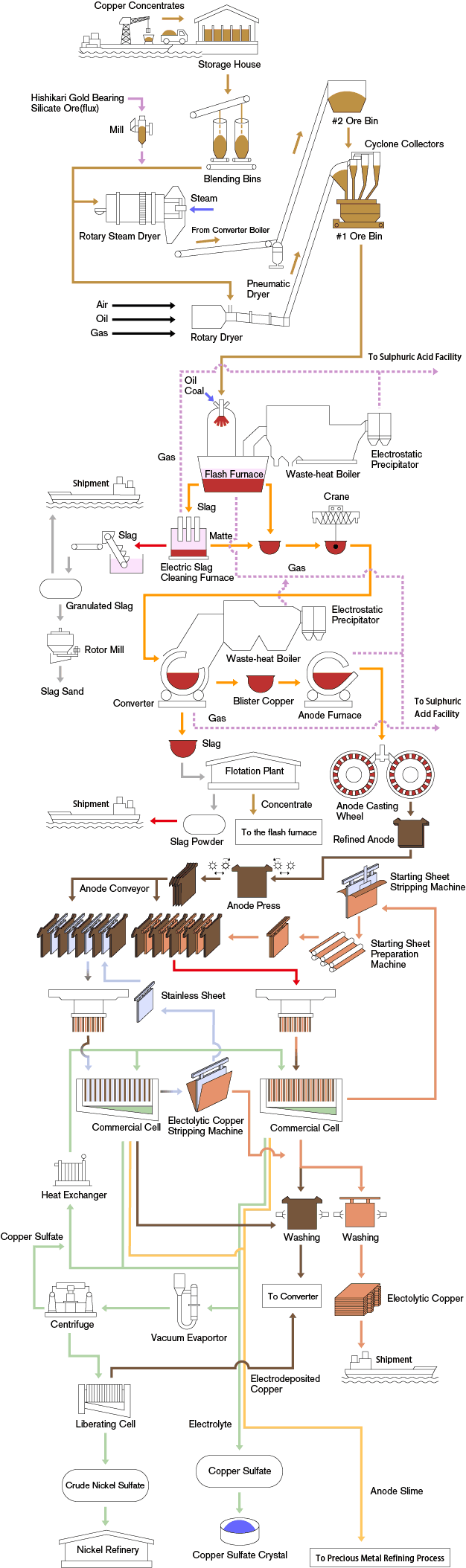
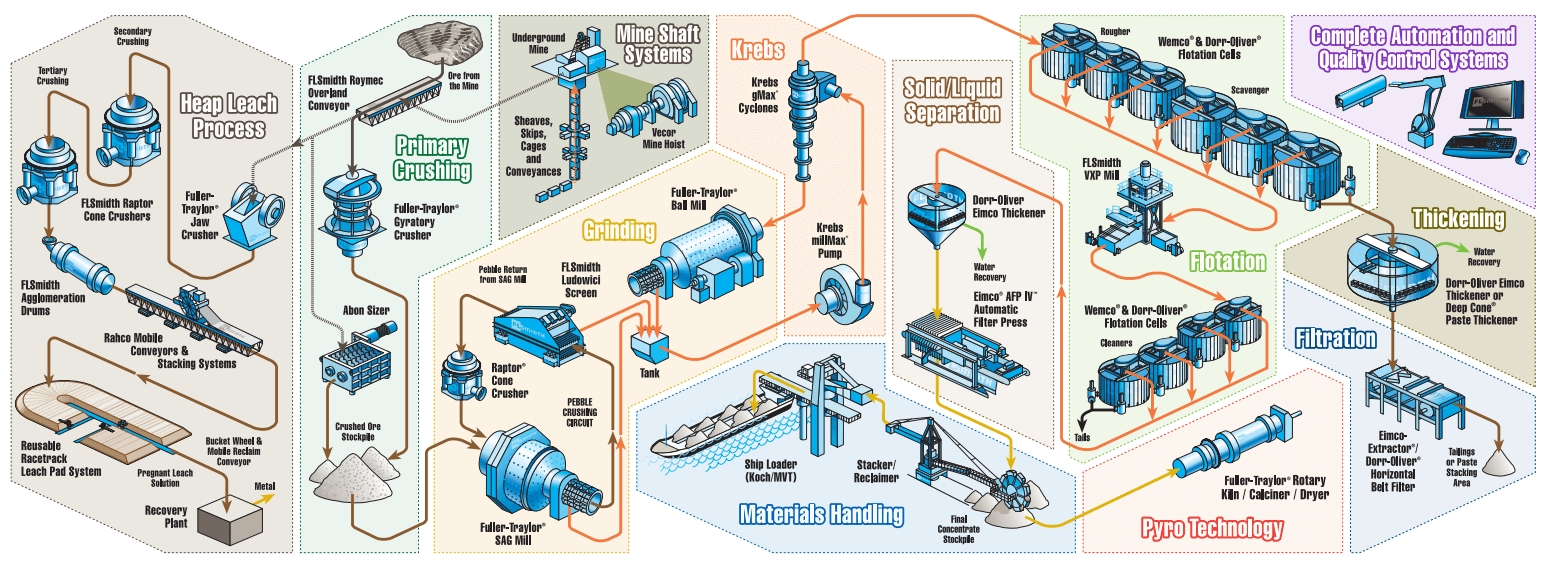
This flowchart made of machinery icons explains or expresses in simple but clear terms the step of the Copper Mining and Copper Extraction Process. Starting from either open-pit or underground mining and using a different relevant treatment method for oxide or sulphide copper mineral (ore).
Having a quick look now at how porphyry ores are treated and the metals extracted. There are two main process streams; one for sulfide ores and the other for ore that is being weathered to oxidize sulfides the so-called oxide ores. All ore in the pit is drilled and blasted and loaded into trucks and hauled for treatment if the ore is un-oxidized sulfidic ore then it needs to be crushed and milled to a fine slurry then it gets past through flotation cells in a concentrator to separate and concentrate the sulfides. The top picture shows the interior of a large concentrator with rows of individual flotation cells the floatation agent is added to the slurry and stirred. The floatation agent preferably sticks to the sulfide minerals rather than the waste minerals and then air is bubbled through the mixture and the floatation agent traps the fine bubbles which carry the sulfides to the surface of the cell where they are carried over aware and separated. From there they are dried to provide a concentrate which then goes on to a smelter. This is the same process for both copper and molybdenum porphyries. The smelter is basically a large furnace which melts the concentrate and drives off the sulfide to leave molten copper metal this is still contains impurities and it needs to be refined further to make it a salable product.
Returning to the overall process; that is the process for the sulfide ores and the oxide ore as I said are treated differently. Direct from the pit the oxide ore is piled onto large lined leach pads and the sulfuric acid. The top photo shows one of these leach pads with the new thick black plastic liner visible on the right of the pad. The copper oxide minimum minerals are dissolved by the acid to give a blue copper rich solution mainly of copper sulfate. This solution is tapped off from the bottom of the pad and placed into big tanks with steel plates an electrical current is passed from the tank to the steel which is then electroplated with pure copper. This process as the advantage of avoiding the smelting and refining stages required for sulfide ores.
Thanks to https://www.flsmidth.com/
Copper Smelting Process