Simply said; a Flotation Collectors is a chemical used to make the surface of minerals, which is metallic, hydrophobic or in layman’s term ‘afraid of water’ or ‘water hating’ as others like to say.
This chemical used will behaved, in a way, as a wax would when you applied it to your car. After wax (or hydrophobic agent) is adhered to the surface, that surface with see water pearl-away.

Another example of a home-made collector or hydrophobic agent is Naphthalene, the chemical reagent used to make Mothballs. It is very much ‘afraid of water.
Anyway, a similar phenomenon happens to minerals when a flotation collector is applied to them. These flotation collectors are naturally attracted to metallic surfaces making them wanting to get away or get out of water. The most common group of flotation collectors are known as Xanthates.
They are attracted to the mineral and to the air of the bubble, attaching themselves to both.
The difference in the collectors are primarily in selectivity based on their strength.
Based on their chemical compositions and affinities, some flotation collectors will tend to be attracted to one mineral more than another. These will produce a cleaner product but may tend to lose some of the values that are still bound with other minerals that they are not attracted to. 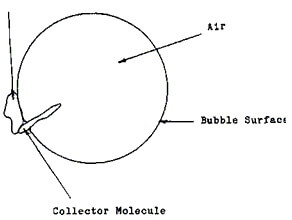
This might cause a move in the grade VS recovery curve. Some may have frothing characteristics as well, this again may or may not be desirable.
Now, image minerals afraid of water, in a tank of mud in which you inject air bubbles… Those minerals will attach to the air bubbles that are passing-by and rise to the surface of the tank just to get away from water. This is the process allowing us to separate minerals from waste rock.
Flotation relies on differences in surface properties of different minerals to achieve separation. The study of these surface properties is known as surface chemistry. In flotation, it is necessary to understand the surface properties of minerals. Most importantly, surfaces can be either hydrophilic (water loving), or hydrophobic (water fearing). Mineral surfaces can also be electrically charged and, under certain conditions, can oxidise.
HYDROPHOBIC
Particles with a hydrophobic surface prefer not to be in contact with water. Such particles are said to be hydrophobic. The reason they are hydrophobic is that the molecules on their surface do not form bonds, specifically hydrogen bonds, with water. Hydrophobic particles prefer to attach to passing air bubbles, rather than stay in the water. Examples of naturally hydrophobic materials are graphite (pure carbon) and solid hydrocarbons such as coal. The surfaces of these particles are non-polar and do non-polar hydrogen bonds with water.
HYDROPHILIC
Particles whose surfaces are hydrophilic prefer to be in contact with water. Such particles are said to be hydrophilic. The reason they are hydrophilic is that the molecules on their surface are able to form hydrogen bonds with water; we control that bond with Flotation Collector dosage.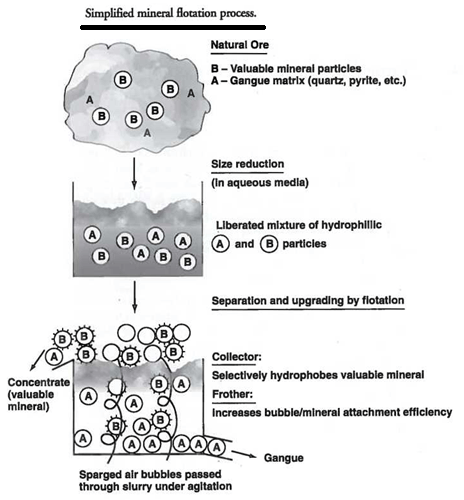 Hydrophilic particles prefer lo slay in the water, rather than attach to a passing an bubble. Examples of hydrophilic particles are sulphide minerals.
Hydrophilic particles prefer lo slay in the water, rather than attach to a passing an bubble. Examples of hydrophilic particles are sulphide minerals.
DEGREE OF HYDROPHOBIA
A particle may have both hydrophilic and hydrophobic patches on its surface. The overall property of the particle depends on the balance which a Flotation Collector will affect. We can estimate the overall hydrophobic of a particle by looking the way a water drop contacts the surface of that particle. If the particle is hydrophilic, the contact angle will be large. It the particle is hydrophobic contact angle will be smelter.
Creating Hydrophobic Particle for Flotation with “Collectors”
To obtain adherence of the desired component to gas bubbles, it is necessary that a hydrophobic surface film be formed on the fine particles of the component to be floated. On the other hand, the fine particles of the other components) must be kept hydrophilic. It is, of course, necessary when considering the use of flotation as a separation process, that some, if not all, of the components originally be somewhat hydrophilic in nature when immersed in water. This situation then allows for the possibility that selective development of hydrophobicity can be made to occur. In almost all flotation processes, the selective development of hydrophobicity is achieved by the use of specific hydrophobing chemical reagents, called collectors in die flotation literature.
Such collector chemistries are the primary driving force that initiates the flotation process If appropriate collectors can not be identified for a given mixture of component particles, normally the flotation process is not feasible. In other words it is not possible to manipulate other parts of the flotation process to overcome the lack of appropriate collector chemistry. Even in the situation where some components, such as coals, or hydrocarbon and organic droplets, have natural hydrophobicity, the use of selective hydrophobing reagents (collectors) is still required because the collectors enhance the selectivity of die separation and also speed up the time rate of the separation process allowing more feed material to be separated with less equipment.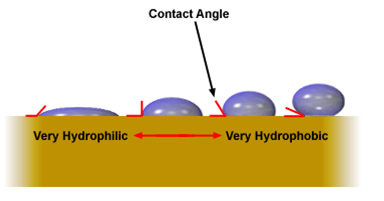
Most of the collector chemistries used industrially fall into the general chemical product category of surfactants (i.e. soaps) and, hence, demonstrate many of the properties exhibited by all surfactants. Since the selection and optimal use of collectors is so key to the flotation process we need to expend about such chemistries and how they work. There is a great deal of complexity involved in selecting and optimizing the choice of a collector (or collectors) for any given separation. Not only is the type of chemistry important, but usually also the dosage, the purity of the water being used (especially with regard to multivalent cation levels present), the pH of the pulp, the influence of excessive fines present in the pulp, and so on. While the efficient selection of appropriate collector chemistry often requires significant technology awareness, it is the low amount of collector used to impart a separation that offers such favourable economics. As mentioned previously, there is no cheaper solid-solid separation process than flotation.
Introduction to Solid-solid Separation of Fine Particles by Froth Flotation. Richard R. Klimpel
What is a Flotation Collector
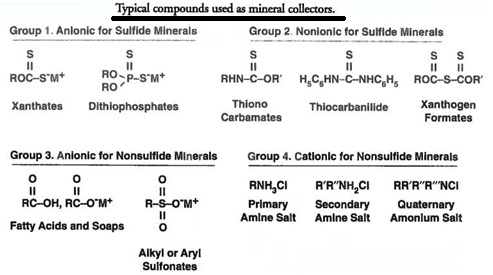
For industrial sulfide mineral flotation, the xanthates and dithiophosphates are the major classes of Flotation Collectors compounds used, where M+ is either Na+ or K+, and R is a relatively short hydrocarbon chain typically containing two to five carbon atoms. The solubility, pH application range, physical state (solid or liquid), and adsorption characteristics vary widely for different compounds within Groups 1 and 2, indicating the difficulty of selecting an optimal collector from a priori reasoning.
Typical sulfide collector dosages range from 0.01 to 0.1 kg/ton of dry feed to flotation, with some applications ranging up to 0.5 kg/ton.
Specifically, the uptake of xanthate by a given sulfide mineral has been shown in idealized laboratory tests to depend on the rest potential of that mineral. The reduction potential for the formation of the disulfide (dixanthogen) is +0.13 V, which from research studies of mineral surface potentials, includes flotations involving chalcopyrite, molybdenite, pyrite, and pyrrhotite. Thus, xanthate ions are oxidized at the mineral surface to dixanthogen, the most likely chemical form imparting hydrophobicity to the mineral.
The reaction product at the mineral surface for these minerals having a rest potential less than +0.13 V, such as is the case with galena, chalcopyrite, and bornite, is most likely the simple metal xanthate.
Obviously, from a practical viewpoint, the effectiveness of the hydrophobic character imparted by the relatively short hydrocarbon chain thiol collectors of Group 1 implies that the hydrophobic sulphur atoms of the collector and those of the mineral work together in whatever surface arrangement may be present. One sulfur atom is equivalent to 3.5 carbon atoms in terms of imparting surface hydrophobicity. Thus, sulphur containing minerals can be weakly hydrophobic without any collector attached.
For non-sulfide (e.g., oxide) mineral flotation, the range of Flotation Collectors used is again quite variable, depending on the ore type and operating conditions. The hydrocarbon chain length is typically from 12 carbon atoms to 22 carbon atoms. In the case of non-sulfide flotation, the dosages are somewhat higher than in sulfide mineral flotation, with amounts of 0.1 to 1.0 kg/ton being common. It is interesting to note that in the case of collectors of Group 3 and 4, the hydrophobicity imparted by the collector requires a collector with a much larger hydrocarbon backbone than in the thiol collectors used for sulfide minerals. In almost all cases, these non-sulfide collectors will have at least 10 carbon atoms; the single most common anionic collector is oleic acid (or its salts) with 18 carbon atoms. likewise, the cationic amines used in industry typically will also contain 12 to 18 carbon atoms; in a few cases, even more.
Extensive hydrogen bonding of water molecules occurs on oxide and silicate surfaces causing them to be highly hydrophilic. Thus, while the theoretical mechanism of attachment of collectors to non-sulfide minerals can be straightforward, involving simple charge neutralization between the mineral surface and the collector, the burden of these collectors to impart the necessary hydrophobicity is also greater than with collectors on sulfide minerals. This situation explains the greater dosage required of Group 3 and Group 4 collectors than with thiol collectors on sulfide minerals.
[/fusion_builder_column][fusion_builder_column type=”1_1″ background_position=”left top” background_color=”” border_size=”” border_color=”” border_style=”solid” spacing=”yes” background_image=”” background_repeat=”no-repeat” padding=”” margin_top=”0px” margin_bottom=”0px” class=”” id=”” animation_type=”” animation_speed=”0.3″ animation_direction=”left” hide_on_mobile=”no” center_content=”no” min_height=”none”]

The higher number of carbon atoms required also partially explains the often-observed poorer selectivity of valuable minerals over gangue in non-sulfide mineral systems. Often, the desired non-sulfide mineral and the undesired gangue will have similar surface charges Also, with nonsulfide minerals, the charged collectors (and/or simple hydrocarbon extenders) must be at the particle surface in sufficient dosage so that there may be extra reinforcement of the particle hydrophobicity through micelle formation. As will be described later, this practice can have a negative impact on flotation rate. Improving the selectivity of a nonsulfide ore system in flotation is a major area of new chemical research.
By far, the most important industrial anionic collectors for nonsulfide minerals are the car- boxylic acids (and their salts), with a special preference for the partially unsaturated fatty adds containing 18 carbon atoms (e.g., oldc, linoldc, or linolenic adds). Fatty adds dissociate in aqueous solutions into negatively charged carboxylate ions RCOOH(aqueous) = H+ =RCOO- with pKa values ranging from 5 to 6. At pH’s less than 5, the add is undissolved and not effective for attachment to positively charged minerals. Above a pH of 6, the add is ionized; thus, the range of effectiveness of the collector is limited specifically to a pH of 5 to 6 or high¬er. This pH range, of course, must then be matched to the specific surface charge exhibited by the particular material under study at that pH to allow for electrostatic attraction of opposite charges to occur. The advantage of using sulfonic adds (or their salts) is that they are stronger than carboxylic adds. Hence, they dissociate at a much lower pH range (approximately at a pH of 2), giving them a wider window of potential use as collectors.
With carboxylic acid collectors, the presence of multivalent cations, especially Ca++ ls quite detrimental because insoluble carboxylate salts which do not act as collectors are formed with calcium. Such flotations in hard water require even higher collector dosages to be effective because the initial carboxylic acid dosage is essentially involved in just cleaning up the water hardness.
[/fusion_builder_column][fusion_builder_column type=”1_1″ background_position=”left top” background_color=”” border_size=”” border_color=”” border_style=”solid” spacing=”yes” background_image=”” background_repeat=”no-repeat” padding=”” margin_top=”0px” margin_bottom=”0px” class=”” id=”” animation_type=”” animation_speed=”0.3″ animation_direction=”left” hide_on_mobile=”no” center_content=”no” min_height=”none”]
The use of industrially viable cationic Flotation Collectors is limited almost totally to amines which ionize in aqueous solution by protonation RNH2 (aqueous) + H2O = RNH3+ +OH-. The actual form of the amines used is quite variable, including primary, secondary, ternary, and quaternary forms. Quarternary amines are strong bases, so they are ionized over essentially the complete range of pH, while the ionization of the remaining amine forms is pH- dependent. Typically, for a primary amine, the ionization starts to foil off in the range of pH 10 or greater, thus defining the window of potential use as a collector to be less than pH 10. The major practical use of amine collectors is in the flotation of silica (quartz) gangue particles away from other materials, a process denoted as reverse flotation.
It is also important to point out that in many non-sulfide flotation systems, regardless of the components to be separated, the actual chemical reagent schemes used industrially may be much more complex than a mechanism of simple physical or electrostatic adsorption. Each specific industrial system needs to be evaluated scientifically as a unique system, starting with the simple mechanisms of physical adsorption, chemical absorption, and so on.
Flotation Collectors
At one time the only available method of increasing the floatability of the minerals was by means of collecting or “ oiling ” reagents, which consisted of oily substances such as coal and wood tars, heavy fuel oils, and various creosotes. A considerable amount of agitation is needed to emulsify these reagents in the flotation machines before their absorption at the mineral/water interfaces becomes possible. Such substances are seldom selective since they often affect not only the valuable minerals but also imperfectly deflocculated gangue and unwanted mineral particles, yielding a low grade of concentrate. The present tendency is to replace them partly or wholly by the more selective chemical promoting reagents, which, incidentally, require much less agitation for their effective action. Nevertheless, this may not be possible in all instances ; cases exist of ores whose minerals are not affected by chemical promoters but which adsorb collecting oils if thoroughly emulsified. There is a considerable range of coal and wood tars and their cresotes available for such ores. Whilst occasionally a locally produced tar product may be utilized, it is usually preferable to employ one manufactured to a definite specification for flotation purposes. The consumption of these tarry reagents often exceeds one pound per ton of ore.
Even when chemical promoters are used, the presence of a small quantity of one of the collecting oils is often found to give a tougher and more stable froth than would otherwise be obtainable. Coal-tar creosote is generally employed for this purpose in amounts which seldom exceed 0.5 lb. per ton of ore. The use of wood-tar creosotes is less common, being confined almost entirely to the flotation of lead ores and tailings.
Flotation Promoters
The term promoting reagent or promoter is used to describe the completely soluble chemicals which are gradually replacing the older collecting or oiling reagents. These terms are used somewhat indiscriminately, however, and it is not unusual to group both types under the single heading of “ collectors ”, although the word “ promoter ” better describes the chemical reagents from the standpoint of modern practice. They may be divided into three main groups : (1) The xanthates, (2) thio-carbanilide, and (3) phospho-cresylic acid and its derivatives.
The xanthates are in general the most powerful of the promoting reagents. Potassium ethyl xanthate, CS.SK.OC2H5, is the commonest of the group, but the employment is increasing rapidly of xanthates in which the ethyl radicle, C2H5, is replaced with one of a higher alcohol, notably of a benzyl, propyl, butyl, or amyl alcohol. These reagents are made with a sodium or a potassium base. The use of sodium methyl xanthate, which resembles the ethyl salt in its action, is not very common.
The characteristic of the ethyl and methyl xanthates is that, although they promote the flotation of most of the common sulphide minerals, they first attack the surface which is most highly flocculated. By carefully controlling the amount added to the pulp the promoting action can thus be made to a large extent selective. The higher xanthates are more powerful than the ethyl and methyl salts, but are as a rule less selective. While their greatest application. is in the flotation of sulphide minerals, they are often effective with oxidized minerals, especially with copper carbonates, which are generally unaffected by the lower xanthates. In the selective flotation of a clean sulphide ore, however, the action of a higher xanthate is often so powerful that accurate control becomes difficult and dual control with ethyl xanthate is necessary in order to prevent the presence of unwanted mineral in the concentrate. When this method is practised, no more of the higher xanthate is added than is needed to float the minerals that are not attacked by the ethyl salt; the latter reagent is added at the same time in sufficient quantity to give the greatest possible recovery of the valuable minerals without bringing up those that are not wanted. Thus flotation is controlled by the more selective ethyl xanthate without losing the advantage of the powerful action of the other.
The xanthates generally work best in a slightly alkaline circuit. Although the alkalinity has not as a rule to be regulated very accurately, it is usual nevertheless to maintain it at the value which is known to give the best results by periodical pH determinations.
All the xanthates are very soluble in water and are added to the circuit in the form of 10 to 20% Solutions. Their consumption on the average is low, ranging normally from 0.05 to 0.15 lb. per ton of ore.
Thio-carbanilide is the commercial name for di-phenyl-thio-urea, CS(NH.C6H5)2. It is a solid that is almost insoluble in water but dissolves in ortho-toluidine giving a solution known as “ T.T. Mixture ”, which generally contains 15% of thio-carbanilide and 85% of the solvent. In this form it was very common as a promoter before the introduction of the xanthates, but it is not so popular now because the latter reagents provide a more sensitive method of control and have a wider range of application. Thio-carbanilide, however, has comparatively little action on pyrite, for which reason it is often used to promote the flotation of galena in the presence of sphalerite and pyrite in cases when xanthate, if added in sufficient quantity to float the lead minerals, brings up too much of the others, in spite of the fact that they may previously have been rendered unfloatable by a depressing reagent. For such a purpose thio- carbanilide is generally added as a solid in the ball mill, which provides enough agitation to dissolve it without the use of a solvent. As long as a certain maximum amount is not exceeded, small alterations in the quantity added do not cause corresponding variations in the proportion sphalerite and pyrite which float with the galena—that is, control by thio-carbanilide is not so delicate as by xanthate. The consumption normally ranges from 0.05 to 0.2 lb. per ton of ore.
Phospho-cresylic acid, or di-cresyl-di-thio-phosphoric acid, HS.SP(OC6H4.CH3)2, is a compound made by the American Cyanamid Company by treating cresylic acid with phosphorus penta-sulphide, and is known commercially as “ Aerofloat ”. It is marketed as a liquid in three forms, Aerofloat “ 15 ”, “ 25 ”, and “ 31 ”, the figure representing the promoter strength of the reagent in terms of phosphorus penta- sulphide. It always contains some free cresylic acid. Aerofloat resembles thio-carbanilide in its lack of sensitivity and its slight action on pyrite as compared with xanthate. It is, therefore, a useful promoter for pyritic ores in circuits in which a xanthate, if added in sufficient quantity to float the valuable minerals, brings up too much pyrite as well. It is generally used in preference to thio-carbanilide for this purpose, except for the flotation of galena in a lead-zinc ore, when the latter reagent is usually preferred. As a general rule aerofloat works best in an almost neutral circuit. There is often no difference between its action and that of a xanthate in the flotation of a straightforward ore which does not contain much pyrite or other unwanted mineral and therefore does not need very accurate control. Aerofloat, however, sometimes has the advantage that it can be used in a neutral circuit, whereas xanthate may require a higher degree of alkalinity to give the same result with the consequent necessity for the addition of a certain amount of lime or soda ash.
The normal consumption of aerofloat varies from 0.1 to 0.3 lb. per ton of ore. It is very corrosive and should preferably be added from a covered feeder made of special metal. Since it already contains cresylic acid, which is a frother, it is diluted when necessary in the feeder with no more of the latter reagent than is needed for the production of the froth. Even when not diluted in this way it still contains cresylic acid, which may interfere with flotation should it happen that the promoting properties of aerofloat are needed in a circuit which already contains enough frothing reagent or which requires one of a different type.
Sodium aerofloat, or sodium di-cresyl-di-thio-phosphate, was introduced to meet such a case. It is a solid, soluble in water, consisting mainly of the active constituents of aerofloat combined with caustic soda. Its action as a promoter is not as powerful as that of the acid, but it is often employed in conjunction with ethyl xanthate for the flotation of ores containing pyrite, when for one of the reasons already mentioned aerofloat itself is not suitable. The dual control method, similarly to that used for the addition of two xanthates, is generally adopted in such cases ; sodium aerofloat is added to bring up the minerals for which it is especially suitable, the final recovery being controlled by the xanthate, the latter addition being regulated to bring up as much as possible of the remaining valuable mineral with the minimum amount of pyrite.
Another aerofloat derivative, sodium di-ethyl-di-thio-phosphate (American Cyanamid Company’s Reagent “ 208 ”), is to a certain extent replacing sodium aerofloat in the flotation of sulphide copper minerals, its mode of addition in conjunction with xanthate being the same as that of the other reagent. Di-thio-phosphates of the higher alcohols are also made and can sometimes be used with advantage in place of the cresyl and ethyl compounds. These reagents are best made up in the form of 5 to 10% Solutions. The amount needed in the circuit is very small, usually between 0.01 and 0.05 lb. per ton of ore.
Chemical promoting reagents are, in general, more powerful and more selective than collecting oils, and, with the exception of aerofloat, they have the advantage that their control is separate from that of the frothing reagents. For these two reasons, in cases where they can be employed with advantage partly or wholly to replace collecting oils, it is usually found that a better recovery of the valuable minerals is obtained in a drier froth and a higher grade concentrate. More care must be exercised, however, in controlling their additions than is necessary with collecting oils, since any excess will nearly always bring up unwanted mineral which may be very difficult to remove in subsequent cleaning operations.

