The Gold Fire Assay Process of ores is almost universally conducted in the dry procedure by furnace methods. Exceptions will be noted later. The plan of operation is to concentrate the precious metal in a button of lead either (1) Fire Assay By fusion in a crucible; or, more rarely, (2) Fire Assay By scorification. The button of lead obtained by either method is then subjected to cupellation, by which the lead is oxidized and removed, and the resulting bead of precious metal is weighed. In these operations, silver and the metals of the platinum group remain with the gold, and are subsequently separated by inquartation and parting, and, in the case of platinum and its allies, by further special methods.
The exact method to be used in any particular case varies with the composition of the ore. As a general rule, gold ores are better assayed by the fusion process, so that a comparatively large quantity of material may be operated on. Very rich ores may be assayed by scorification, the errors arising from the small amount of material used in the process being less important in their case.
The assay by scorification was preferred to pot-fusion by the German assayers of long ago. When fusion in a crucible was recommended, it was only as a preliminary to scorification. Ercker describes many of the methods and precautions are given in this chapter, such, for instance, as the treatment of cupriferous ores with nitric acid, the use of a salt cover in the crucible, and of “ proof-Centners,” which correspond to assay-tons. He also mentions the forerunner of the buck-board, and instances assay-offices in which 200 assays of ores were made in a week.
In many ores, both gold and auriferous silver occur natively in grains or threads. These “metallics” are not readily reducible to a fine state of division, and, though a part always passes through the sieve, some of the larger pieces which have resisted abrasion fail to do so. In some assay offices, part of the pulverized ore is thrown back into the mortar with the metallics, and grinding is continued until everything passes the sieve.
Sampling the Ore and Preparation of the Sample for Fire Assay
This is a dangerous practice, as it is impossible to ensure the even distribution of metallics through the sample. The smaller pieces which pass through the sieve in the first instance constitute an unavoidable evil that is increased by every piece of metal that follows them. The safer plan is to cupel by themselves the whole of the metallics left on the sieve and calculate their value per ton of ore independently of the result obtained by the ordinary assay. The total value of the ore is found by adding these two results together.
The prepared sample may be mixed and divided by the Bridgman divider, and is stored in tin boxes or glass jars, which should be labelled by numbers, none of which are ever repeated. Before weighing out the powdered ore for assay, the whole sample should be turned out into a wide bowl or on to glazed paper, or, better still, rubber cloth (which does not crack and wear out like paper), and thoroughly mixed with a spatula. The sample should never be mixed by shaking, and care should be taken to avoid jarring it after mixing, as metallics in that case tend to settle to the bottom from their superior density, and a fair sample cannot easily be obtained. For the same reason the part of the mixed sample which is taken for assay should not be hastily shovelled on to the balance pan from the top of the pile, but a vertical slice should be taken, some of the lowest layer being carefully scraped up from the rubber cloth.
Method of Gold Fire Assay by Fusion or Crucible
This Gold Fire Assay Process is divided into four parts:
- Fusion;
- Cupellation;
- Parting;
- Weighing the gold.
Gold Fire Assay Process
The ore is weighed accurately to 0.01 gramme unless very poor when a less exact approximation is sufficient. The charcoal is always weighed with great care; the litharge is best measured by a ladle or shot measurer; the fluxes may also be measured out by a ladle more rapidly than they can be weighed. The various ingredients are thoroughly mixed together on rubber cloth or in the crucible in which the fusion is to take place. Part of the borax is often kept separate and used as a cover, being put on the top of the rest of the charge after transference to the crucible.
The crucible is carefully annealed in the ashpit of the furnace before using. It is lowered into a hollow in the fuel of the furnace made by piling the coke round an old pot and then carefully withdrawing the latter. Both ordinary and basket tongs are useful. The fire should be at a low red heat on charging-in— that is, at a temperature of 600° or 700°—and should not be urged at first. The objects to be obtained are as follows:
- The gold is to be brought into such a condition that it will be readily taken up by the molten lead before the subsidence of the latter to the bottom of the pot.
- The mixture is to be finally brought to a state of quiet fusion, with low viscosity, so that the reduced lead may subside completely and collect into one button.
- There must be no mechanical losses.
Reduction of the lead begins below a red heat, and is probably almost complete before borax glass and carbonate of soda begin to melt (at about 700°) and to attack the gangue. In an ordinary charge which was heated in a muffle at 640° for twenty minutes, and then withdrawn and allowed to cool, it was found that fusion had begun though no effervescence had occurred. No lead was visible until after the charge had been panned, when a large amount of metallic lead in fine particles was recovered. The lead begins to collect into globules visible to the naked eye as soon as the charge begins to work and effervesce, but does not sink until the effervescence has proceeded for several minutes. The slower the melting the more chance is afforded of bringing the lead into intimate contact with all parts of the pasty mass and of collecting the gold from the ore. Also the slower the melting, the less chance there is that part of the charge will be projected from the pot. The final temperature need not, as a rule, be above 1,100°, and may be lower in certain cases.
The influence of the rate of heating on the result is very great. In an experiment designed to test this, four charges of a pyritic ore were made up in accordance with the table given above. Two charges were heated very slowly, and remained in the furnace for fifty minutes before fusion was complete. The other two charges were heated as rapidly as possible, and were in the furnace for only twenty minutes. The results were as follows:


The mean deficiencies in the charges heated rapidly were thus: Gold, 34%; silver, 42%, but the results were also less regular than when slow heating was used. The effervescence is due chiefly to the escape of carbonic acid from the sodium carbonate as it unites with silica, and is of value in moving about the lead globules.
After a lapse of from 40 to 50 minutes the charge is in a state of tranquil fusion, with the exception perhaps of slight action round the sides or next the iron, if any is used. The crucible is then lifted out of the fire by the tongs, the nails withdrawn and any lead adhering to them shaken off into the pot. The pot may now be tapped on the floor to assist the lead to settle in the slag, allowed to cool and broken by a hammer to extract the lead button, or the charge may be at once poured into an iron mould. The mould must be cleaned, blackleaded, and warmed before being used, and, after pouring, it is tapped on the bench to collect the lead at the bottom.
If the charge does not fuse completely, so that the slag is pasty or has lumps in it, it is advisable to recommence the assay, making such alterations in the charge as experience suggests.
When the mould is quite cold its contents are readily separated from it if the precautions mentioned above are taken, and the slag is detached from the lead button with a hammer on an anvil. The slag should be glassy and homogeneous; if it is streaked it is probable that the fusion has not been perfect. It is green and transparent if the ore is nearly pure quartz; black and opaque if much iron is present; but is red if the ore contains much copper.
The lead button should be soft and malleable. If it is hard or brittle it may contain sulphur, arsenic, antimony, copper. Sulphur and arsenic are kept from entering the lead by the addition of iron, and may form with the iron a regulus or spiess which separates as hard blackish-grey or white layers found just above the lead. They are richer than the slag, and may often yield appreciable quantities of gold on further treatment by scorification or roasting and fusion. If the quantity of sodium carbonate is large enough, the sulphur is retained in the slag in the form of sodium and ferrous sulphides. Antimony makes the lead hard, white, brittle and sonorous; according to Rivot, it is not a source of loss if forming less than 1 per cent, of the lead button (see under Cupellation). The lead must be completely freed from the slag, very small quantities of the latter interfering seriously with the cupellation, forming a scoria, and occasioning loss.
FLUX RECIPES Cleaning the Slag Blowpipe Assay The Treatment of Base Ores
ASSAY BY SCORIFICATION Detection of Gold in Minerals SPECIAL METHODS OF GOLD ASSAY
Fire Assaying Gold Alloys
It is often impracticable to apply the ordinary parting assay to the examination of low-standard alloys of gold with other metals. These are then tested by various other methods, of which a summary is given below, the alloys being grouped in four series for convenience:
A. Alloys requiring scorification.
B. Amalgams.
C. Alloys containing members of the platinum group.
D. Tellurium compounds.
Scorification of Alloys

Alloys of arsenic or antimony are reduced to a fine powder and scorified with thirty parts of lead and a half part of borax. If the slag becomes pasty towards the end of the operation more borax is added, a little at a time. If the lead button obtained is hard, a second scorification is necessary, with the addition of more lead. There is some loss of gold in the slag.
- Iron or Manganese Alloys
The operation is tedious and difficult with these alloys, as they are difficult to fuse, having higher melting points than pure gold, and the oxides of iron do not form easily fusible compounds with the litharge. An extremely high temperature is required; ten parts of lead, one of borax, and one of silica usually suffice.
- Cobalt and Nickel
Twenty parts of lead are used, but no borax at first, so that the oxidation of the nickel may not be hindered. A very high temperature and the subsequent addition of two parts of borax are necessary. Several successive scorifications are required as nickel and cobalt are difficult to oxidise. - Zinc
Oxide of zinc does not form a fusible mixture with litharge, and the slag is only rendered pasty by borax, unless it is added in large quantities. Gold is lost in the slag, but the loss is minimised by slagging off the zinc as rapidly as possible. Use fifteen to twenty parts of lead and two to three parts of silica, with a little borax. - Tin
Twenty parts of lead are required ; oxide of tin is rapidly formed, but the slag is not easily fusible. Large amounts of borax are necessary, or still better, borax mixed with potash which forms a fusible stannate with SnO2. - Aluminium
Alloys containing this metal cannot be assayed by scorification and cupellation. As soon as fusion takes place in the muffle, aluminium floats to the top of the bath, being of low density, and is rapidly oxidised, producing alumina which forms an exceedingly infusible scoria not easily removed by litharge. The production of the latter, moreover, is checked by the scum.
Mr. Edward Matthey observes that the removal of aluminium by digestion in hydrochloric acid, and collection of the residual gold, does not yield satisfactorily results. The process he recommends is as follows:
Accurately weighed portions of 50 grains each of the alloys are fused with litharge, under a flux of potassium carbonate and borax with a small proportion of powdered charcoal, and the resulting slag re-fused with a further small quantity of litharge and powdered charcoal. The lead buttons containing all the gold (the aluminium having combined with the fluxes employed) are cupelled, and the resulting gold cupelled with silver and parted with nitric acid in the usual manner. The assays must be worked with checks or standards of fine gold and pure aluminium. In the majority of the preceding cases it is better to analyse the alloys by wet methods.
Fire Assaying of Amalgams
The alloy is placed in a weighed porcelain crucible and gradually heated so as to drive off the mercury. After the greater part of the mercury has been driven off the temperature is raised to a full red heat which is maintained for half an hour. About 0.1 per cent, of mercury still remains in the gold after this treatment and can only be completely removed by cupellation and parting. Checks must be used, as the loss of gold in the operation may amount to 1 per 1,000.
A better method is to dissolve the mercury in nitric acid mixed with an equal volume of water. A porcelain crucible or basin should be used and gentle heat applied. If the action is not too violent the gold does not break up, but is left as a spongy coherent mass. It is washed, first with nitric acid, and finally with water, and ignited. Mercury may be made use of to collect very finely-divided precipitated or parted gold into a coherent mass, the mercury being removed by nitric acid.
Fire Assaying Platinum Group Elements
Cupellation must be performed at a higher temperature than usual, and the iridescent bands are seen to remain longer, although they are less numerous.
Platinum Gold Alloys Fire Assaying
The button obtained by cupellation is dull and crystalline. If the alloy contains as much as 7 or 8 per cent, of platinum the cupellation proceeds slowly, brightening is only obtained at a very high temperature and the button appears flattened, and has a rough crystalline surface and a grey colour. If more than 10 per cent, is present, brightening does not occur at all, and the other features just mentioned are more strikingly exhibited. On parting, the platinum is partly dissolved with the silver, but the assay-piece must be boiled in acid for a long time, and the parting is incomplete. A second and even a third parting may be necessary. Unless proofs are made up of similar composition the results are not satisfactory if more than 1 or 2 parts of platinum are present per 1,000 of alloy.
According to results obtained in the laboratory at the Royal Mint by Prof. Stansfield, it is impossible to free buttons containing platinum from lead at the ordinary temperatures attainable in a muffle. It is necessary to employ the oxy- hydrogen blowpipe for the purpose, with the result that the losses are irregular. In the cupellation of pure platinum, buttons weighing from 0.002 to 0.01 gramme retain about 10 per cent, of their weight of lead, and those weighing from 0.04 to 2 grammes retain about 33 per cent. f parting is effected by boiling in concentrated sulphuric acid instead of in nitric acid, almost all the platinum remains undissolved with the gold. Chaudet recommends the following proportions of silver for this purpose:
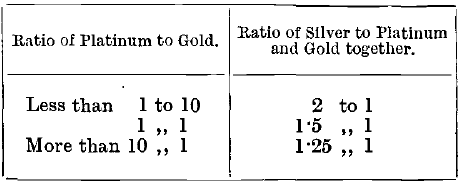
The reason for the reduction, shown in the table, in the amount of silver when the platinum is increased is that, according to Chaudet and Haindl, considerable quantities of platinum are dissolved, and the error caused this is corrected by leaving some silver undissolved in the cornet. The losses of platinum, however, are very irregular, and the method is not a good one. The reason for the variation is probably the lack of homogeneity in the buttons and prills, as gold-platinum alloys show a considerable amount of liquation.
The platinum is not dissolved by nitric acid if zinc or cadmium is substituted for silver.
Mr. Edward Matthey recommends the following method for alloys of platinum 900, and gold 100 parts :—50 grains each are taken of the alloys and treated with an excess of nitrohydrochloric acid which gradually dissolves the whole. The resulting solutions of platinum-gold chloride are then evaporated nearly to dryness to drive off the free acid and diluted with distilled water to about 20 c.c., a degree of strength ascertained by experiment to be the best for the precipitation of the gold. The metallic gold is thrown down by means of crystals of oxalic acid, and is carefully washed, dried, and weighed. It is necessary to use checks.
The estimation of platinum in its alloys with gold and silver has also been studied by M. Forest, assayer at the French Mint.
Fire Assaying Palladium-Gold Alloys
The palladium is dissolved in parting if the weight of silver is at least three times that of the gold, yielding an orange coloured solution. Matthey recommends double parting. Separation may also be affected by fusion with six to eight parts of potassium bisulphate, and dissolving out the dark brown palladious sulphate by boiling water. A second fusion is usually necessary to render the gold residue quite pure.
As in the case of platinum, palladium may be separated from gold by dissolving the alloy in aqua regia, evaporating to dryness, taking up with water and precipitating the gold by oxalic acid from a very dilute solution : the palladium remains in solution.
Fire Assaying Rhodium and Iridium-Gold Alloys
Iridium, if present, always sinks to the bottom of the cupelled button as it is very dense (specific gravity = 21.38), and is not usually fused at the temperature of the muffle, but occurs in the state of fine black crystalline particles. Hence, when the button is rolled into a cornet with the lower face outwards iridium occurs as black sooty spots or streaks which are seen by a lens to fill up depressions in the surface of the gold.
Both rhodium and iridium are almost insoluble in aqua regia. If gold alloys containing both of them are parted in the ordinary way with nitric acid, only a small quantity of rhodium goes into solution with the silver. The residue consisting of the gold, the iridium and most of the rhodium may then be attacked by dilute aqua regia when the gold is dissolved together with only traces of the other metals. These may be separated by evaporating the solution to dryness and heating to dull redness, when the reduced metals being no longer alloyed may be completely separated by dissolving the gold in aqua regia. Iridium may be separated from gold by fusing it with fluxes, the charge being made up as follows:
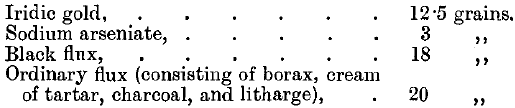
The iridium forms a speiss with the iron and the arsenic, and the lead button formed at the bottom of the fused mass contains all the gold. The estimation of osmium, iridium, and ruthenium in gold bullion has also been studied by Riche, Leidie, and Quennessen, assayers at the French Mint.
Assaying Tellurium Compounds
These must be treated by wet methods. The alloy may be dissolved in nitrohydrochloric acid, and the solution containing both gold and tellurium evaporated with a large excess of hydrochloric acid until no more chlorine is given off, when both gold and tellurium are readily precipitated by a current of sulphur dioxide gas. On attacking the precipitate with dilute nitric acid the tellurium is dissolved in the state of tellurous acid, and the gold residue may be dried and weighed, and its purity ascertained by inquartation and parting. The greater part of the tellurium may be removed from gold-tellurium alloys by boiling in nitric acid, and the residue can be cupelled and parted with very little loss of gold.
Wet Assay Methods of Gold Alloys & Compounds
Assays or complete analyses of gold bullion, natural minerals, can be made by the ordinary chemical methods given by Fresenius, Crookes, and others. From 1 to 5 grammes of bullion are usually enough, but a much larger amount is necessary if the alloy is nearly pure gold. M. Forest takes 300 grammes of gold bullion when examining it for small quantities of impurities. In general the residue left after prolonged action of nitric or sulphuric acid is not sufficiently pure to weigh as gold, and complete solution in aqua regia is usually necessary. From the solution the gold may be precipitated by:
- (a) ferrous sulphate,
- (b) sulphurous acid,
- (c) oxalic acid,
- (d) sulphuretted hydrogen,
- (e) ammonic sulphide, followed by the addition of hydrochloric acid.
The following remarks may be of value in aiding the chemist in his choice of a precipitant in any particular case. Nitric acid must always be expelled from the solution by warming with successive additions of hydrochloric acid. The acid solution must not be heated too strongly or loss of gold chloride by volatilisation occurs. Some other chlorides escape more freely. Ferrous sulphate and sulphurous acid act well in strongly acid (HCl) solutions ; oxalic acid, sulphuretted hydrogen, and ammonic sulphide best in presence of small quantities of HCl. The solution should be dilute (say 1 part of gold in 300 of water), so that other metals may not be carried down by the gold. Sulphate of iron gives a very finely-divided precipitate which is difficult to wash by decantation without loss ; precipitation is slow in cold solutions. Oxalic acid causes plates and scales to form which are readily washed and are very pure; it acts best in boiling liquids, but a temperature of 80° for forty- eight hours suffices; in the cold or in the presence of much hydrochloric acid or alkaline chlorides the action is very slow and partial; a large excess of the precipitant must be present. Oxalic acid is used for solutions containing metals of the platinum group, which are not precipitated by it. Alkaline oxalates act better than the free acid.
Sulphurous acid is an excellent precipitant for most solutions. It acts rapidly and completely in the cold, and does not readily precipitate other metals, except tellurium. Sulphuretted hydrogen is used in the absence of all other metals whose sulphides are insoluble in hydrochloric acid.
In all cases careful consideration must be given to the nature of the base metals present, and the precipitant which will not render any of them insoluble must be selected.

