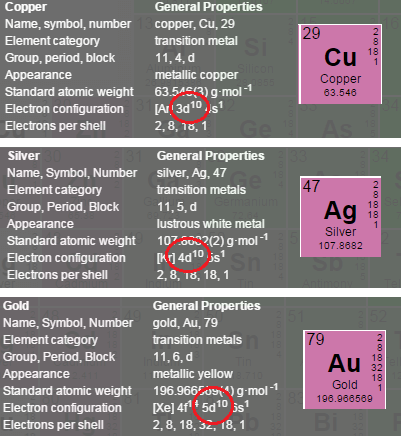
With regards to the aqueous chemistry of gold and the various states of gold oxidation ( 5, 3, 2, 1, −1 (an amphoteric oxide)), we must look at the electronic presentation typical of transition metal elements (Copper, Silver, Gold) AKA Group 11 element from the periodic table, because they allow for the extraction of multi electrons. As shown in the red circle of Cu/Ag/Au below, the difference of energy needed to go from electron ns to (n-1)d is low. Typically, group 11, is where we come upon Gold Oxidation States +1, +2, +3.
 |
Gold Oxidation State I: In group 11, only silver can create an aqueous ion in oxidation state I. Cu (I) and Au (I) recombines unproportionately in aqueous solution unless they be in restricted solubility like an Au (I) VS a highly complex Au(CN)2-,
The complex anion, Au(CN)2-, is produced when gold is dissolved as part of the cyanide process. This aurocyanide complex has has a highly stable constant (200,000,000,000,000,000,000,000,000,000,000,000,000) is so much stable over the common copper or silver cyanide complex used in cyanidation of gold processes. They are other gold based complex ions we can be interested in for gold extraction metallurgy such as thiourea, chloride, and thiosulfate.
Gold Oxidation State II: Paramount in copper chemistry, not so much in silver chemistry and rarely in gold chemistry, is the importance of Oxidation State II. The ultra rare Au(II) may occur as a temporary bridge during reaction from Au(II) and Au (II); a redox process. Au (II) can also be seen on gold-gold strongly bonded compounds and finally, it may complex with some unsaturated sulfur compounds.
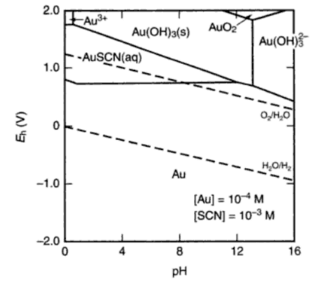
Gold Oxidation State III: This is the most significant gold oxidation state that we seldom see applied on silver or copper. The gold-cyanide complex [Au(CN)4]- (auricyanide) like its counterpart Au(CN)2- is ultra stable (100,000,000,000,000,000,000,000,000,000,000,000,000, 000,000,000,000,000,000). Few examples of stable Au (III) complexes exist but do include thiocyanate [Au(SCN)4]-, ammonia [Au(NH3)4] and chloride [AuCl4]- complexes.
 |
 |
 |
Oxidation State V: With a good Lab, you could prepare an AuF5 compound and have a potent oxidizer without any parallel for silver or copper.
How do you determine what is the oxidation state of gold:
Starting with Auric complexes being the strong oxidizers, the primary or most common gold oxidation states are for pretty much non-existent Aurous and Auric ions – Aurous (Au1+) and Auric (Au3+). Gold will covalently bond and compound generally in complexes.