The concentration of low grade cinnabar, HgS, with a specific gravity of 8.0-8.2 is effectively accomplished by gravity and flotation methods.
Cinnabar ores of relatively high mercury content can be retorted directly to expel the sulphur and condense metallic mercury, but for lower grade ores, preliminary concentration is advisable to reduce the tonnage for retorting.
Retorting and recovery of metallic mercury is widely practiced at small or large tonnage mines. Cinnabar decomposes, yielding mercury at a temperature from 500 to 600°C. The ore in question is assumed to contain cinnabar associated with a siliceous gangue. Grinding to minus 48 mesh is the economic liberation point, but considerable free cinnabar is released at about 10 mesh and warrants concentrating the mineral as soon as it is freed.
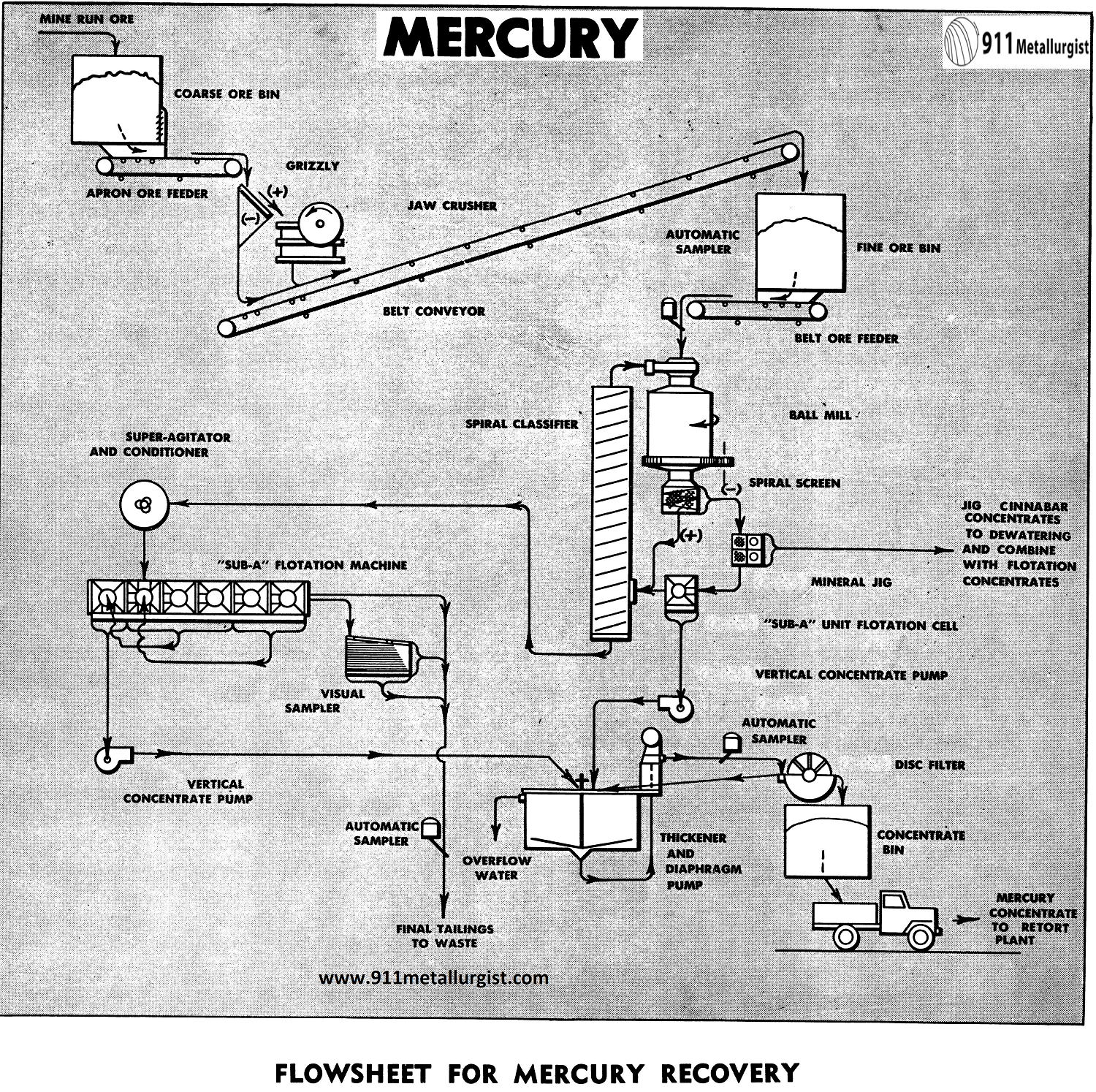
A Mercury Ore Beneficiation Process Flowsheet
Crushing Circuit
Conventional single stage crushing comprises this section, since the tonnage is less than 100 tons per day. The mine ore is fed from the coarse bin by means of a Apron Feeder to the Jaw Crusher. Crusher feed passes over a grizzly with an opening similar to the crusher setting. Grizzly undersize and crusher discharge product are delivered to the fine ore bin by means of a belt conveyor. A Adjustable Stroke Belt Ore Feeder delivers the fine ore to the grinding circuit at a uniform rate.
Mercury Ore Grinding Section
The Mineral Jig and Unit Cell are installed in the ball mill-classifier circuit to recover an important amount of the freed cinnabar at a relatively coarse particle size, desirable because of cinnabar’s tendency to be friable and slime easily. In addition, the coarser concentrates are preferred for retorting, since dust losses can be substantially reduced.
The Ball Mill of the grate discharge type uniformly discharges through a Spiral Screen to the Duplex Mineral Jig which recovers a coarse cinnabar concentrate. The jig tailing passes to the Unit Cell, which produces a granular flotation concentrate, while the tailing flows by gravity to the Spiral Classifier. Additional water for the proper classifier dilution is added to the Unit Flotation Cell feed. The classifier overflow, minus 48 mesh, passes to a Conditioner, then to flotation.
Mercury Flotation Section
Conditioned classifier overflow is sent to a 6-cell “Sub-A” Flotation Machine, arranged to provide three rougher cells, two scavenger cells, and one cleaner cell. The rougher flotation froth of cinnabar passes to the cleaner cell,while the scavenger froth is returned to the first rougher cell. This arrangement, including re-circulation of the cleaner tailing, is accomplished without the use of pumps, a typical illustration of the great flexibility of the “Sub-A” Flotation Machine.
 Selective Mineral Jig recovers mineral by gravity as soon as free.
Selective Mineral Jig recovers mineral by gravity as soon as free.
Filtration
The cleaned flotation concentrate and Unit Flotation Cell concentrate are combined, thickened, and filtered. Thickening is optional, depending upon the tonnage and settling characteristics of the concentrates. Since the Unit Cell concentrate is relatively coarse and easily filtered, direct filtration of the combined flotation concentrates, utilizing the Disc Filter is often feasible.
Dewatered Mineral Jig concentrate is combined with the filtered cinnabar flotation concentrate and stored preparatory to retorting and recovery of the metallic mercury.
Sampling
Automatic Samplers are used to sample the feed, concentrate, and tailing. A portion of the final tailing is removed and treated on the Visual Sampler, consisting of a ¾” Suction- Pressure Diaphragm Pump and a No. 13A Wilfley Concentrating Table. The flotation results and conditions may easily be controlled by observing the concentrate streak on the table.

“Sub – A” Unit Flotation Cell in the grinding circuit, recovers mineral by flotation at a coarse size.
Flotation of Mercury
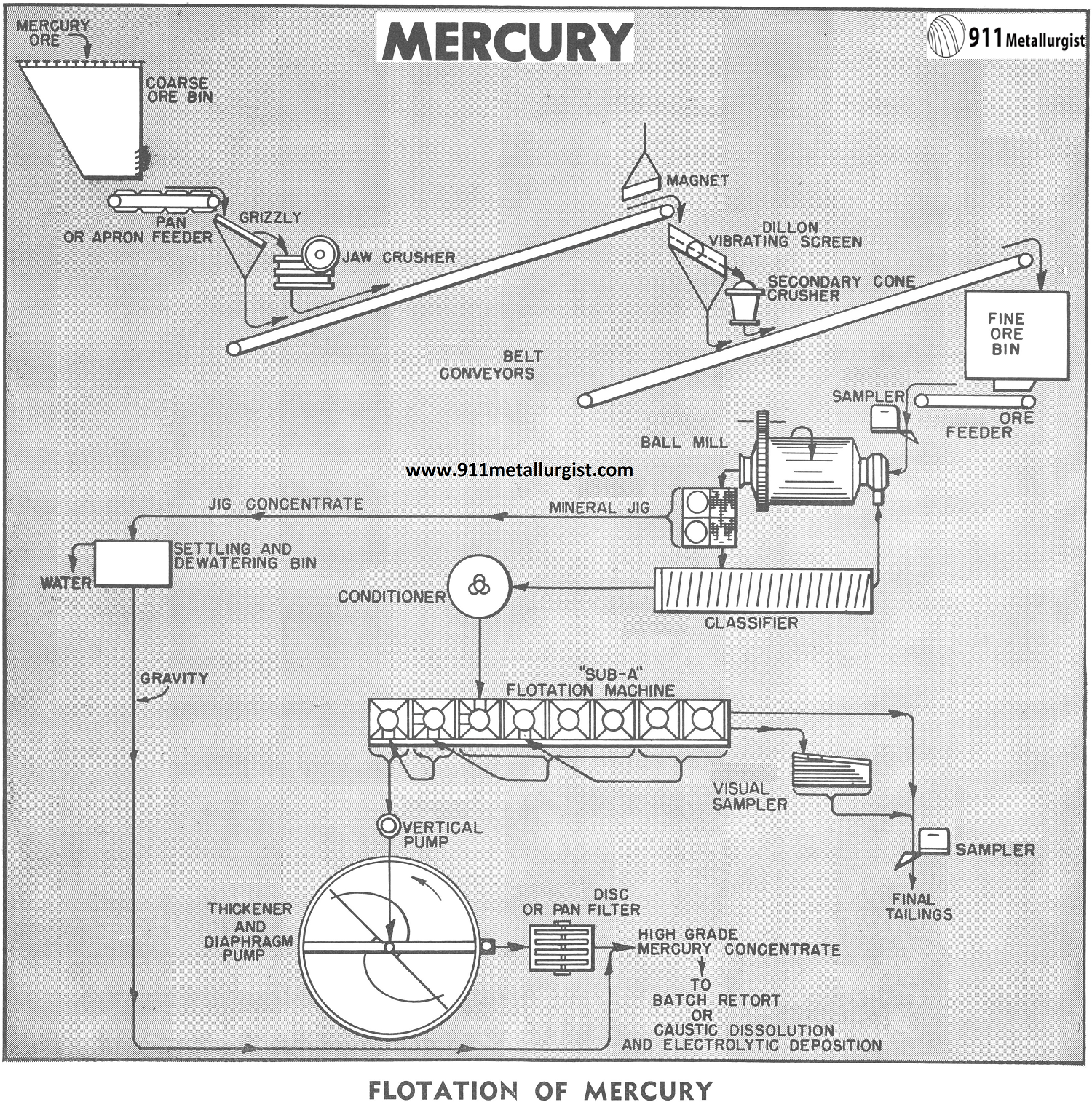
In Flowsheet Study M7-F22 a typical flotation mill for concentration of mercury ores was given. No mention of reagents to effectively float cinnabar was made although normally this mineral is readily floatable.
Since publication of this information plants have gone into operation in Nevada, Oregon, and Idaho which fully demonstrated the advantages of concentrating low grade mercury ores by flotation. Experience has also shown that particular attention must be given to the selection of the proper flotation reagents for most effective results and high recoveries.
The Mercury Ore Beneficiation by Flotation
The flowsheet given above is typical for the average mercury concentrator. Two stage crushing followed by grinding in closed circuit with a classifier is used to reduce the ore down to flotation size, 48 to 65 mesh. Many ores contain granular particles of cinnabar recoverable by gravity. The Mineral Jig is incorporated in the mill circuit to scalp out this coarse heavy mineral and any metallic mercury which may be present in the ore.
Flotation with one or more stages of cleaning is used to float the finely ground mercury minerals into a high grade concentrate. It is not uncommon to produce concentrate grades of 40 to 50% mercury on ores carrying 5 to 15 lbs. of metal per ton. Ratio of concentration is therefore, high. A mill treating 200 tons of 10 lb. mercury ore and making 95% recovery will produce 4750 lbs. of 40% mercury concentrates per day. A thickener and filter is required to dewater the flotation froth concentrate prior to drying and retorting, or chemical dissolution. In small plants the thickener may be eliminated to minimize tie-up of high grade concentrates. Pan Filters have a good application in small plants.
Flotation Reagents
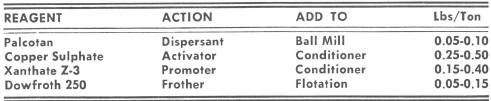
Normally cinnabar floats quite readily with pine oil and a xanthate. A typical reagent combination is given in DECO Bulletin M4-B24 describing the operation at the “Compania Mercurio En Sain Alto and Beneficiadora De Mercurio, S.A.” (Mexico). Here starch or reagent 637 was used to disperse the slimes and depress carbonaceous matter.
Plant operation has shown that dispersants such as soda ash and sodium silicate should be avoided. These reagents have a tendency on certain ores to depress cinnabar and cause low recoveries by flotation. Palcotan, a redwood lignin product, is a very effective slime dispersion reagent. It has no apparent adverse effect on cinnabar. On exposure to the atmosphere after grinding, cinnabar has a tendency to decrease in flotability. This may be due to some surface oxidation. Metal salts such as copper sulphate and lead acetate or nitrate when added in small amounts overcomes this difficulty. Tests should be made, however, to establish the best flotation reagent combination. Frothing may present a problem so each type of ore must be tested to obtain the proper combination and ratio of frother to collector and activator. A typical flotation reagent combination is as follows:
“Sub-A” Flotation Cells recovering cinnabar in mercury flotation mill in Oregon. Better than 90% recoveries are made with concentrates averaging more than 1000 pounds per ton.

Generally pH control is not too critical. A pH varying from 6.5 to 8.5 is satisfactory. The usual pH regulators such as lime, soda ash and sodium silicate should be avoided.
Advantages of Flotation for Mercury Processing
Treatment of low grade mercury ores by flotation show generally a lower capital investment than for a comparable tonnage furnace plant. Operating costs are about equal, however. Advantages of concentration are:
- Higher recoveries on low grade and/or wet, sticky ores. Wet sticky or dusty ores cannot be furnaced efficiently.
- Less health hazard. Mercury fumes are poisonous.
- Tonnage of plant can be more flexible. Plant can start and stop on short notice.
- Higher resale value on flotation equipment. Furnace equipment only applicable on mercury ores.
- Impurities can be rejected from complex ores by flotation; as for example, arsenic.
Batch Retorting—High grade mercury flotation and gravity concentrates can be batch retorted for recovery of the distilled metal. With just a few hundred pounds to be treated, retort operating conditions can be carefully controlled and the normal operating hazards kept to a minimum. DECO drawing A-7930 shows a form of batch retort suitable for handling small quantities of high grade concentrates or high grade ore.
Chemical Treatment and Electrolysis—Considerable experimental and pilot plant work has been done to eliminate retorting and its accompanying hazards. Impurities such as arsenic and antimony also cause operational trouble in any retorting method. Mercury concentrates respond well to caustic leaching with a mixture NaOH and Na2S. Mercury is taken into the caustic solution which is decanted, clarified and treated through a special electrolytic cell. Metallic mercury is tapped from the cell bottom.
It is reported that ore carrying 7 lbs. of mercury per ton can be treated economically at a profit with an overall recovery of 95%. This is by a combination of flotation, caustic leaching, and electrolytic deposition.
Source: This article is a reproduction of an excerpt of “In the Public Domain” documents held in 911Metallurgy Corp’s private library.
