The stamp battery must be regarded from two different points of view, (a) as a crushing machine, (b) as an amalgamating machine, and it should be remembered that the modifications designed to make it a more efficient crusher often reduce its power as an amalgamator, and vice versa.
Stamps were originally designed as crushing machines, and the tendency is just now towards a return to this view of its proper functions. A practice which now seems to be growing in popularity consists briefly in crushing the ore and effecting its discharge from the battery as rapidly as possible. With this object in view heavy stamps are used, running very fast, with a moderate drop; the screens are coarse, the screen area is large and placed as low down as possible, and the mortar is made narrow, with nearly vertical sides. These arrangements all increase the output of the battery. Amalgamated copper plates are either not placed inside the mortar, or there is a plate on the discharge side only. In the extreme case, no mercury is fed into the battery, and the coarsely-crushed ore passes over amalgamated plates and is then re-ground in tube mills, and either again run over the plates or passed at once to the cyanide plant. Under these conditions, the output is very large, and may even be as high as 8 to 12 tons of ore per stamp per day. The percentage of gold extracted by amalgamation is low.
When the stamp battery is used as an amalgamator, the depth of discharge (height of screen above dies) is great, amounting even to 12 inches or more, the screens are finer, the supply of water is reduced, and all efforts are directed to fine crushing in the battery. Mercury is fed into the mortar, and amalgamated plates are put inside the mortar on both feed and discharge sides. The object of these arrangements is to keep the ore in the mortar for a long time, so as to increase the chance of catching the gold on the inside plates. . The duty of the stamps is, of course, greatly diminished, and may be as little as one ton of ore per stamp per day. Under such circumstances a high percentage of gold is amalgamated. Between these two extremes there are many gradations.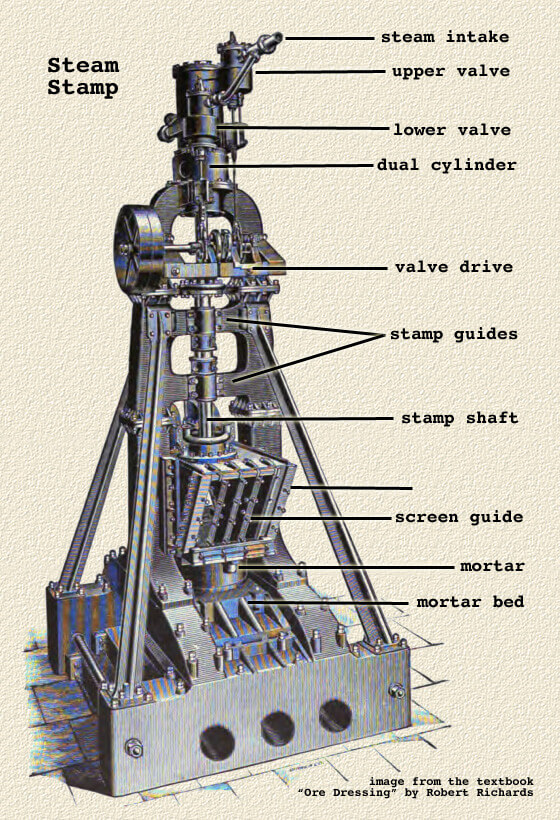
It is probable that no two ores can be treated to the best advantage under exactly the same conditions. A millman experienced in the treatment of the ores of one district may be quite at fault when attempting to amalgamate an ore unlike those to which he has been accustomed. A silver mill, in particular, has been pronounced to be the worst possible school for a gold amalgamator, whose work must be closer in proportion as his amalgam is richer than that obtained from silver ores.
It is obvious that the stamps and screens must be such as are calculated to produce the largest possible output, without rendering the pulp unsuitable for the processes of amalgamation or of concentration, or both, which are to follow. The ideal crushing would be to “crack the nut and leave the kernel entire,” or in other words, to liberate the particles of gold without breaking them. This suggestion, however, is not a helpful one, inasmuch as it has been found in many cases that amalgamable gold remains locked up in particles of ore of all sizes except the very finest slimes. The tendency is now in the direction of finer crushing, if not in the stamp battery, then in re-grinding machines.
The subject of delivery is closely connected with that of crushing and must be considered at the same time. The screens are not usually placed quite close to the level of the dies in the mortar on account of the rapid wear caused by the violent projection of pulp against them when in that position. Their height above the dies is varied according to the ore, the delivery being slower in proportion to this depth of discharge when it exceeds 2 or 3 inches or, according to Peplar, 5 inches. The banking of sand against the screens checks discharge through their lower part if the depth of discharge is small.
The best size for the mesh of the screens must be determined by direct experiment. It has often been contended that, as the crushing must be fine enough to liberate the particles of free gold from their matrix, therefore the size of the screen mesh depends on the state of division of the precious metal in the ore. Even if this be so, however, it does not follow that the apertures in the screen need be small. When coarse screens are used, it is found that, in the course of the crushing, much of the ore has been reduced to a comparatively fine state of division, and usually this portion is found after amalgamation to contain but little gold ; from this, the coarser material, in which the gold is still locked up, may be separated by sizing in “pointed boxes” and reground in an amalgamating mill or pan. If, on the other hand, the slimes are found to be as rich as the coarse sand, it is obvious that no finer crushing is required, as the output would be thereby diminished without any corresponding increase in the yield per ton. If the slimes, after separation of the sulphides, are found to contain more free gold than the coarse sand does, this fact points to the conclusion that a coarser screen might be used without detriment, and experiments in this direction should be made, and the limit of economy thus found by trial.

The evils of overstamping, due to slowness of discharge, have been often dwelt on, and probably are frequently exaggerated. It is true that the excessive production of slime thus caused may sometimes be disadvantageous, but, setting this aside, it has been frequently asserted that particles of gold are reduced in size by overstamping, so that they will float off in suspension in water, whilst, even if not reduced in size, they are hammered and flattened so as to be rendered incapable of amalgamation. “ Float ” gold has been considered above, and as regards hammered gold, it does not seem to be beyond doubt that flattening and hardening alone will prevent gold from being amalgamated. Prof. T. Egleston has described a number of experiments which tend to show that amalgamation is retarded by this treatment of gold, but, on attempting to repeat these experiments at the Royal Mint, the author could not obtain similar results to those of Professor Egleston. Pieces of pure gold when subjected to repeated blows with a clean 7-lb. hammer on a clean anvil occasionally showed a disinclination to amalgamate, but if these pieces were washed with dilute ammonia, so as to remove any grease that might be adhering to them, they were instantly wetted by mercury and were dissolved by it at about the same rate as clean annealed gold. It thus appeared that, in these cases at least, grease formed a more potent preventive of amalgamation than the hardness of the gold. Moreover, gold-leaf which has been subjected to an extended course of hammering is readily amalgamable. It seems probable that the supposed difficulty of amalgamating “hammered” gold has no real existence.
There is still much difference of opinion as to the desirability of attempting to amalgamate the gold inside the battery. It is considered by some millman that the addition of mercury to the mortar is a mistake, and that no copper plates should be put inside. In spite of numerous practical illustrations of the soundness of the contrary view, they hold that no machine can be successful at once as a crusher and an amalgamator. The practice of adding mercury to the mortar when no inside plates are used is certainly not now much favoured, although it is still adhered to in Ballarat and some other districts in Australia. In treating rich ores, however, when the gold is coarse and of high standard, there does not seem to be any valid objection to be raised against catching the gold on inside plates in a concentrated form, instead of letting it all go to the outside. In this case mercury must be added to the mortar, and this is probably not so important a cause of loss of mercury by flouring and sickening as is often assumed. If a decomposing ore is mixed with lime beforehand, and the acidity of the water used is corrected, the conditions do not appear to be favourable to the production of salts injurious to the mercury, and the latter when charged in is probably almost instantly washed through the screens or else dashed against and retained by the plates. The mercury does not remain on the die, subjected to repeated blows, which would no doubt cause much flouring. There is also the point of view that the distribution of mercury through the ore favours amalgamation, and that amalgam is more easily caught on stationary or shaking plates than very finely-divided gold. This last point remains a matter of some doubt. It is often denied on the ground that amalgam is of less density than gold.
Finely-divided gold, however, particularly if it is of low standard, containing much silver, requires more mercury for its amalgamation than coarse gold, and in this case it is difficult to keep the plates in good order, so that it is usually advantageous to save the extra trouble and labour, caused by looking after and cleaning-up inside plates, by putting all the plates outside. This has been the experience in a number of mills, including that of the Montana Company, where the inside plates have been entirely discarded.
Amalgamation outside the battery has also been the subject of much discussion and some careful investigation. Numerous amalgamating machines have been patented, the inventors in every case praising their own contrivances and decrying the copper plate, but the latter has not as yet been superseded, and its principle is applied to almost all its more promising rivals.
To secure successful amalgamation it is necessary that the particles of gold should be brought into absolute contact with the mercury. This contact is obtained in one of three ways:
- The mercury and ore are ground together in pans, arrastras and similar machines, contact being secured by pressing the gold and mercury together.
- The ore is allowed to flow over or even through a bath of liquid mercury, or the endeavour is made to ensure contact by letting the pulp fall from a height upon the mercury.
- The ore is allowed to flow over either stationary or shaking amalgamated copper plates, drops being sometimes introduced between the plates to break up the pulp and to assist in catching the amalgam and “ float ” gold.
The first method is undoubtedly the best for ensuring contact, but the operation is tedious and, in most cases, unnecessary. As to the relative merits of the two latter methods, the great majority of metallurgists are advocates of the superiority of the plates. They point out that in a mercury bath, in spite of the first impression to the contrary felt by everyone on approaching the subject, contact with the ore is very difficult to obtain. If wet pulp is introduced at the bottom of a bath of mercury, it rises to the surface in lumps, surrounded by films of water, and dry pulp is still more effectively protected from contact with the mercury by films of air. If a thin stream of pulp is run over the surface of a bath of mercury of sufficient size, the chances of the particles of gold coming in contact with the quicksilver (by settling through the stream) are greatly improved, but in this case the more convenient copper plate could be substituted for the bath. Moreover, it is well known that rich gold or silver amalgam catches gold more readily than pure mercury, and, whilst the surface of the plates can readily be covered with such pasty amalgam, it would involve a larger and unnecessary sinking of capital to keep any considerable percentage of precious metals in the baths. For these reasons, and for the practical reason that plates are found to work better than baths, the use of the latter has been gradually more and more restricted, until they are now only to be found in narrow wells and riffles for the purpose of catching hard amalgam and floured mercury, and as purely supplementary aids to the plates, whilst they are sometimes dispensed with altogether. When a proper disposition of the plates is made, it is rare to find amalgamable gold escaping into the tailings. Even if the latter contain several pennyweights of gold to the ton, this does not show that the amalgamation effected on the plates is unsatisfactory, and, where tailings have to be reground or roasted, or treated in any way that may alter the condition of the gold, before a further extraction by mercury is obtained, no proof is afforded that the plates are not doing their work.
