The Rule and Function of Oxygen in Cyanidation.—Oxygen appears to be an indispensable factor, either directly or indirectly, in the dissolution of gold and silver by cyanide solutions, except in the case of the haloid compounds of silver. Whether the action of oxygen in the dissolution of gold be a direct one, as illustrated by the Eisner equation, or an indirect one, in the sense of acting merely the part of a depolarizing agent, as maintained by Julian and Smart, will not affect the general statement that it is a necessary adjunct for the dissolution of the precious metal.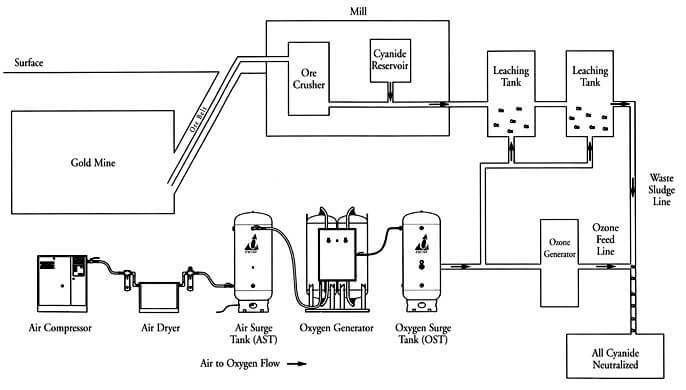
The most generally useful agent for Cyanidation is atmospheric oxygen, and in many instances sufficient oxygen is absorbed by the solutions in their circulation through the plant to accomplish all that is necessary. Ores that contain reducing constituents may need more oxygen than is obtained in this way, and the additional amount is most easily supplied in the case of percolation by draining dry between washes, and by the use of air lifts or centrifugal pumps, in agitation.
The addition of chemical oxidizing agents, such as potassium ferricyanide, permanganate, sodium peroxide, and ozone, has not been attended with much success, both on account of their cost, and also because of their tendency to oxidize the cyanide to cyanate (KCNO), which is useless for the purposes of the process. They have sometimes been found useful, however, for oxidizing a highly reducing pulp before adding cyanide, as in the case of slime which has stood in dams for long periods, undergoing partial oxidation with formation of ferrous compounds.
The only reagent of the oxidizing class that has attained to any importance commercially is bromocyanogen, BrCN, and that only in the raw treatment of telluride ami mispickel gold ores.
Julian and Smart consider that the activity of this compound is not due to the liberation of cyanogen, though that probably occurs, but to a liberation of oxygen, in the sense of the equation.
2BrCN + KCN + 4KOH = 2KBr 4- 2KCN + KCNO + 2HsO + 0
Reducing Agents.—Oxygen being necessary in almost every instance for the dissolution of gold and silver by cyanide, it follows that any substance that has the property of denuding the solution of its dissolved oxygen will retard or completely stop the action of the cyanide. It is a common practice to test working solutions for their “reducing power” by acidifying and titrating with standard permanganate. Such a test, however, is misleading in its bearing on the dissolving efficiency of a solution, because it includes the reducing effect of such substances as ferrocyanides and sulpliocyanatos which have not the power of absorbing oxygen from solutions and therefore do not retard the dissolving effect of the cyanide on account of their reducing character, in fact, they are. not reducers when considered from that standpoint. If, then, an estimate of the detrimental reducers present be desired the ferrocyanides and sulpho-cyanates should be determined separately and their equivalent in terms of standard permanganate deducted from the “total reducing power,” as already found.
1 cc N/10 permanganate = 0.001619 grm. KCNS
1 cc N/10 permanganate = 0.036831 grm. K4Fe (CN)6
P 3-4 MANUAL OF CYANIDATION BY E. M. HAMILTON, B. A. (oxon) 1st Edition 1920
