Table of Contents
Chemistry plays a more important role in milling operations than is generally recognized, particularly in fluid-solid separation processes. Chemical aspects are also very important insofar as plant water quality is concerned, and in the manner in which this water quality effects plant design and operation.
One aspect of the use of mining chemicals which has received scant recognition is the realization that in many cases, the equipment in which the chemical is to be used must be designed specifically for use with the chemical, or the equipment, or its method of operation must be modified in order to obtain optimum results. This is especially true of equipment such as filters, thickeners, clarifiers, reagent mixers, reagent feeders and distribution systems, pumps, etc.
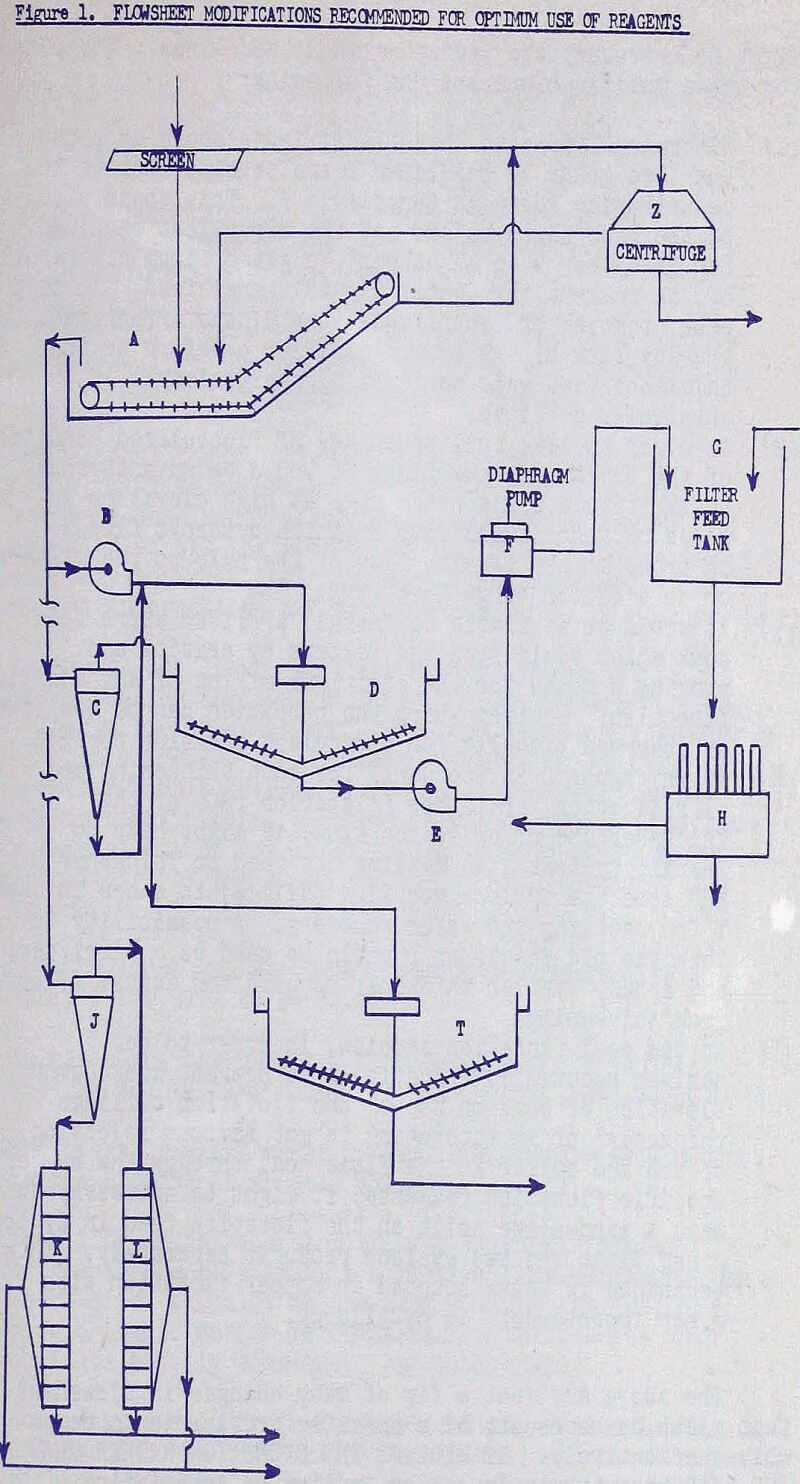
Based on the flowsheet, the reasons for these modifications are the following:
- flocculent added at Drag Classifier A could decrease the load going to Thickener D and perhaps improve the centrifuging rates in Centrifuge Z.
- in order to take full advantage of flocculated condition of the thickener underflow.
- it would be desirable to install a filter surge feed tank which would feed the filters by gravity and provide a means for the efficient mixing.
- if Thickener D is too small for both thickening and clarification, or if the filtration rate of the thickener underflow is too slow.
- in the coal flotation section, in order to get maximum recoveries (especially to prevent subsequent flotation of coal on top of the flotation tailings thickener) or in attempting to get maximum rejection of ash and sulfur in the float coal through the use of specific flotation reagents, it might be necessary to make a sand-slime split on the flotation feed in Cyclone J and float the two cyclone products separately.
The above are just a few of many changes in flowsheet that might be necessary if a specific problem is to be resolved effectively. IT WILL BE THE EXCEPTION RATHER THAN THE RULE that chemicals can be applied to an existing flowsheet (that originally was not designed for conjoint use) and function properly; yet this is the common expectation of most operating people. It is most important, therefore, that operators have a good understanding of why equipment, chemicals, and operating techniques must be carefully co-ordinated to obtain optimum results at the lowest costs.
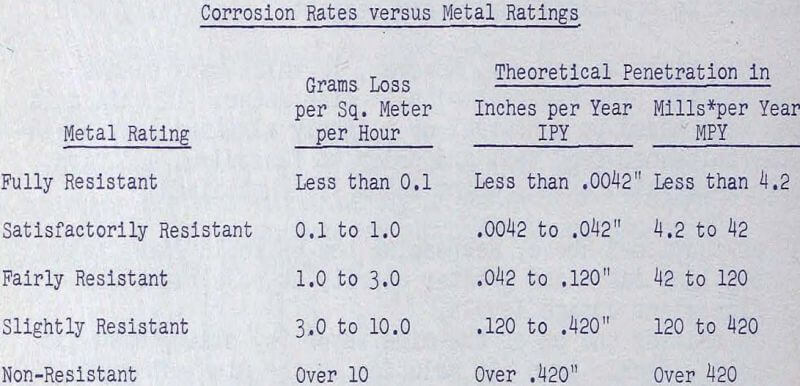
If abrasion will occur and remove the protective film, corrosion may be rapid if dissolved oxygen is present in plentiful amounts. Oxygen is actually the factor which will control the corrosion rate of steel in the weakly acid pH range. This dissolved oxygen can be controlled by the addition of reducing agents, one of the best of which is ferrous sulfate, a principal constituent in acid mine water which has a reasonably high solubility over the pH range 4 to 9. It might be well to recall that acid mine water acquired its reputation as a highly corrosive medium in underground operations primarily because of its ferric (in contrast to ferrous) iron content in the pH range 2 to 3. Above pH 3, the ferric iron is essentially completely precipitated so that its concentration at pH 4 is practically zero.
Inter-relationships of Chemistry, Equipment Design, and Costs
In order to stimulate some thought on this subject, we shall examine one complex relationship between flocculents, thickener design, and capital and operating costs. First, if the settling rates of a particular thickener feed pulp are determined in laboratory tests at various flocculent dosage levels, and the data are plotted as settling rates versus flocculent consumption. Hence an important axiom is that settling rates obtainable with flocculents are not infinite, and actually might be confined to a fairly narrow range, especially where fine pulps of minerals such as clays, iron oxides, and heavy sulfides are concerned. Thus, contrary to what is often expected, flocculents alone cannot be relied upon to remedy extreme variations in thickener operations that may require high settling rates to maintain tight overflow clarification specifications. Chemicals alone will not compensate for skimpy or poor design, or poor operating procedures. To try to use them in this manner is wasteful and costly; Is quite often unsuccessful, and can sometimes lead to serious damage of a thickener.
Many flocs formed from very fine particles occlude water so that their apparent specific gravity might be as low as 1.1. Recognition of this condition makes it apparent why “loading” of a fine thickener feed with larger particles is helpful in obtaining good clarification. The trapping of a large, fast settling particle in a floc of fine particles will give the higher settling rates required.
Capital Costs versus Operating Costs
Based on next figure, if we design thickeners
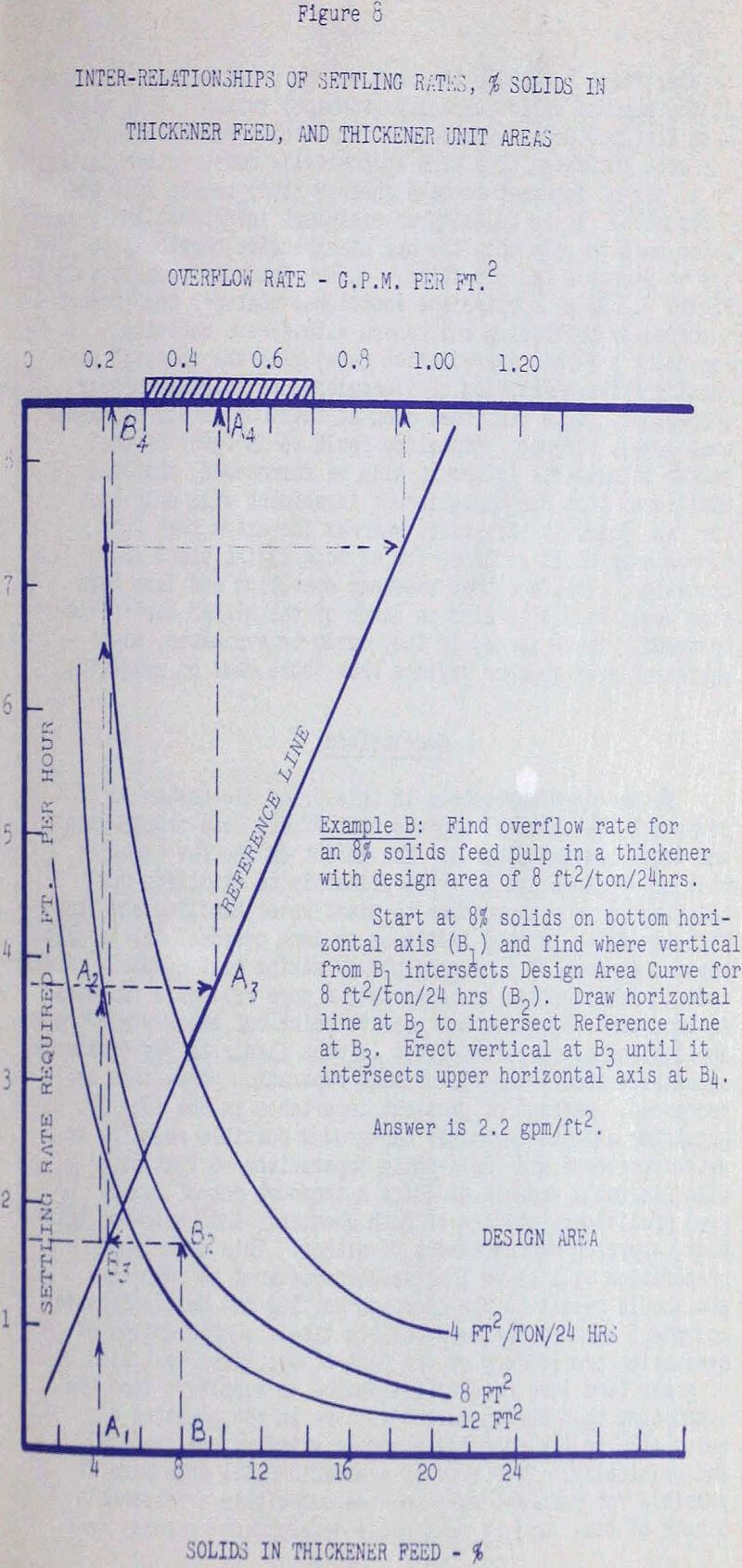
or clarifiers to handle the most dilute pulps we can anticipate at the maximum solids capacity (tonnage) rating. This might mean that our design would call for a thickener with double the area indicated by a more economically conservative design. It is now of interest to make another study to see just how “economical” It is to skimp on equipment investment and then later have to rely upon the use of expensive chemicals to try to remedy a bad situation.

