Table of Contents
This report stems from a study by the Bureau of Mines of the possibility of reducing coal filter-cake moisture by applying radiant heat directly to the filter cake while under vacuum during the drying phase of the filter cycle. One disk of a 4-foot-diameter vacuum filter was equipped with gas-fired radiant-heat generators and the effect of radiant heating on the moisture content of a variety of filter cakes was studied.
The results of this investigation show that this process can best be described as a filter aid capable of increasing the effectiveness of conventional filtration approximately 6 percentage points of moisture. The operating cost of removing water by this method is somewhat lower than the average operating cost of thermal drying and is considerably cheaper when the total cost is considered.
Removal of water from coal that has been wet-cleaned is an important part of the overall coal preparation process. Water is a contaminant, and it is foolish to wash coal to reduce its ash content and add inert water in the process. In some instances the removal of water is actually more important than the removal of ash-forming minerals; its weight adds to the cost of transportation, and its evaporation during burning or carbonizing absorbs useful heat. Fine coal that contains more than about 10 percent moisture is notoriously difficult to handle. A problem thus exists for which there are sound practical and economic reasons to seek a solution.
Products resulting from a wet-washing process contain as much as three times their weight of water. The removal of enough water from the coarse sizes (plus 3/8-inch) to meet consumer requirements is relatively easy and usually may be accomplished by dewatering screens and by natural drainage. The dewatering of intermediate-size coal is effectively accomplished by basket and solid-bowl centrifuges. The removal of water from the sizes finer than 48-mesh is, however, a major problem. Hydraulic cyclones and static thickeners are capable of thickening dilute suspensions of minus 48-mesh coal and water to about 50 percent solids; vacuum filters can reduce the moisture to 20 to 30 percent. Moisture content of coals in this fine-size range can be further reduced only by thermal drying.
A laboratory investigation of the possibility of reducing filter-cake moisture by radiant-heating was completed by the Bureau of Mines and the results published. The Bureau knew that commercial scale results would differ from the laboratory results for several reasons: (1) laboratory heat input was 50,000 British thermal units per hour per square foot of filtering area, but in practice the heat input would be reduced to about 22,000 British thermal units per hour per square foot of filtering area, (2) no attempt was made during the laboratory study to contain and use the sensible heat from the radiant heaters, and (3) the cake thicknesses used in the laboratory study were not related to the length of time the filter cake was heated, whereas in actual practice, the cake thickness formed is related to the rotation speed of the filter and hence to the time available for heating, and vice versa. Because of these inherent difficulties in scaling-up laboratory data, a pilot-sized disk vacuum filter was equipped with radiant heaters to simulate actual plant operation.
Theoretical Considerations
The separation of liquids from solids by passing the liquid through a porous medium involves several interrelated variables. Coal slurries exhibit individual filtering characteristics. Although much progress has been made in determining the effect of operating variables on filter capacity and final filter-cake moisture content, coal filtration is still more an art than a science. However, filtration theory is valuable in interpreting laboratory test data and in seeking optimum filtration performance.
An adaptation of Poiseuille’s equation, which is generally considered to be the basic cake-filtration equation for streamline flow of liquid through a filter cake, may be given as
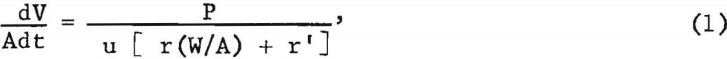
which expresses the differential or instantaneous rate of filtration per unit area
where
V = volume of filtrate collected,
A = area of filtering surface,
t = time,
P = total pressure drop through filter medium and cake,
u = filtrate viscosity,
r = average specific cake resistance,
W = weight of dry cake,
and r’ = resistance of unit area of filter cloth, and pressure drop in lines.
When the cake is composed of hard granular particles that make it rigid and incompressible and filter medium resistance is neglected, equation (1) becomes
dV/dt = AP/ur(W/A)……………………………………..(2)
An examination of the factors in equation (2) yields information on the effects of filtration variables. The rate of water removed from a slurry is increased in proportion to the filtering area and pressure drop across the filter medium and cake and is inversely proportional to the filtrate viscosity, average specific cake resistance, and weight of dry cake per unit filtering area.
For a given feed to a filter, a cake forms on the filter medium, and a pressure drop across the cake and filter medium is established commensurate with the filtering characteristics of the feed. A dewatered cake and a low percent solids filtrate are obtained as final products. In searching for means to reduce further the moisture content of the cake, researchers have investigated the individual and combined effects of the factors involved. One of the possibilities for lowering the cake moisture is decreasing the viscosity of the filtrate. The viscosity of water is affected markedly by temperature, going from 1.55 centipoise at 40° F. to 0.56 centipoise at 120° F. Equation (2) shows that the rate of water removal is an inverse function of the filtrate viscosity so that increasing the filtrate temperature should have significant effect on the final filter-cake moisture.
Silverblatt and Dahlstrom have studied the effect of increased filtrate temperatures and have shown the possibility of reducing the moisture content of a filter cake from 23.4 to 18.1 percent by heating the slurry from 40° to 120° F. a reduction of 5.3 percentage points. Applying these data to an operating coal preparation plant where the circulating water would be heated from 40° to 80° F. by the direct introduction of 100 pounds per square inch absolute steam for three alternate cases, they estimated that the heat requirements would range from 5.8 million to 17 million British thermal units per ton of water removal. The heat requirement for thermal drying, by comparison, is about 4.4 million British thermal units per ton of water evaporated.
During filtration, the rate of water removal decreases rapidly after the cake emerges from the slurry when the water remaining in the cake is mainly a thin film covering the particles and held at the points of particle contact. As the filtration cycle continues, moisture reduction is small. At this point, the application of heat would be most effective. The object is to heat only that part of the filtrate that is not removed by normal filtration rather than to heat all the water before feeding it to the filter. This procedure should result in reducing the overall heat requirements.
The time available for heating the water entrained in the filter cake is about one-third of the filter cycle (about 1 minute); therefore the heat source must be operated at high temperature to transfer the necessary heat during the limited exposure time. The gas-fired radiant heater appeared to be best suited for this application.
Test Apparatus
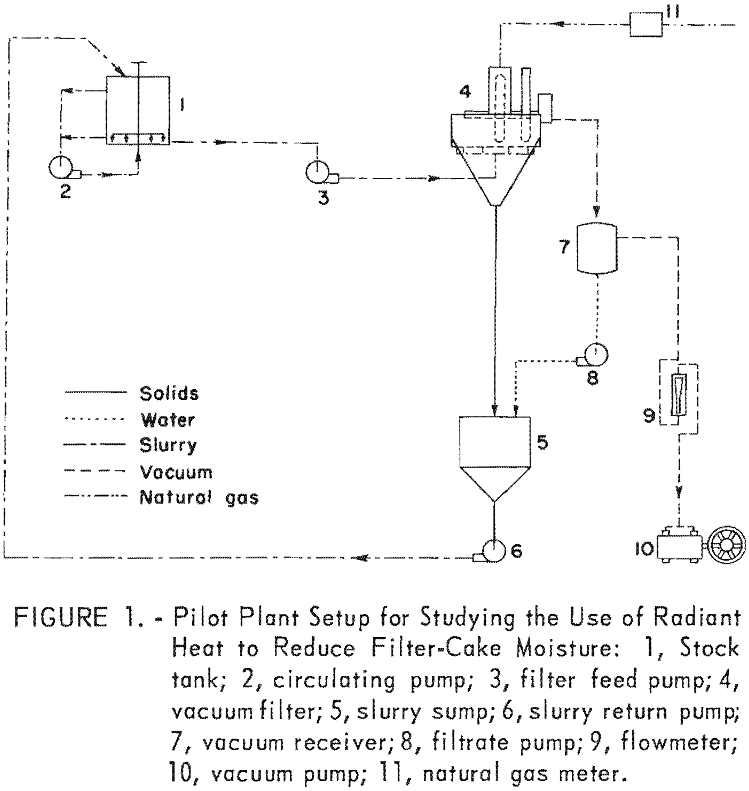
The flowsheet of the radiant-heated filter setup is shown in figure 1. Essentially, it consists of a 1,000- gallon stock tank, a vacuum filter that is fitted with radiant heaters, a collecting sump, and a return pump.
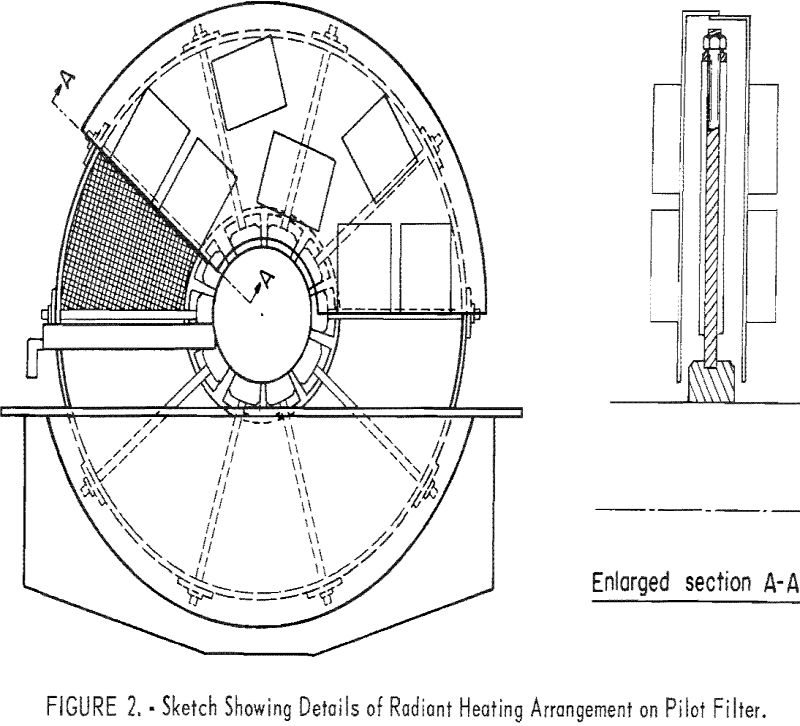
The filter is a conventional four leaf, 4-foot-diameter disk vacuum filter; each disk consists of eight pie-shaped sectors covered with 42- by 60- mesh stainless steel screen-cloth filter medium. For this study, 2 of the disks were removed and 1 of the remaining disks was equipped with 14 (7 on each face) gas-fired radiant-heat generators. Overall these generators were 11 inches long, 6-3/8 inches wide and 3 inches deep and were rated at 12,000 British thermal units per hour at 5-ounce gas pressure. The generators were mounted in as dense a pattern as possible on a ¼-inch-thick steel plate that was shaped like the sector of a circle and folded over at the top to form a telescoping adjustable envelope. This whole assembly was clamped to the filter tank. Figure 2 shows the details of the arrangement of the heaters. Figure 3 is an overall view of the radiant-heated filter; figure 4 shows the discharge side of the filter where the cake emerges from between the heaters.
Total heat input to the heated disk was 168,000 British thermal units per hour or 22,400 British thermal units per hour per square foot of heated filter area. The required 168 cubic feet per hour of natural gas for the radiant heaters was reduced from mainline pressure to 5-ounce pressure by means of gas regulators. It then passed through a gasmeter and was divided into two manifold pipes for distribution to the individual heaters. A heated-probe safety- control system was included in the gas supply to each of the two banks of heaters. Basically, the system is one in which the gas supply to the heaters is controlled by a normally closed electric solenoid valve. This solenoid valve can be opened only if a power supply is available and the pilot light is

burning and heating the probe. Failure of either the power or gas supply causes the solenoid valve to automatically close, thus preventing an accumulation and possible reignition of gas in the hood containing the heaters.
Filter rotation time was quickly and accurately set by means of a stroboscopic tachometer on the output shaft of the variable-speed filter drive unit. The total volume of air passing through the filter was measured by a flowmeter, and the pressure drop across the filter cake was measured by a pressure gage on the filter manifold. Feed and filtrate solids concentration, cake thickness, and moisture content of the filter cake were determined from samples collected during a test.
To conserve coal and permit direct measurement of the effect of operating variables, both the filter cake and the filtrate were collected in the slurry sump and returned to the stock tank for reuse.

Test Procedure
A feed slurry of the desired coal solids concentration was prepared by mixing the quantity and type of coal with the required quantity of water in the stock tank. The desired filter rotation time was set, and the slurry was pumped to the filter. Excess feed slurry overflowed two weirs in the filter tank and was returned to the stock tank. After a filter cake formed, the radiant heaters were lit and the filter operated until a uniform cake formed on both disks and the heaters reached operating temperature.
Once the system reached equilibrium, duplicate samples of both the heated and unheated filter cake were taken. In most instances one full sector was taken as a sample to permit calculating dry-coal solids throughput and water removal rate. A timed sample of the filtrate was also taken. Data recorded for a given test condition included the volume of air and the pressure drop across the filter cake, filter-cake thickness, filter rotation speed, volume and pressure of the gas supplied to the heaters, and the qualitative condition of the filter cake. The filter-cake samples were weighed immediately and then dried to constant weight; the feed and filtrate samples were filtered, and the solids were dried to constant weight.
No flocculants were used to increase filter-cake rates. The synthetic slurries were a simple mixture of coal and water; the samples of slurries collected in the field were tested in their original condition.
Synthetic Slurries
A sample of Pittsburgh-bed coal was pulverized through 14-mesh and mixed with water in the stock tank. This slurry, containing 33 percent solids, was then pumped to the filter. The time of rotation and the gas pressure to the heaters was varied to study the effect of drying time and heat input on the filter-cake moisture content. Some difficulty in forming a uniform cake on the four filter faces was found because of the coarseness of the coal solids. A mechanical mixer was added to the filter tank and did prevent the settling of the coarse solids, but it caused an unequal distribution of solids on the filter faces, which was reflected in the formation of irregular and nonuniform cake thicknesses. The air rate through the filter cake was 1.25 cubic feet per minute per square foot of filter area at 24 inches of mercury vacuum. Both faces of the heated and unheated filter cake were sampled by taking full sector samples. A summary of the data from test 1 is given in table 1.
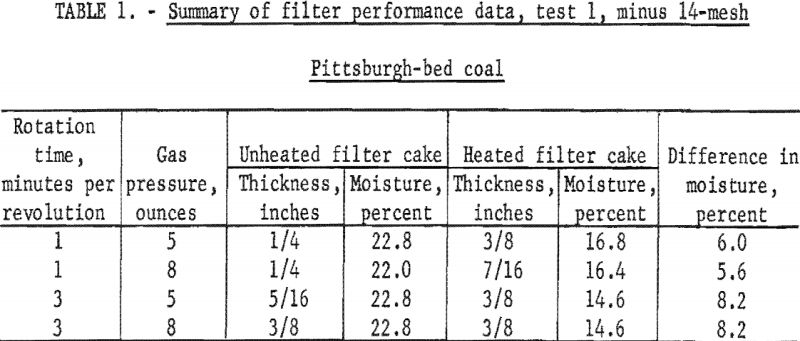
The data in table 1 show average moisture reductions, owing to radiant heating of 5.8 and 8.2 percentage points at 1 and 3 minutes per revolution, respectively. Increasing the heat input by increasing the gas pressure from 5 to 8 ounces had little or no effect on the moisture reductions achieved.
Another sample of Pittsburgh-bed coal was crushed through 20-mesh, and another series of tests determined the effect of heating time and heat input on the filter-cake moisture content. Samples of the heated and unheated filter cake were collected and a summary of the data from test 2 is given in table 2. Air rate through the filter cake was 3 cubic feet per minute per square foot of filter area at 12 inches of mercury vacuum. Filtrate solids amounted to 3 percent by weight.
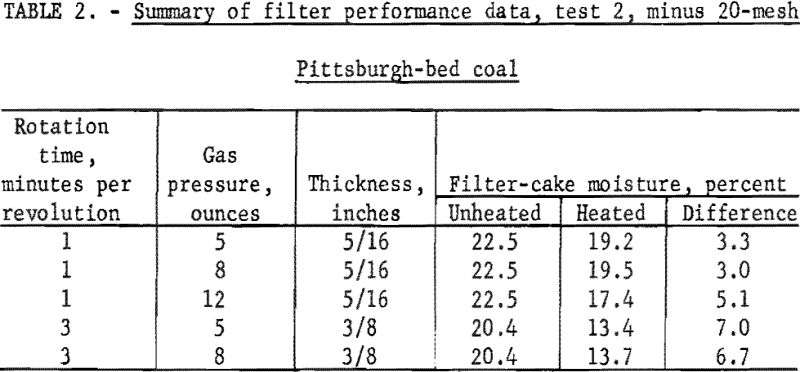
The data in table 2 show that thicker filter cakes containing less moisture were formed at 3 minutes per revolution than at 1 minute per revolution and that a threefold increase in the heating time resulted in about a twofold reduction in moisture as a result of heating. As in test 1, increasing the gas pressure from 5 to 8 ounces had no significant effect on the results; increasing the gas pressure to 12 ounces resulted in an additional 2 percentage points of moisture reduction but at a prohibitive rate of gas consumption.
Another sample of Pittsburgh-bed coal was crushed to minus 48-mesh and a slurry containing 25 percent solids was pumped to the filter (test 3). Air rate through the filter cake was 4 cubic feet per minute per square foot of filtering area. Duplicate samples were collected from both the heated and unheated disks at three rotation times. These data are given in table 3.
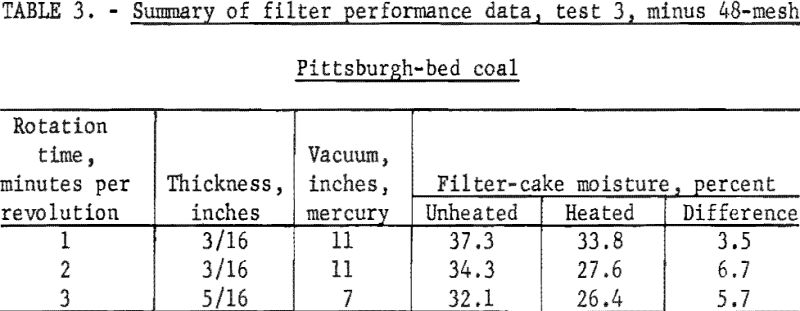
Tests 1, 2, and 3 followed generally the expected trend of dryer, thicker filter cakes and greater reduction of moisture because of applying radiant heat with slower rotation times. The increase in cake thickness (and hence moisture content) at slower rotation times was more than compensated for by the longer heating time. The overall effect of the fineness of coal used was increased moisture content of both the unheated and the heated filter cake and a reduced effectiveness in applying radiant heat.
In order to study the effect of radiant-heating filter cakes resulting from the filtration of deslimed and ultrafine (minus 200-mesh) coal slurries compared with resultant coals, a lot (about 1,000 pounds) of Pittsburgh-bed coal was crushed through ¼-inch and then ball-milled to a nominal minus 28-mesh, containing a high percentage of minus 200-mesh material. This coal was pulped with water to 33 percent solids in the stock tank and pumped to the radiant-heated filter. Duplicate samples of the heated and unheated filter cake were collected at rotation times of 1 and 3 minutes per revolution.
After this test was completed, the original feed slurry amounting to about 400 gallons was pumped to a bank of four 3-inch-diameter cyclones for size classification. The 250 gallons of cyclone overflow, containing 23 percent solids was returned to the stock tank; the 150 gallons of cyclone underflow, containing 48 percent solids, was collected and stored for subsequent testing. The wet screen analysis of the cyclone feed, overflow, and underflow product is given in table 4.
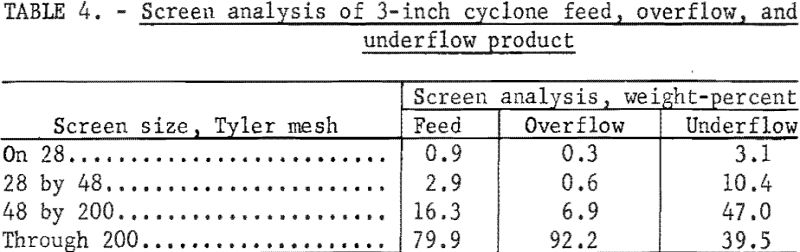
Both the cyclone overflow and underflow product were tested under the same conditions as the original slurry. The cyclone overflow was tested at its original 23 percent solids concentration, but the cyclone underflow was diluted to 40 percent solids before being fed to the filter. Duplicate samples of the unheated and heated filter cake were collected at the rotation time of 1 and 3 minutes per revolution . A summary of the data from test 4 is given in table 5.
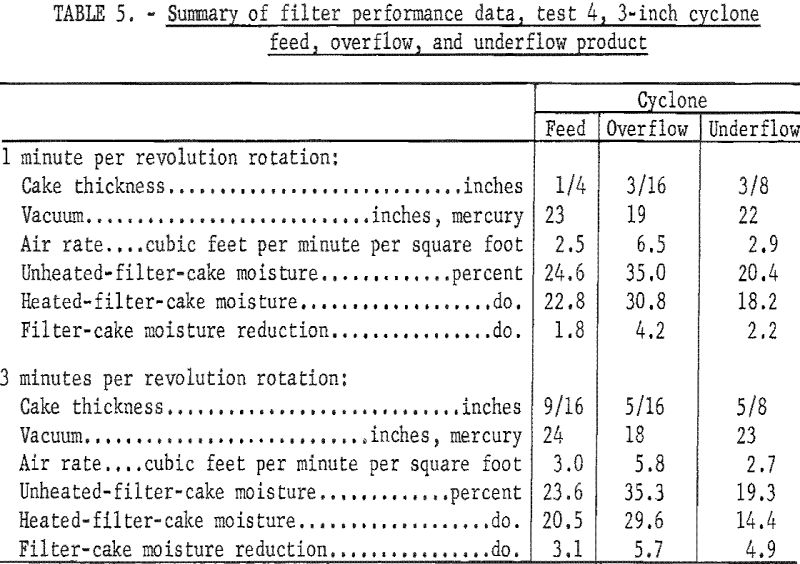
Filtering the original minus 28-mesh slurry at a rotation time of 3 minutes per revolution produced a 9/16-inch-thick cake, containing 23.6 percent moisture, and a filtrate, containing 2.9 percent solids. Applying radiant heat to this filter cake for 1 minute reduced the filter-cake moisture to 20.5 percent, a reduction of 3.1 percentage points. The relatively high moisture content of both the heated and unheated filter cake may be attributed to the fact that the filter feed contained 79.9 percent of minus 200-mesh material.
Excellent results were achieved at a rotation time of 3 minutes per revolution -when filtering the deslimed cyclone underflow. A 5/8-inch-thick filter cake that formed easily at a vacuum of 23 inches of mercury contained 19.3 percent moisture, and the application of radiant heat further reduced this moisture content to 14.4 percent, a reduction of 4.9 percentage points. The filter feed contained 39.5 percent of minus 200-mesh material, and the filtrate contained 1.1 percent solids.
Filtration of the cyclone overflow, containing 92.2 percent of minus 200-mesh material, at a rotation time of 3 minutes per revolution was characterized by thin (3/16-inch), wet filter cakes. Because of the formation of thin hairline cracks, it was not possible to maintain a vacuum drop across the filter cake greater than 19 inches of mercury. The filtrate, which contains about 2 percent solids when filtering normal coal on a 42- by 60-mesh screen filter medium, contained 13.8 percent solids. As this cyclone overflow contained only 1 percent of material coarser than 48-mesh, there was not enough coarse material to bridge the openings in the filter medium. The use of finer filter medium would have reduced the solids concentration of the filtrate and increased the moisture content of the filter cake. Despite the high concentration of fines, the application of radiant heat effected a 5.7 percentage point moisture reduction in the filter cake.
Coal slurries are known to exhibit individual filtering characteristics and it was felt desirable to verify the results obtained on synthetic slurries by tests on actual preparation plant slurries. Arrangements were made to collect two types of coal slurries commonly dewatered by filtration, a thickener underflow product, and a froth flotation concentrate.
Thickener Underflow Product
A sample of thickener underflow product was collected from a preparation plant in northern West Virginia. This plant uses a thickener for water clarification; the overflow is sufficiently low in solids to be returned to the plant circulating water. The thickener underflow contained 54 percent solids (the screen analysis is given in table 6). This product, after filtration, contained 30 to 35 percent moisture and was so tacky that it could not be handled by conventional means nor could it be thermally dried or combined with the thermally dried fine coal. Although this filter cake, amounting to about 25 tons per hour, is of suitable quality to be considered a salable product if its handling properties could be improved, it is presently being wasted.
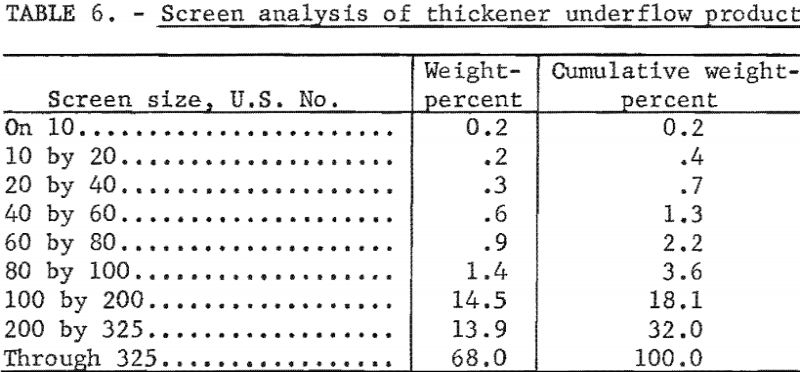
The 440-gallon sample was put into the 1,000-gallon stock tank and pumped to the filter, and duplicate samples of both the heated and unheated filter cake were collected (test 5). These data are given in table 7. The filtrate contained 12 percent solids and the air rate during the test was 6 cubic feet per minute per square foot of filter area.
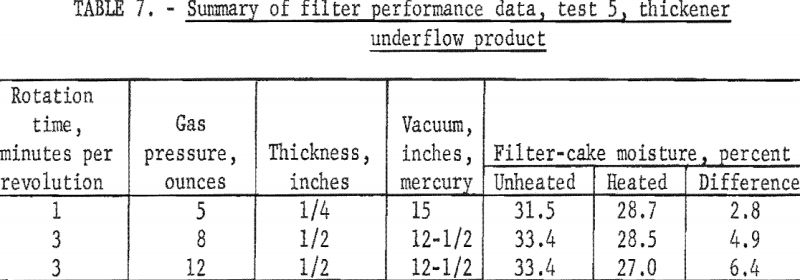
In view of the high concentration of extremely fine coal, the moisture reductions shown in table 7 are indeed encouraging. The improvement in the handling properties of the filter cake as a result of heating was equally encouraging. Without heat, the cake was tacky, difficult for the scrapers to remove, hung up in the filter-cake chute under the filter, and could not be mixed with dry coal. The heated cake, by comparison, could be handled, was freely discharged from the filter medium, and appeared as though it could be blended in a ribbon mixer and either thermally dried or blended with thermally dried fine coal.
Froth Flotation Concentrate
A 600-gallon sample of froth flotation concentrate was collected from a preparation plant in western Pennsylvania and put into the stock tank ahead of the filter. The concentrate contained 10 percent solids; the screen analysis is given in table 8. A collector and frother were used as flotation reagents at the rates of 1.24 and 0.11 pounds per ton, respectively. The effect of rotation time and heat input on the moisture content of the filter cake were tested, and a summary of these data is given in table 9. The filtrate contained 0.5 percent solids; the air rate through the filter cake was 4 cubic feet per minute per square foot.
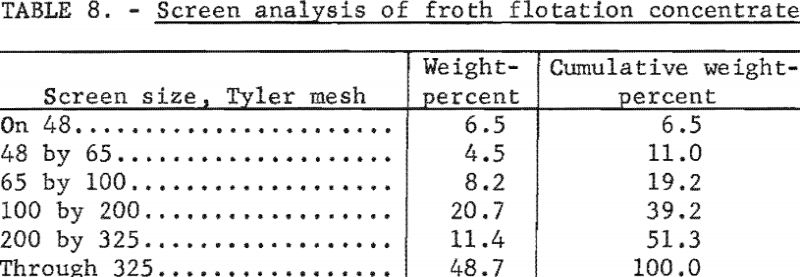
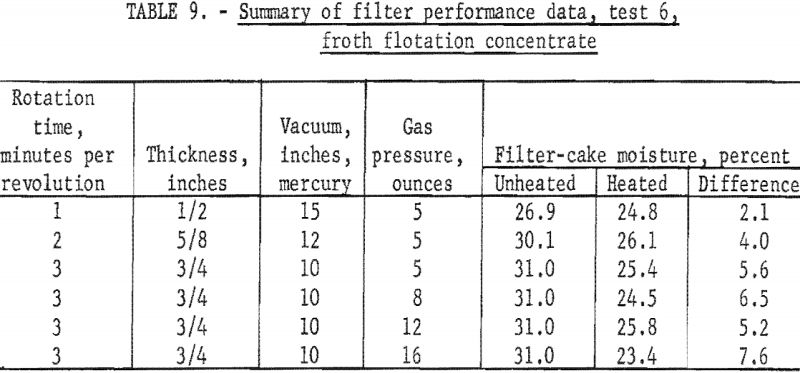
The data in table 9 show that applying radiant heat resulted in a moisture reduction of about 6 percentage points at a rotation time of 3 minutes per revolution regardless of the heat input. The moisture content of the unheated filter cake of 31.0 percent was considered high, and the remaining unheated disk was removed in an effort to increase the pressure drop across the filter cake by halving the effective filter area. The test conditions and data from samples collected during this test (test 7) are given in table 10.
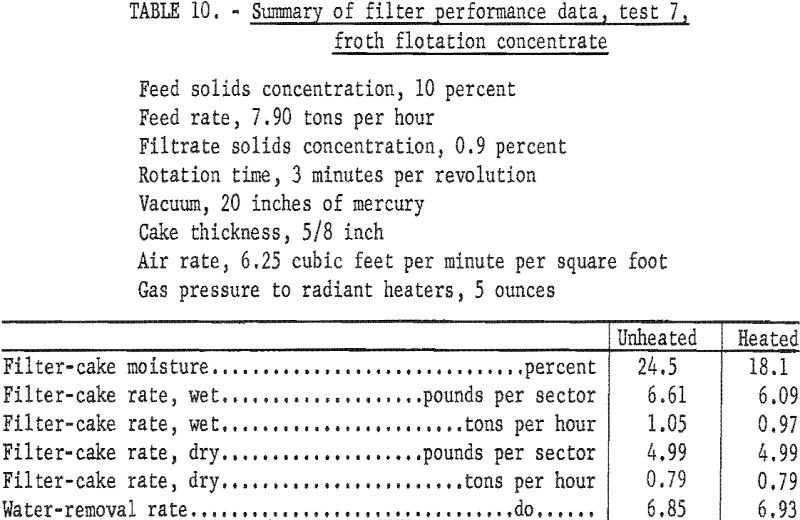
Thus, the overall effect of halving the effective filtering area was an increase in the pressure drop from 10 to 20 inches of mercury, and a lowering of the unheated-filter-cake moisture about 7 percentage points. The moisture reduction due to radiant-heating remained substantially unchanged at about 6 percentage points.
Economic Considerations
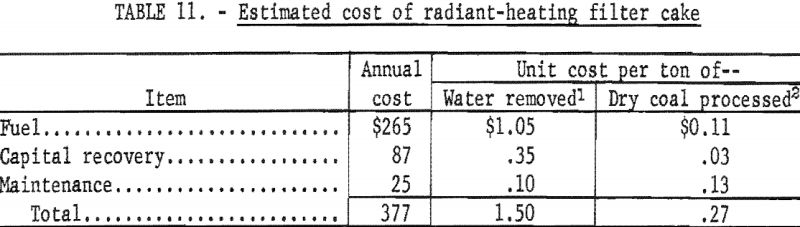
Using the preceding data, it was possible to calculate the cost per ton of water removed and the cost per ton of dry coal processed as a direct result of applying radiant heat. The quantity of water removed (table 10) was the difference between the water-removal rate of the unheated and heated filter cakes or 0.08 ton of water per hour; 0.79 ton per hour of dry coal was processed.
The total cost of radiant heating includes the direct cost of fuel, recovery of capital investment, and maintenance. The installed cost of the 14 radiant heaters was estimated at $1,000 to be amortized by uniform annual payments at 6 percent interest over 20 years of operation. It was further assumed that the filter would operate 3,150 hours annually (14 hours per day and 225 days per year) and that additional operating personnel would not be required. Maintenance cost was estimated at $0.10 per ton of water removed. The 14 radiant heaters burn gas at a total rate of 168 cubic feet per hour for an operating cost of $0.084 per hour when gas that costs $0.50 per 1,000 cubic feet is used. On this basis, the estimated annual and unit costs are as shown in table 11.
The moisture reductions achieved by radiant-heating are the result of the combined effect of lowered fluid viscosity and evaporation. Attempts to determine the relative proportions of moisture removal associated with viscosity reduction and evaporation have failed. However, some insight into the mechanics of the process may be gained from the following analysis.
About 2.2 million British thermal units are theoretically required to evaporate a ton of water; during test 7, the heat required to evaporate 0.08 tons of water was 168,000 British thermal units, which is 2.1 million British thermal units per ton of water removed as a direct result of applying radiant heat. If the moisture reduction is considered to be wholly the result of water evaporation, the process approaches 100 percent thermal efficiency. As process heat is lost by heating the solids and water discharged in the warm filter cake, by sensible heat losses, and by radiation, the process cannot achieve such a high thermal efficiency.
If the filter cake is assumed to enter the drying zone at 50 percent moisture, the quantity of water and coal entering the drying phase during test 7 were both 0.79 ton per hour. The quantity of water removed during the drying phase was 0.53 and 0.62 ton per hour for the unheated and heated cakes, respectively. To remove 0.09 ton per hour of water on the basis of lowered filtrate viscosity would require a reduction of filtrate viscosity from 1.00 to 0.85 centipoise along with an average temperature rise from 68° to 80° F. The theoretical heat required to increase the temperature of the 0.79 ton per hour of water 12° F. is 18,960 British thermal units per hour. On the basis of a heat input of 168,000 British thermal units per hour, an efficiency of about 11 percent is indicated. This is obviously too low.
Therefore, the mechanics of this process appears to be a complex combination of both water evaporation and lowered fluid viscosity. The radiant heat apparently evaporated the moisture on or near the surface of the filter cake and leaves the process via the escaping sensible heat. The sensible heat being pulled through the cake is apparently transferred to the water entrained in the cake increasing its temperature and decreasing its viscosity. The temperature of the air was 125° F.; after it passed through the heated filter cake the average temperature of the heated filter cake was about 110° F. The fact that the surface moisture is removed by radiant heat and that the amount of sensible heat pulled through the cake is determined by the air volume pulled through the cake may explain the failure of increased heat input to increase moisture reductions.
Conclusions
- The application of radiant heat to filter cakes that are under vacuum during the drying phase of the filter cycle can increase the effectiveness of vacuum filtration by about 6 percentage points. This process lacks flexibility inasmuch as heat can be applied for only one third (about 1 minute) of the filter cycle.
- The mechanics of radiant-heating filter cake under vacuum is a complex combination of both evaporation and viscosity reduction. Categorically, this radiant-heating process is more a filter aid than a thermal dryer.
- Handling properties of the filter cake are markedly improved by radiant heating even when the moisture reductions were insignificant.
- Equipped with adequate control devices, this process should not require any additional operating personnel nor does it introduce any special hazards.
- The total cost of applying radiant heat is about $1.50 per ton of water removed and $0.27 per ton of dry coal processed; the total estimated cost for removing water, which was interpolated from laboratory data using a 5/8-inch thick cake that was heated for 1 minute, was $1.36 per ton. An operating cost of $1.15 per ton of water removed compares favorably with the average cost of $1.80 cited by Judy and Washburn and the economy of this process becomes even more attractive when capital costs arc included because the only expenditure required is the cost of the radiant-heat generators.
