Table of Contents
Several years ago, the Bureau of Mines initiated a program to develop effective methods for reclaiming and recycling metal and mineral values contained in urban solid waste.
The Bureau’s initial work at its College Park (Md.) Metallurgy Research Center dealt with establishing reliable methods for sampling and characterizing the residues left from the incineration of urban waste. After the general composition of residues had been established, research was aimed at developing mechanical methods for recovering and separating the residues into fractions that, if necessary, could be further treated to produce products suitable for recycling. The most promising procedures that evolved from this t research were incorporated into a continuous beneficiation pilot plant which came on stream in November 1969. A commercial-size plant based on the Bureau’s process will be constructed at Lowell, Mass, in the near future.
The Bureau’s research is now focused on developing mechanical methods for separating unburned urban refuse into recyclable fractions. Other researchers have found that refuse is composed of approximately 51.6 percent paper, 1.4 percent plastic, 9.9 percent minerals, 10.2 percent metals, 7.6 percent fibers and nonconductors, and 19.3 percent organic matter. To separate the refuse into its various components, the Bureau has been investigating various combinations of multistage shredding, air classification, magnetic separation, screening, electrodynamic separation, and optical sorting operations.
Most of the major pieces of equipment required for this pilot plant have been purchased and are currently being installed. Efforts are now being devoted to prove, further develop, and refine the proposed separation procedures. One important phase of the research on unburned urban refuse is aimed at producing a paper-free plastic concentrate as well as a plastic-free paper product. The method being investigated utilizes a high-tension electrodynamic separator. This report presents the progress the Bureau has made in developing this technique to separate plastics from paper.
Sample Tested
The mixed paper-plastic sample used in the experimental work was a fraction of shredded refuse having an average moisture content of about 15 percent. The material was shredded to between 1 and 3 inches in size. No analysis was made as to the distribution of various plastics in the sample. Small amounts of the most abundant types of plastic, which included polyethylene, polystyrene, vinyl acetate, polyvinyl chloride, Teflon, nylon, and Mylar D, were added to the sample to insure that all types of plastics were present. The average composition of the material with regards to the paper and plastic content is given in table 1. The total plastic content of the sample was 9.3 percent.

Apparatus and Procedures Used
Electrostatic separators have been used by the minerals industry for many years for processing a variety of ores. Principles of operation are described in detail by Taggart and Dyrenforth. As shown in figure 1, the apparatus used by the Bureau for processing urban waste consists of (1) a combination 4-inch-diameter aluminum electrode with a wire electrode, (2) a vibratory feeder, (3) a 12-inch-diameter by 22-inch-wide variable-speed rotating drum, (4) a brush, and (5) an adjustable stream splitter. A view of the actual unit is shown in figure 2. Power supplied to the unit can be varied between 0 and and 200 kilovolts.
In separating the plastics and paper, the material is fed to the electrodynamic separator by a vibratory feeder. The material falls onto the rotating drum and is carried into the corona formed between the wire-beamed electrode and the grounded drum. The paper is drawn toward the electrode, while the plastics adhere to the drum. As the drum rotates, the plastics are brushed free at the bottom.
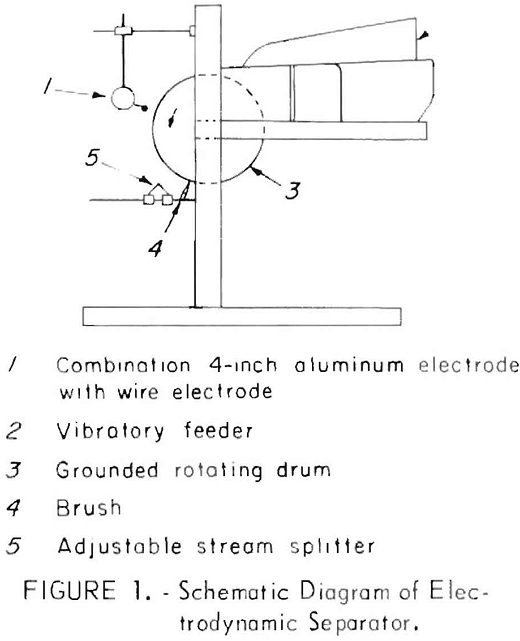
Initial separation tests established that the operating voltage required is dependent on the electrode-to-drum spacings. Good separations of the 1- to 3-inch shredded material were obtained at voltages ranging from 35 to 50 kilovolts at a spacing of about 6 inches. The tests reported herein were made at 45 kilovolts with the drum maintained as the negative pole and the wire electrode as the positive pole.
Preliminary tests made on the shredded material revealed that although the plastics and paper could be separated, several passes were necessary to produce a paper-free plastic product. Samples of the mixed feed material were subsequently dried, and the test procedure was repeated. The results of these tests were surprising. The separation efficiency decreased substantially, apparently in accordance with previously reported data on materials of different water repellency. A series of tests were then made to determine the effect of moisture on the separation of plastics and paper. The moisture level of the material was varied by tumbling the shredded product vigorously in a perforated drum while adding a predetermined quantity of water to insure that all of the material was sufficiently dampened. The data obtained from these tests are shown in table 2.
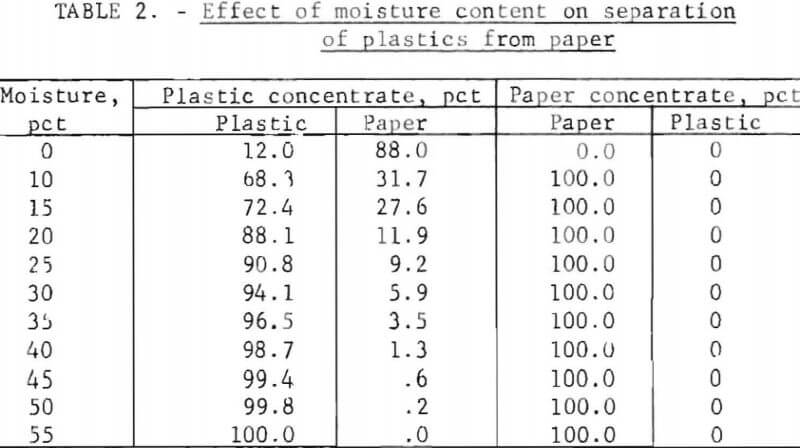

The data show that the moisture content of the refuse had a pronounced effect on separation efficiency. Using a feed containing 15 percent moisture resulted in a paper fraction free of plastic; however, the plastic material was contaminated with a substantial quantity of paper. Increasing the moisture content above 50 percent resulted in a complete separation of the plastics and paper in one pass.
Separation of plastics from paper is based on the fact that paper is a partial conductor and plastic is a nonconductor. When water is added to a mixture of plastic and paper, the paper absorbs the bulk of the water and becomes more conductive. The plastics do not absorb enough moisture to affect their conductivity. This principle enables a clean separation to be made on a mixed paper-plastic product.
To verify these data, a series of repetitive tests were made using the shredded product with water added to a moisture content of 55 percent. The results uf these tests given in table 3 show that the separated plastic fraction analyzed at least 99.0 percent plastic and that the lowest recovery obtained was 98.9 percent. The plastic material was contaminated because small bits of moist paper adhered to the plastic surface.

The data in table 3 also show that the paper fraction recovered from the electrodynamic separations analyzed at least 99.8 percent paper. Some contamination occurred as a result of conductive coatings on some plastic film. This material, along with small amounts of rubber and cloth, reported in the paper fraction.
Tests were also conducted using reversed polarities. In these tests, the drum was positive and the electrode was negative. The results of the tests were the same regardless of the polarity used. The plastics adhered to the drum while the paper was pulled toward the electrode.
Conclusions and Future Plans
The Bureau’s research has shown that electrodynamic separation is a technically feasible method for separating plastic from a mixed plastic-paper product recovered from refuse. Recoveries of up to 99.4 percent of the plastic and 100.0 percent of the paper are attainable by the electrodynamic separation technique.
To date, tests have been made on a semicontinuous basis on small batches of materials. Research is now being focused on optimizing the feed rate and feed size to the separator so that the unit may be incorporated into the continuous pilot operation.
Research is also being aimed at separating the plastics into major classes, such as polyethylene, polystyrene, polypropylene, Teflon, nylon, and polyvinyl chloride. Preliminary investigations have already shown that a substantial quantity of polyvinyl chloride can be separated from the other plastics using a 4-inch-diameter electrode.
