Table of Contents
Functional mineral pigments are used in large quantities in paint, paper, plastics, rubber, textiles, agriculture, and several other industries.
Originally extender pigments were employed only to lower cost. They were thought of merely as adulterants to replace more expensive prime pigments and binders. However, over the years their role has become better understood, and today their main functions are:
- Increasing strength, stability, and service life.
- Improving surface properties.
- Enhancing application and processing properties.
- Extending prime pigments.
- Controlling viscosity and package stability
- Lowering overall costs of the finished product.
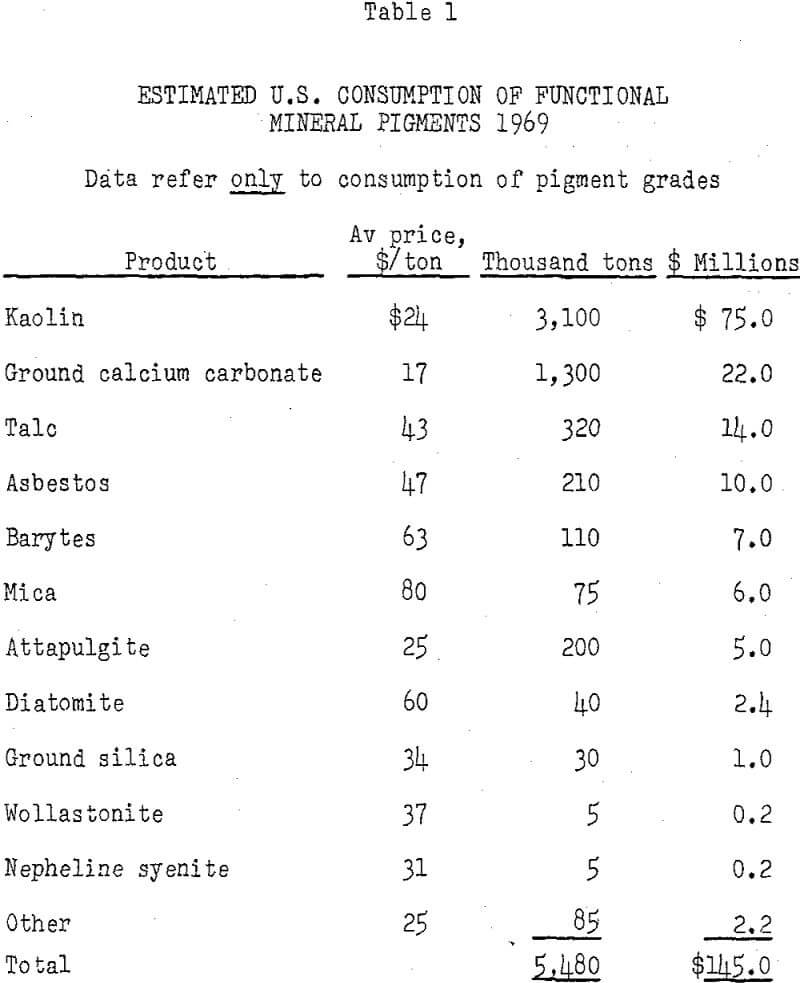
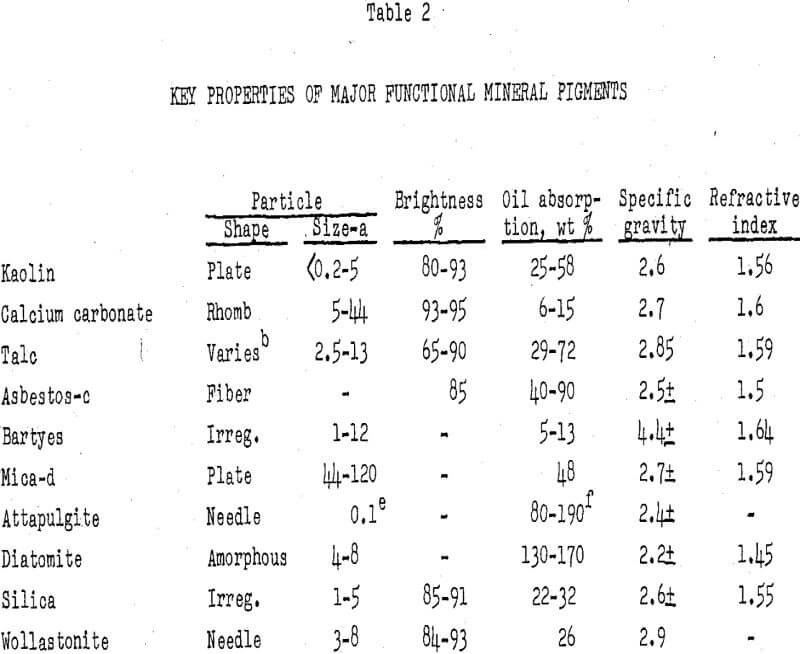
The principal minerals used as functional pigments are kaolin, ground calcium carbonate, talc, and asbestos. Other minerals include barytes, attapulgite, diatomite, mica, ground silica, wollastonite, and nepheline syenite. Several synthetic products, including precipitated calcium carbonate; precipitated, gel, and fumed silica; alumina trihydrate; and barium sulfate, compete with mineral products.
Kaolin
Kaolin is the most widely consumed functional mineral pigment both in tonnage and dollar volume. Its major end uses are coating and filling paper and filling rubber. Other uses include paint, plastics, adhesives, textile coating, and caulks and sealants.
Kaolin, with the chemical composition Al2O3·2SiO2·2H2O, is frequently referred to as aluminum silicate pigment, China clay, or simply as clay. It is mined and processed in the Carolinas and Georgia, and also in England.
According to the degree of processing, kaolin falls into three general grades:
- Air-floated
- Water-washed
- Calcined
As mined, kaolin is a soft friable mass that must be reduced to fine particle size. The clay is removed by surface mining. For air-floated grades it is crushed dry and classified by air flotation. Air-floated kaolin is the cheapest type; it contains more impurities and is more abrasive and less bright than the water-washed and calcined products.
Rubber Kaolin is the most widely used nonblack rubber pigment. It acts as a low cost filler. Unlike ground calcium carbonate the so-called hard clays of finer particle size have a pronounced stiffening effect on the modulus of rubber compounds. Other important properties it imparts to such compounds include hardness, abrasion resistance, and tensile strength. Applications of hard clays include heels and soles, footwear, wire and cable jacketing, and various molded and extruded products.
Ground Calcium Carbonate
Ground calcium carbonate is the most widely used functional pigment next to kaolin, both in tonnage and dollar volume. Finely pulverized and micronized forms of limestone and marble are most commonly used, along with some ground shell and precipitated calcium carbonate. The finest products, which pass a 325-mesh sieve, are known as whiting. The largest applications are in paint, plastics, rubber, putty, and textile coating and backing.
Ground calcium carbonate is made by quarrying the stone, crushing, grinding, and classifying to the required particle size. The stone can be ground by either the wet or dry process. In the dry process the crushed stone is ground with a hammer or roller mill. Compressed air carries the fine particles out of the mill. The pressure of the compressed air determines the particle size of the product.
In wet grinding crushed stone is mixed with water and processed in a ball or pebble mill. Water-ground whiting is more uniform and has a smaller particle size than the dry-ground products. Water-grinding can be followed by flotation and separation in settling tanks, yielding particles as small as 1 to 2 microns if enough settling tanks are used.
Ground calcium carbonate is the leading extender and filler pigment for surface coatings. It is widely used in both exterior and interior flat and semigloss house paints. Paints containing whiting have excellent leveling and resistance to polishing. In interior flat paint, the prime consumer of extender pigments, it performs several functions:
- Lowering cost
- Imparting flatness and dry hiding
- Adding solids
- Increasing viscosity
- Contributing to opacity
Talc
Talc is a natural magnesium silicate with the theoretical formula 2MgO·4SiO2·H2O. Commercial talcs seldom have
the ideal composition and ere often mixed with varying percentages of magnesite, dolomite, quartz, and other minerals. Little effort is usually made to remove these materials. Raw talc is mixed, crushed, and ground; some grades are washed and others are air-floated.
The most important single application of talc is in exterior house points. These coatings have excellent durability, suspension, viscosity stability, and application characteristics. The film weathers slowly and uniformly by gentle chalking or erosion, and eventually provides a desirable repaint surface.
White exterior latex emulsion paints also frequently contain talc to give good consistency control, suspension, color, and application properties. Talc is also used in stain-resistant paints because it does not discolor by wash-down from iron or copper screens and hardware. Large quantities of talc go into traffic paints and a variety of such industrial finishes as structural steel primers, industrial flash, and one-bake primers.
Diatomite
Diatomite consists of the siliceous skeletal remains of aquatic, single-celled algae called diatoms. Large deposits are found throughout the world. The United States produces about one-third of the world’s diatomite; California, Nevada, and Washington are the largest domestic producers. Most diatomite is consumed as filter aids and insulation, but pigment and parting applications are also sizeable outlets.
Because of its intricate structure, diatomite has unique properties among the natural simple silicas. Among these are:
- High porosity
- Low density
- High oil and water absorption
- Inertness
- Great bulk per unit weight
Many grades are produced in various particle sizes and in colors ranging from white to light gray or light pink when calcined. They contain 83 to 95% silica.
The major use of filler grade diatomite is probably as a conditioning agent to prevent the caking of hygroscopic materials, chiefly prilled ammonium nitrate. It is added at a level of about 3%. Because of its high absorption, this small amount prevents caking.
Mica
The principal type of mica used as a functional pigment is the mineral muscovite, a complex silicate of aluminum and potassium with the formula K2O·Al2O3·6SiO2·2H2O. Mica occurs in large sheets of crystals known as books. Those unsuitable for sheets for the electrical industry are ground, mainly for use as filler pigments, by both wet and dry methods.
The largest use of mica is in joint cements, mainly in dry-wall compounds. Mica imparts strength to the compound by interleafing and preventing cracking when the compound dries. It has excellent slip and gives a buttery consistency to compounds applied by knife. It also disperses well so that dry cements formulated with mica can be readily mixed in the field.