Table of Contents
The alternatives considered for accelerating the concentrate leach kinetics were addition of air or oxygen, and Degussa Corporation’s peroxide-assisted leaching (PAL) process. Air addition was tried, but only produced a voluminous froth which overflowed the leach tank, because of the presence of residual concentrations of flotation reagents. Therefore, addition of oxygen was ruled out because of expectations of a similar reaction.
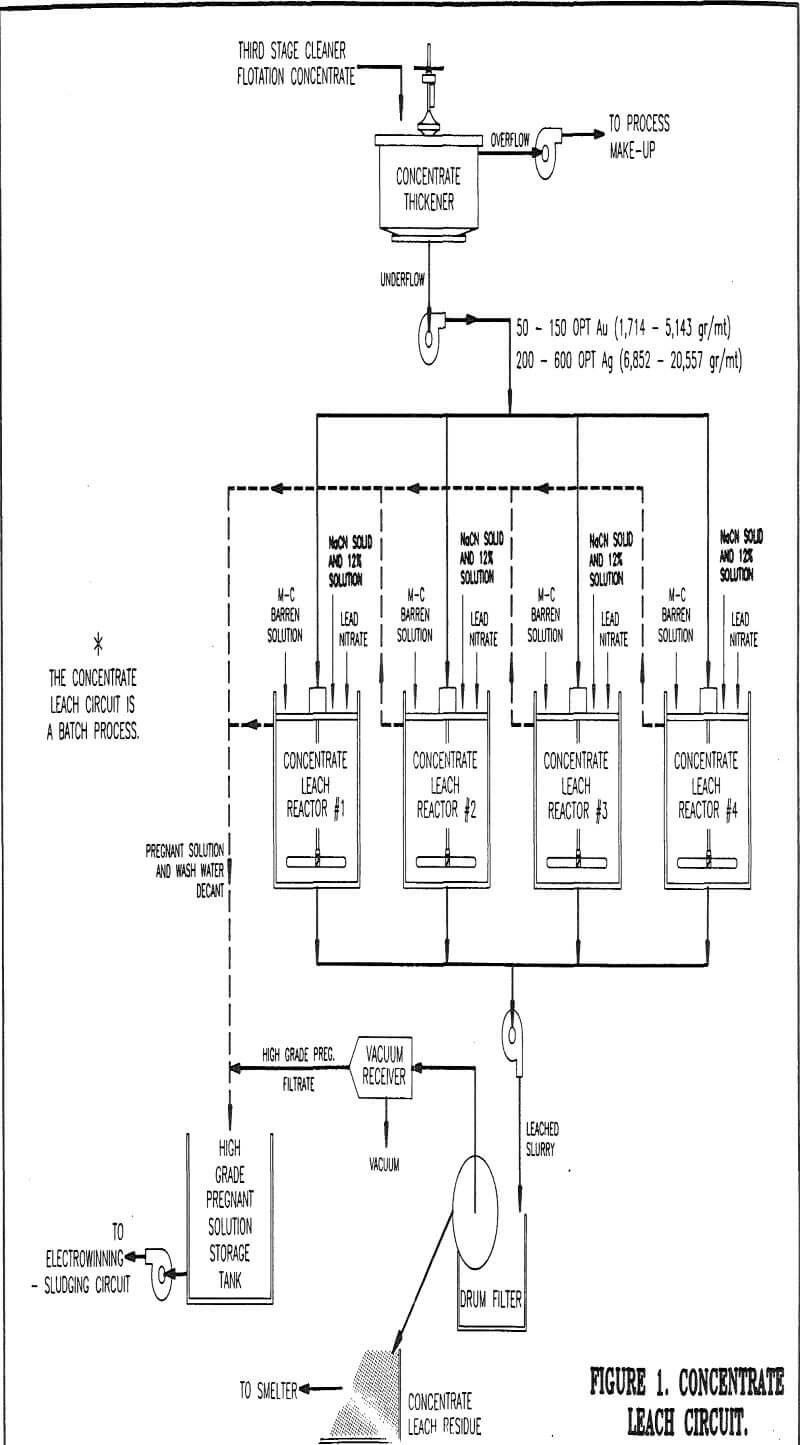
Concentrate Leach Circuit
Third stage cleaner flotation concentrate gravity flows to the concentrate thickener. The thickener feed has a fine particle size distribution, 80% minus 37 microns, which is a result of the rougher concentrate regrind circuit. Regrinding is used to liberate gold from pyrite encapsulation as well as allowing production of high grade concentrate.
At the start of leaching, dry sodium cyanide is added until the solution concentration is maintained at 25 pounds per short ton (12.5 kg/mt). A five pound per short ton (2.5 kg/mt) dosage of lead nitrate is also used to reduce thiocyanate formation. Liquid sodium cyanide is fed throughout the leach cycle to maintain a constant 25 pound per short ton (12.5 kg/mt) solution strength.
Upon completion of the required leach time, flocculant is made up and then added to the reactor. After mixing for a short period of time, the agitator is stopped and the solids are allowed to settle.
Process Theory
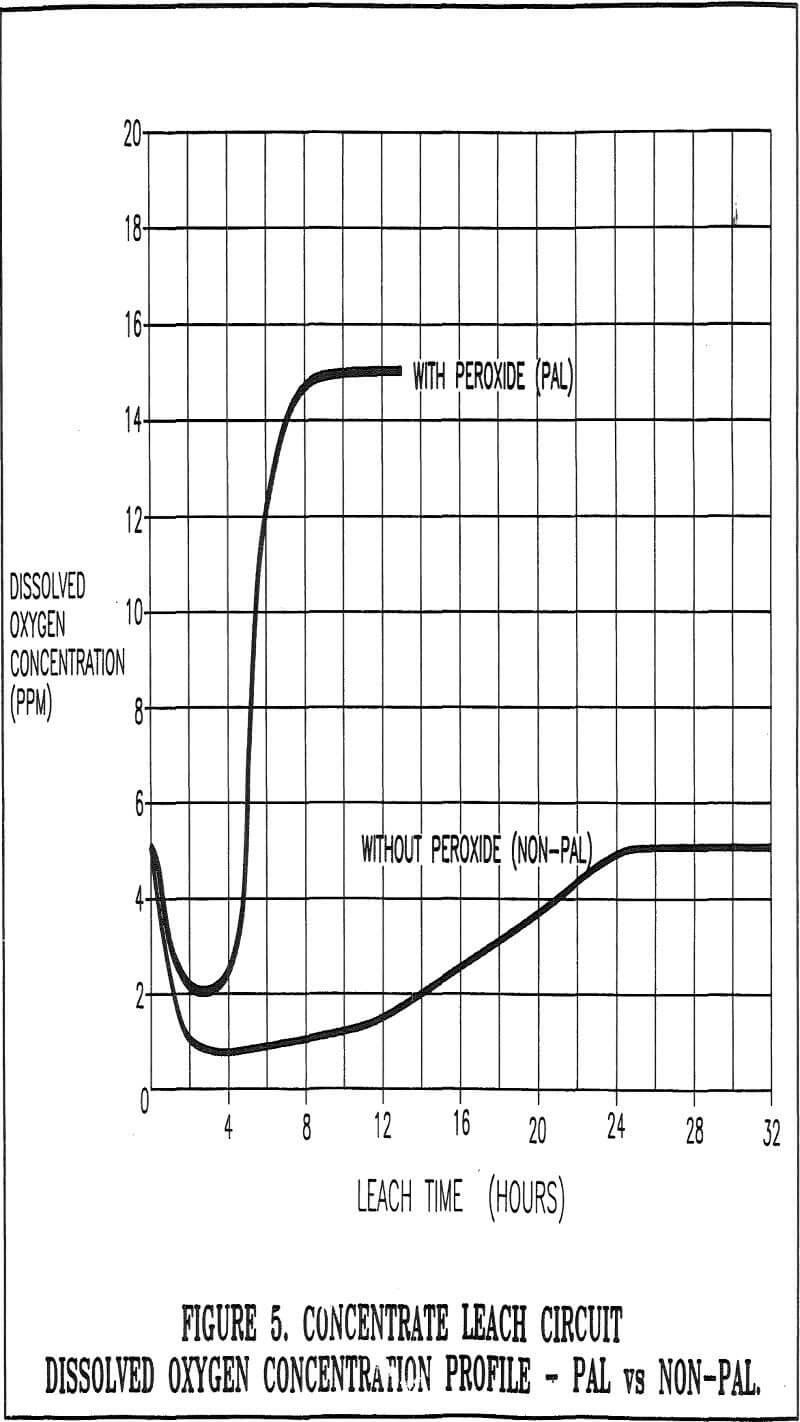
The oxygen formed by the decomposition of the hydrogen peroxide is extensively dissolved in the liquid phase. The oxygen is therefore homogeneously distributed in the leach slurry, which enhances mass transfer characteristics over conventional aeration techniques. Oxygen concentrations above the saturation limit of the slurry (about 8 ppm) can be easily achieved, if required.
Test Program
The objectives established for the PAL test program were the following:
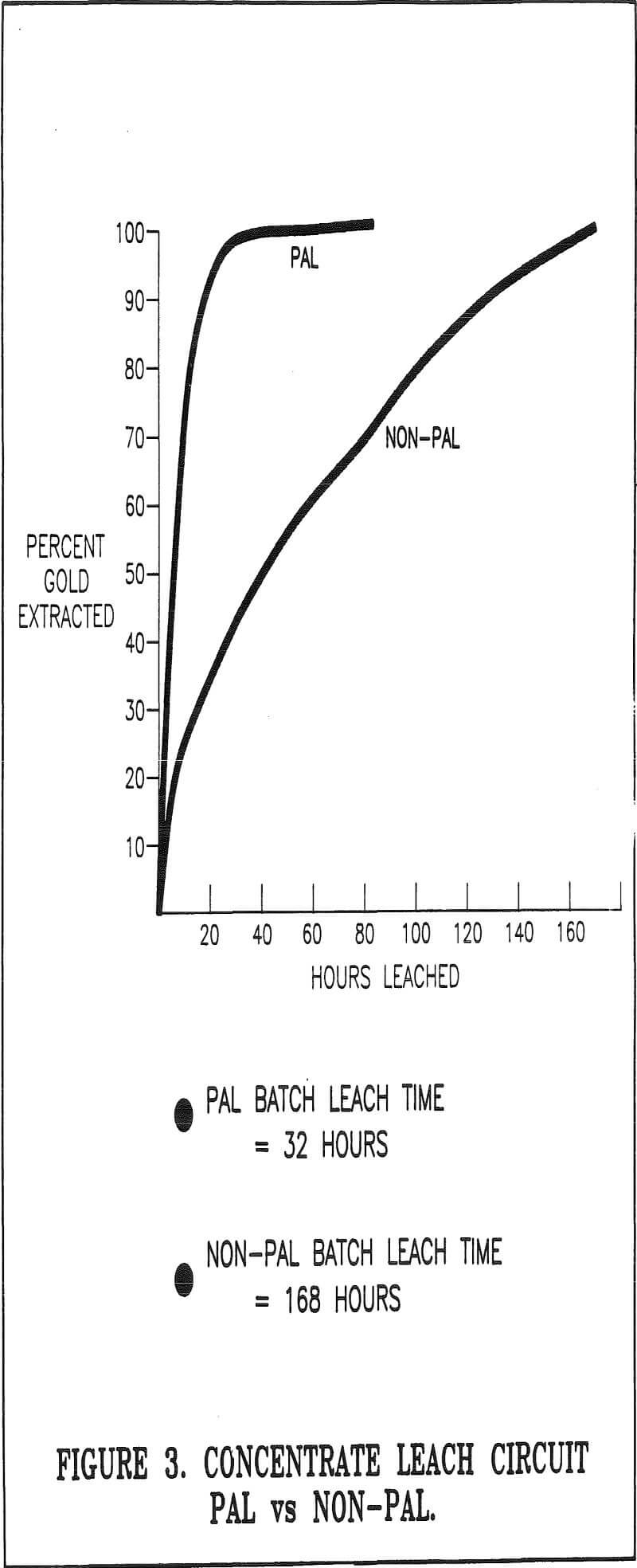
- Reduce leaching time by 35 to 50%. This range of reduction was selected based on the successful performance of PAL at other gold operations.
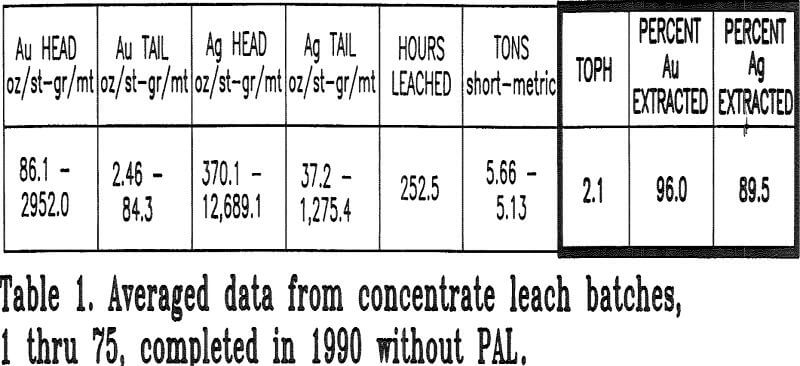
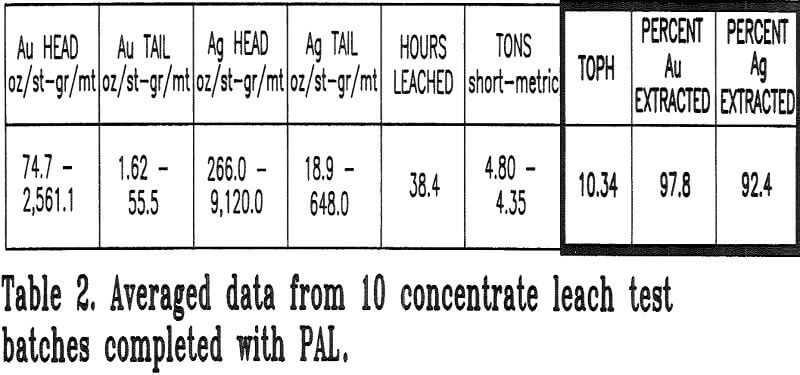
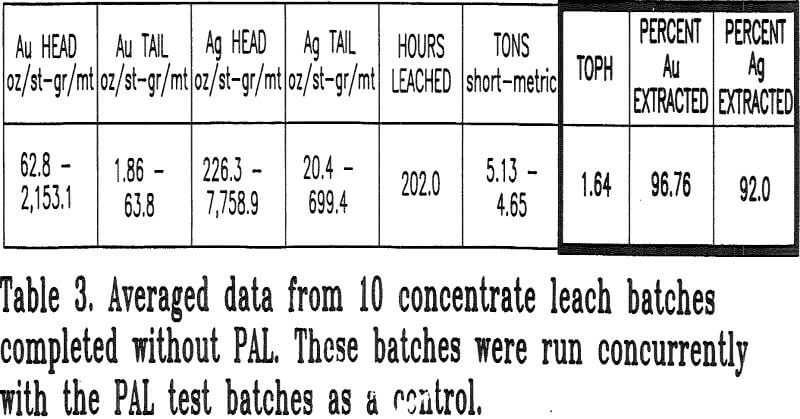
- Recovery of gold and silver could not decrease. The average recoveries prior to PAL testing were 96% gold and 89.5% silver.
- The economics of the concentrate leach operation could not be adversely affected.
- The PAL feed system had to be safe and easy to use.
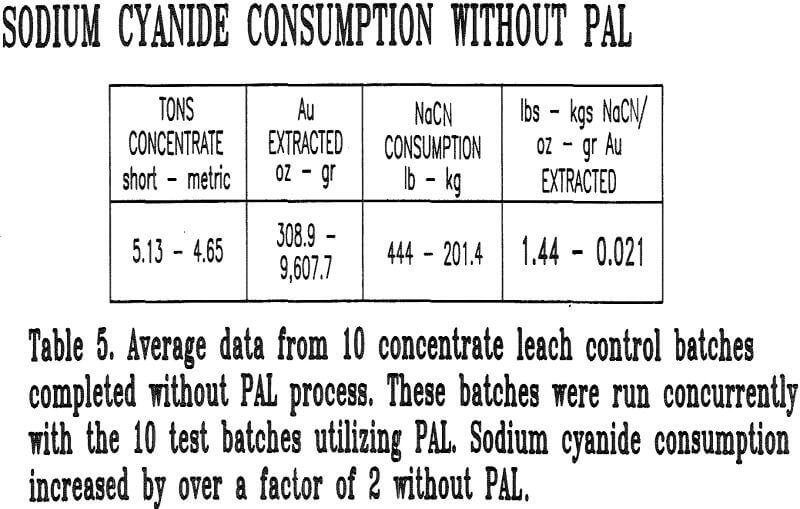
Batches without hydrogen peroxide addition were also leached concurrently with the PAL batches to ensure comparative evaluation of similar concentrates. These batches were monitored for the same parameters as the PAL batches. A total of 20 batches with and without the use of PAL (10 each) were tested.
Results of Testing
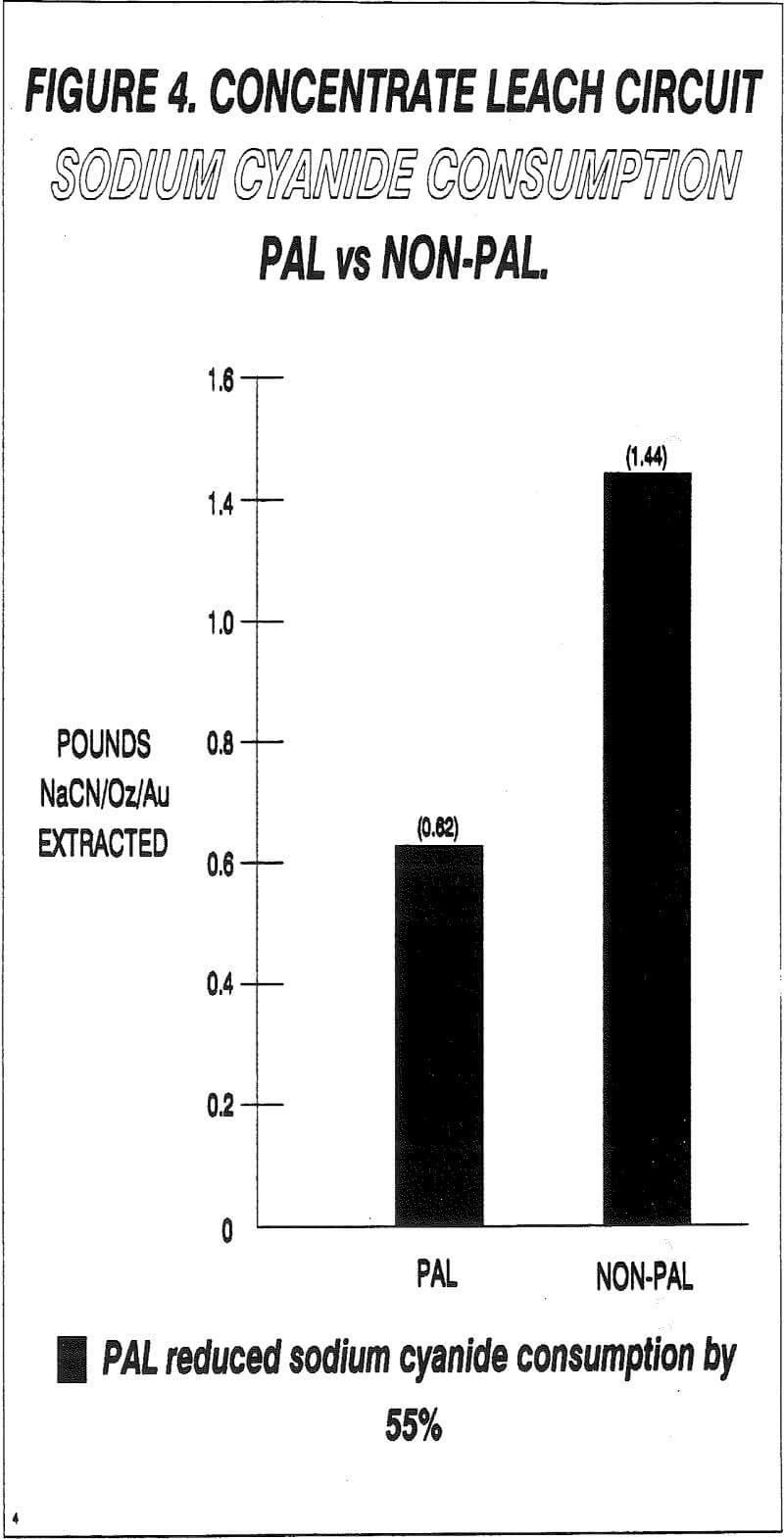
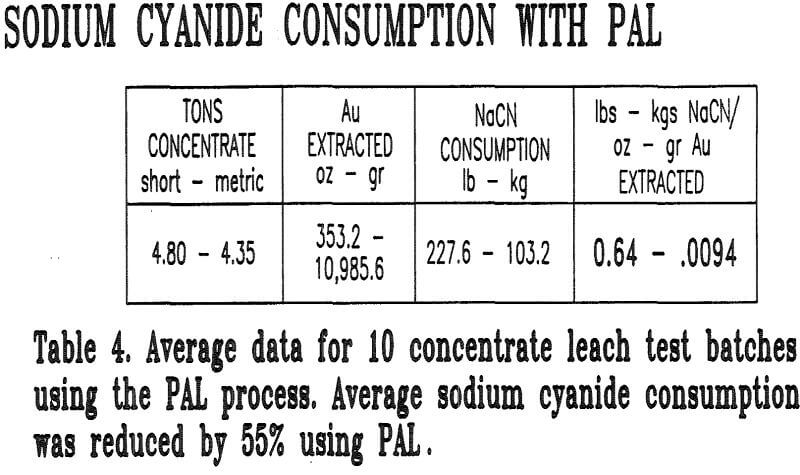
The PAL process was found to reduce sodium cyanide usage by 55%. This reduction can be attributed to three factors:
- The increased rate of gold dissolution decreases the leach retention time required, which in turn reduced the amount of cyanide wasted on the formation of compounds such as copper and iron cyanide complexes.
- There is also evidence that suggests that the PAL process passivates the surface of sulfide minecals such as pyrrothite and arsenopyrite. This seems to hold true for pyrite also. Therefore, it is possible to decrease the amount of thiocyanates formed during the leaching process, thus reducing cyanide usage.
- Cyanide loss by formation of HCN is also decreased because of the reduced leach time.
PAL Economic Benefits
- Gold revenues increased slightly because of greater concentrate leach extraction (1 to 2%) and less shipment of gold to the custom smelter.
- The reduction in leach time requirements saves on equipment wear and power consumption.
- The existing concentrate leach circuit can comfortably process up to 5 times more concentrate production, without expansion, should future needs arise.
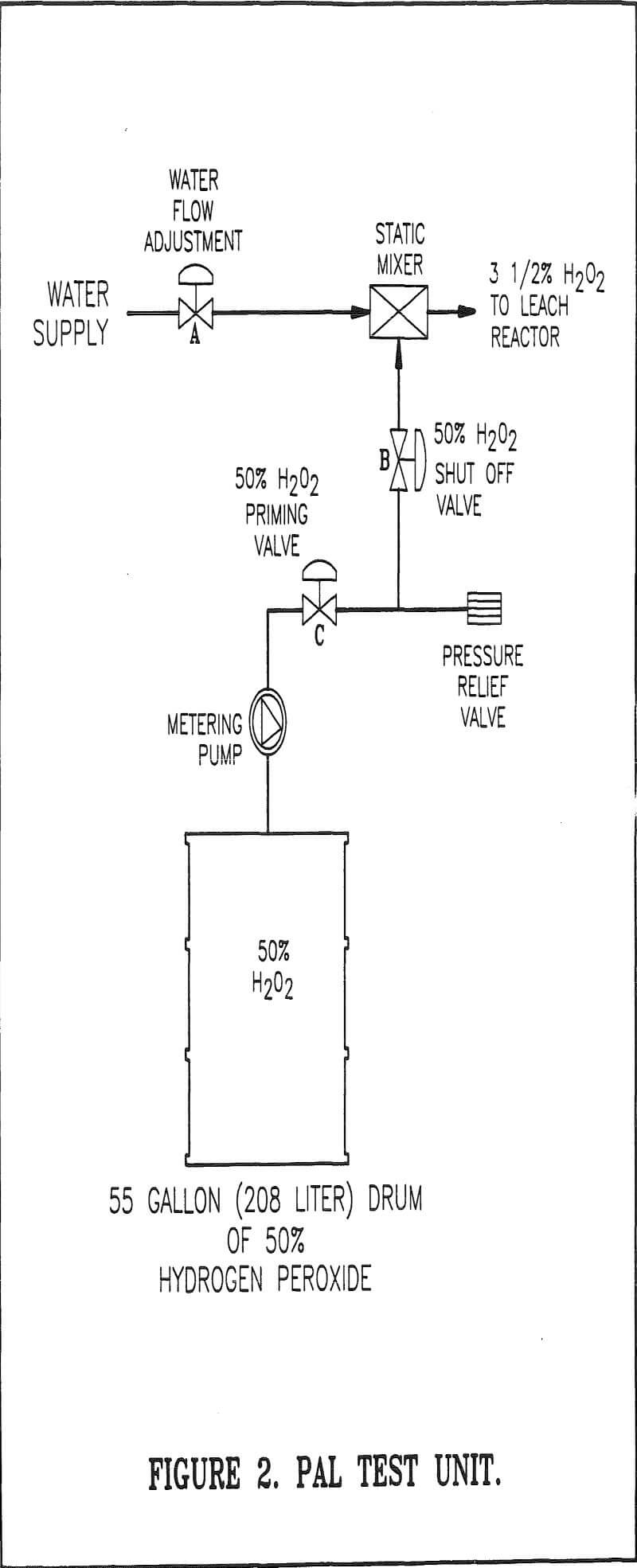
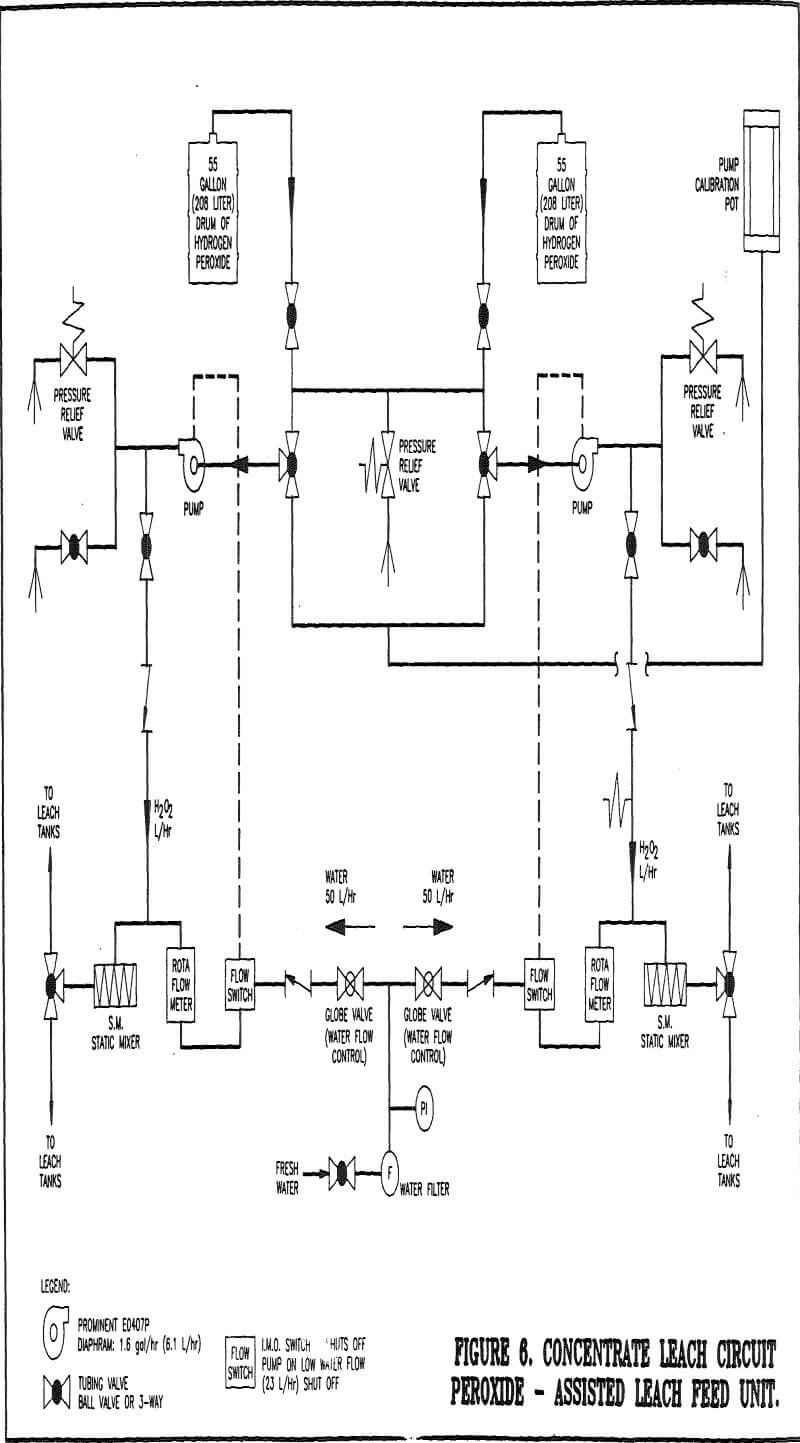
Feed System
Because of the excellent test results achieved PAL has become standard practice in the concentrate leach circuit operation. Dilute hydrogen peroxide is made available to each leach reactor through the use of the PAL feed unit. The PAL feed unit was designed by and purchased from Degussa Corporation. The cost of the feed system will be paid-off in a year through savings in sodium cyanide, alone.