Table of Contents
There has been considerable discussion of late as to the best method of determining the silver-contents of sulphides of silver resulting from the leaching of silver-ores, and also as to the relative merits of the crucible and the scorification method for the determination of the silver-contents of ores.
Owing to the great depression in the silver-market, these are questions of considerable importance, at the present time, to both the producer and the smelter and refiner.
A few ounces of silver sulphide were prepared by dissolving quite pure silver chloride in sodium hyposulphite and precipitating the silver as a sulphide by the addition of sodium sulphide to the solution. After filtration and slight washing, the precipitate was dried, ground, and thoroughly mixed; and the percentage of silver was then determined by carefully weighing out several portions of 0.05 A. T. each, on the assay-balance used for weighing the silver buttons resulting from assays; dissolving each portion in nitric acid of 27° Beaume; boiling to expel the red fumes; diluting with distilled water to 200 c.c., and titrating with a standard solution of potassium sulphocyanate; adding about 1 c.c. of a strong solution of ammonium ferric alum as an indicator (Volhard’s method). This method was adopted because the Gay-Lussac titration with standard salt solution could not be used on account of the sulphur set free during solution and the consequent cloudiness of the solution. This free sulphur would also have interfered with the gravimetric determination (precipitating the silver as a chloride and weighing it as such). Volhard’s method, provided copper is absent, gives very correct results, and has been used by the writer for years with entire satisfaction. The average silver- contents of the sulphides, as thus shown by several closely agreeing determinations, was 19,693 ounces per ton of 2000 pounds.
The silver was next determined by the combination method as follows: Two portions of 0.05 A. T. each were dissolved in nitric acid (27° B.), and after boiling out the red fumes each solution was diluted to 300 c.c. with distilled water, and 110 c.c. of normal salt solution (1 c.c. = 10 milligrammes silver) were added, and the solution was vigorously stirred. After allowing the precipitate to settle slightly, 10 c.c. of a strong solution of lead acetate were added, and then 1 c.c. of strong sulphuric acid, the latter drop by drop. After stirring, the solution was allowed to settle over night. The precipitate was filtered off through a heavy filter-paper and dried. The precipitate was enveloped in the filter, placed in a 2.5-inch scorifier, and the filter burned by placing it in front of the muffle- furnace. After the filter-paper had been consumed, the scorifier was placed back a few inches into the muffle, to consume all the carbon. After cooling, the residue in the scorifier was mixed with 8 grammes of litharge; 20 grammes of test-lead and 0.5 gramme of borax-glass were added as a cover, and the charge was carefully scorified. The resulting lead buttons (weighing about 10 grammes each) were cupelled in front of the muffle, and the resulting silver buttons were weighed, the result being: a, 19,590 ounces per ton; b, 19,625 ounces per ton; average, 19,608; average per cent, of the silver present thus found, 99.56.
The a assay was made on the pure material, while to the b assay were added (in order to introduce such elements as are liable to be present in the sulphides as produced commercially) copper, 0.5; arsenic acid, 0.5; and antimony oxide, 0.5 gramme.
The following table gives the results of the different determinations by both scorification- and crucible-assay:
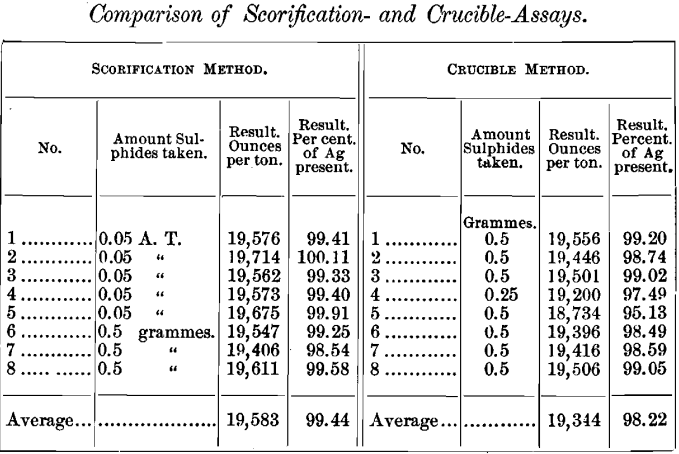
Scorification-Assays
The assays were run in the usual manner, from 30 to 40 grammes of test-lead and from 0.5 to 1 gramme of borax-glass being used. In addition to the regular fluxes, there were added the following reagents:
No. 1. Grammes: 0.5 of Sb2O4, 0.5 of As2O5, and 0.5 of Cu. The button, after cupellation, weighed 19,764 milligrammes; but, as it showed the presence of copper, it was recupelled with 1 gramme of lead, and the weight of this second button is the result reported in the table.
No. 2. The charge was the same as in No. 1. The button weighed 19,841 milligrammes, and was dissolved in nitric acid, the solution showing the presence of copper. The result, as reported in the table, was obtained by titration of the solution from the button with standard sulphocyanate solution, which method would necessarily give a high result, owing to the presence of copper.
No. 3. This was run without the addition of fluxes other than the regular charge of test-lead and borax.
No. 4. Same as No. 8.
No. 5. Added, grammes: BaSO4, 0.5; Sb2O4, 0.5; FeS, 0.4; Cu, 0.5; Zn, 0.5.
No. 6. Added 0.5 gramme of Cu.
No. 7. Added 0.5 gramme of Zn.
No. 8. Added 0.5 gramme of FeS.
Crucible Assays
The fusions were run in 10- gramme crucibles, the fusion being performed in the muffle-furnace. The regular charges of litharge, sodium bicarbonate, borax-glass, lead flux and nails were added. The time of fusion was about thirty minutes in each case. In addition to the regular fluxes there were added the following reagents:
No. 1. Added 0.5 A. T. of pure SiO2.
No. 2. Added, grammes: SiO2, 5; Sb2O4, 1; As2O5,1 ; Cu, 1; S, 1.
No. 3. Added 0.5 A. T. of pure SiO2.
No. 4. The same as No. 3.
No. 5. Added grammes: SiO2, 5; BaSO4, 5; Fe2O3, 5.
No. 6. Added 14 grammes of pure SiO2.
No. 7. Added, grammes: SiO2, 5; Zn, 1; S, 1; Cu, 0.5.
No. 8. Added, grammes: SiO2, 5; BaSO4, 5; Fe203, 5.
In all the assays the same precautions were adopted as would ordinarily be observed in careful commercial work.
The crucible-assays where made, not because this was considered tin proper method for the assay of high-grade silver sulphides, but in order to obtain comparative results by this and the scorification-method.
Silver Sulphide Assaying Methods
It may be objected that the above determinations are too few in number to base conclusions upon. However, it is proper to say that these results but tend to confirm the previous experience of the writer.
The combination-method is by far the most accurate, and yields the most concordant results. On material containing much copper (such as the sulphides produced in the Russell process) this method yields by far the best results, and in the opinion of the writer, is preferable to the corrected scorification- method (where the slag- and cupel-absorption is determined) on account of both the greater accuracy of the results and the difference in time and labor involved. The latter is generally an important consideration in a metallurgical laboratory, where frequently a great number of assays are run in the course of a day.
The regular scorification-assay apparently yields results which may be considered as within the limits of accuracy of a commercial method.
The crucible-method is not suited for the assay of this material.
Concerning the comparative accuracy of the crucible and scorification-methods, as the writer has observed elsewhere, the scorification-assay almost invariably yields higher and more uniform results than the crucible-method, especially on sulphides and base ores. The above results appear to confirm this opinion, which was formed after a large series of assays by the regular scorification-method, one crucible-assay being run with each set of scorifications as a check. On some classes of material, such as the oxidized lead-ores and pure siliceous silver-ores, the crucible-method is preferable; but on the great bulk of ores now mined in Colorado and other States the scorification-method is better.
