Table of Contents
Based on the analysis of the W50 values of 65 permissible-type formulations, with and without added sodium chloride of various particle sizes, it was concluded that:
- The addition of sodium chloride to an explosive has a highly significant effect in reducing its probability of igniting firedamp. Addition of small amounts of sodium chloride (about 3 percent) had a measurable though inconsistent effect, and addition of 10 to 20 percent salt was accompanied by a very definite effect.
- Of the two granulations of sodium chloride used, the fine one reduced the incendivity of the explosive more than the coarse.
- The extensive experience with the up-and-down method obtained in this Investigation confirms the validity of this technique for the quantitative evaluation of the incendivity of permissible-type explosives by the gallery test. Further analysis showed that a randomized series of 20 shots was sufficient, with the specific procedure used, to provide a reasonable estimate of the mean, W50.
The results of the first phase of this investigation were presented in a previous paper, which established that adding up to 10 percent sodium chloride to permissible explosives reduces the likelihood of ignition of firedamp in the gallery test; however, the results were somewhat inconsistent, and the conclusions were qualified. Accordingly, the investigation was continued with a study of the effect of added sodium chloride as a function of the quantity added and of the particle size of the salt.
Eighty-seven permissibles and modifications of permissible formulations have been tested during this investigation, which resulted in over 2,400 gallery shots in 2 years. Many of these formulations were specially prepared by the manufacturers.
Following a preliminary orientation study, a randomized design of the up-and-down method was selected as a suitable means of ranking the incendivity of explosives. In consequence, it became a second objective of the present investigation to determine the validity of this method, one ad¬vantage of which is that it provides data that are readily amenable to statistical analysis. This paper gives the results obtained to date in the program.
Procedure
The Bureau’s large test gallery was used in this investigation. It consists of a cylindrical steel shell 6-1/3 feet in diameter and approximately 100 feet in length. Mixtures of firedamp are provided in the first 20-foot section by admitting a measured quantity of natural gas from a nearby well at a uniform rate. This gas is introduced into a 6-inch sideline near a centrifugal blower that circulates the gas through an external heat exchanger and the gallery.
The boreholes of the steel cannon used in these tests measure 2-¼ inches in diameter and 21-½ inches in length when new. When the diameter of the borehole has expanded to 2-¾ inches in the course of testing, that portion of the borehole is filled with well rammed fireclay and a plug of asbestos rope. Thus, the borehole is, in effect, shortened and restored to a diameter within tolerance limits. Successive plugs of fireclay and asbestos rope are inserted in the cannon as the diameter increase progresses toward the cannon muzzle. This practice is continued as long as the last clear portion of the borehole, after loading, is not less than 2 inches.
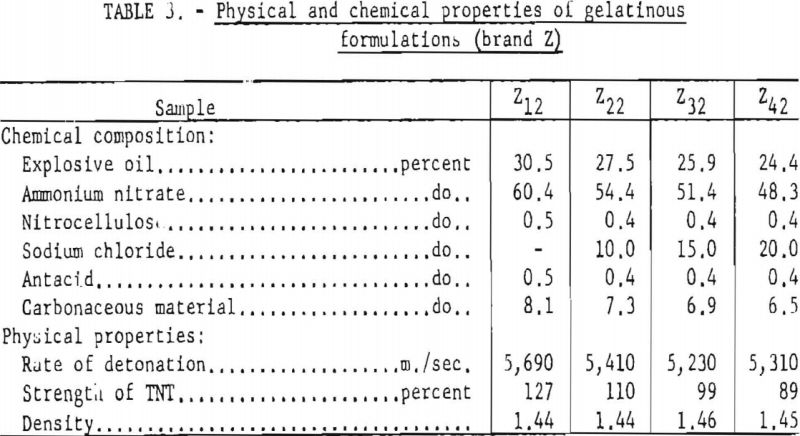
As previously stated 87 explosive formulations were tested over a period of 2 years. During the second phase of the investigation and since the first results were published, 18 specially prepared formulations, based on 1 gelatinous and 2 nongelatinous permissibles, have been studied. Tables 1, 2, and 3 give their compositions and physical properties. For each brand, the same ratio was maintained for all ingredients except the sodium chloride. Each group was prepared by a different manufacturer with varying amounts of sodium chloride (0 to 20 percent). The seven samples for each nongelatin group (X and Y samples) differ from each other in the amount and fineness of the sodium chloride. The particle size of fine salt used in the X formulations ranged from 48- to 150-mesh (Tyler standard); the fine salt used in the Y samples was all 200-mesh. The coarse salt for both samples was approximately 35-mesh. The four gelatinous formulations (Z samples) were prepared with similar concentrations of the coarse sodium chloride.
A randomized design of the up-and-down method was employed to evaluate these samples in 8-percent natural gas-air mixtures. Because the samples differed widely in incendivity, the use of different types of stemming was necessary to keep the charge weights within the limits of the cannon. Thus, certain formulations were tested with 1 pound of fireclay stemming, others with asbestos tamping plugs, and the least incendive samples without stemming. In all tests the charges were slit and tamped in the cannon. Untamped charges without stemming also were fired; these charges were prepared in an asbestos cup designed to contain the small charges encountered in unstemmed shots and insure correct positioning of the detonator.
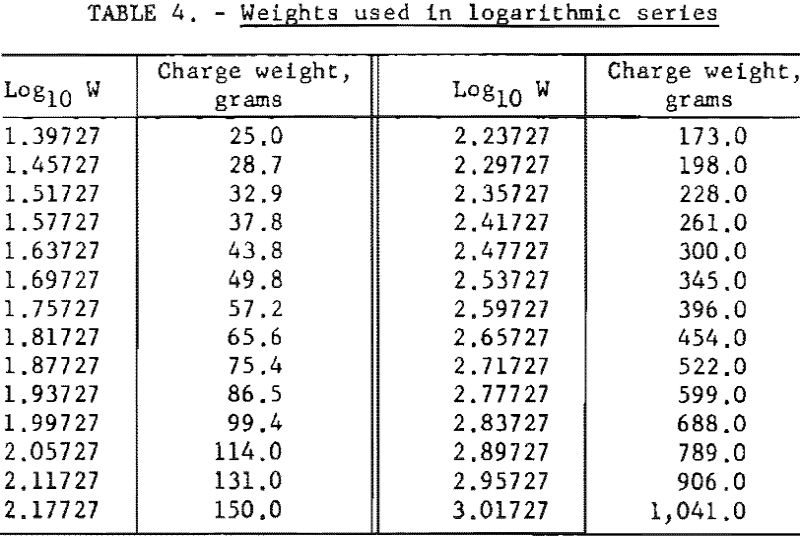
In the first stage of the investigation the validity of the test method was emphasized. For this purpose, four series of tests were conducted on sample X, which contained no sodium chloride. In one series logarithmic increments were used; in the other three series the variable was based on arithmetic increments. Sixty shots were made in each series, giving a total of 240 shots. In all subsequent tests, the increment used was 0.06 log10 W; the corresponding weights are given in table 4. Each test sequence consisted of 20 to 30 shots.
In the second stage, the three special groups of permissible-type formulations were evaluated to study the effect of sodium chloride concentration and particle size on incendivity. In addition, several old and new formulations of permissibles were compared. The latter consisted of basic permissibles modified by adding up to 10 percent sodium chloride and were prepared in manufacturers laboratories; the old formulations were production samples.
By carrying out the gallery shots as a series of randomized experiments, the several objectives of this investigation (effect of sodium chloride and validity of the test method) were pursued concurrently.
Validity of the Up-and-Down Method
If an experiment yields results of the “go-no-go” type, the up-and-down method offers an experimental design suitable for statistical analysis. However, in order that analysis may be applied and may yield valid and useful conclusions, the following three conditions must be satisfied:
- The variable of the experiment must be normally distributed. In these tests the variable is the charge weight; accordingly, the distribution of the frequency of ignitions or nonignitions, whichever is smaller, must be normal.
- The standard deviation of this variable (charge weight) must be known, at least approximately.
- The predetermined intervals of the weight employed in the up-and-down steps should be within the range of one-half to two standard deviations.
In addition, a minimum of 30 trials or shots was recommended, but under fortuitous circumstances a smaller number may lead to a reasonably satisfactory estimation of the mean weight, or W50.
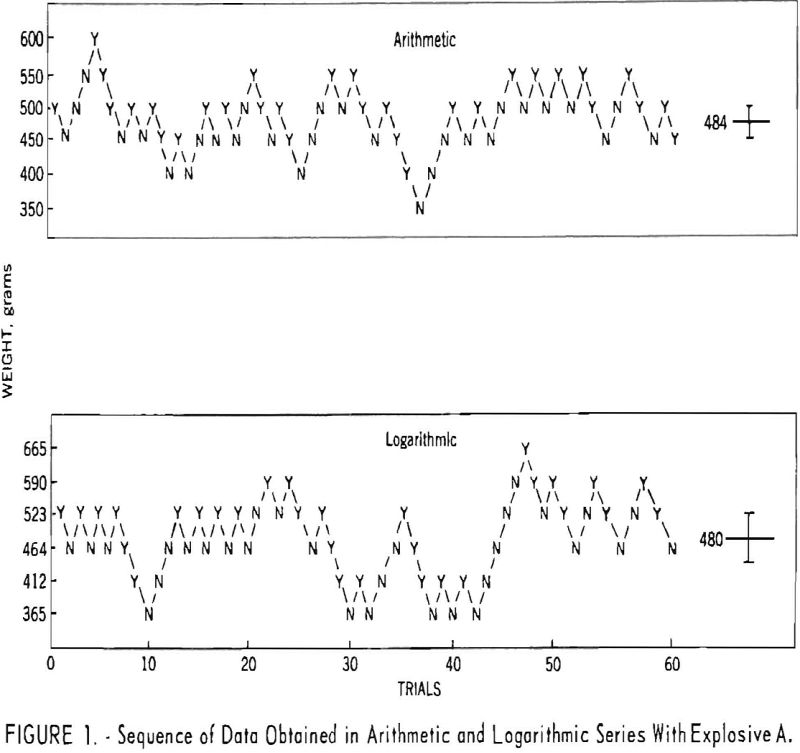
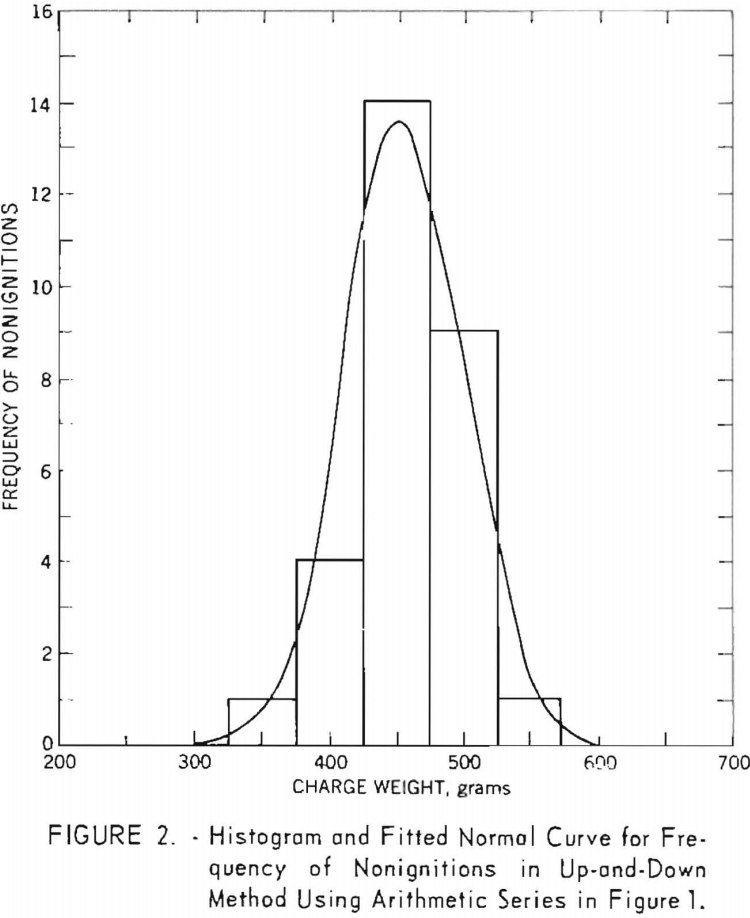
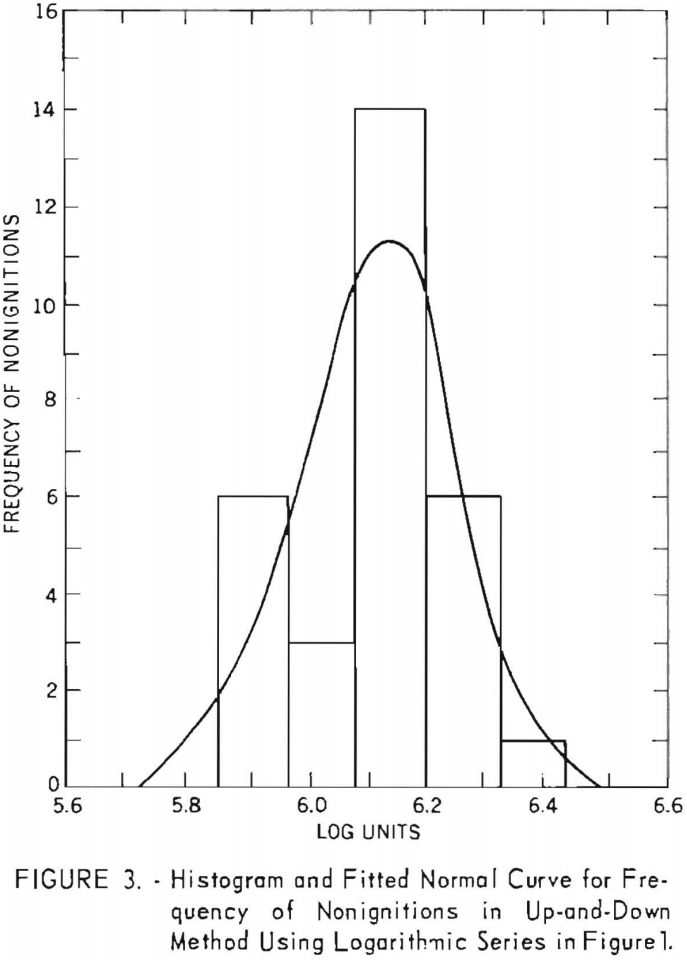
During the investigation the results were examined periodically, and the procedure was adjusted to meet the above conditions. With respect to condition, two special up-and-down series of 60 shots each were carried out with a single explosive at the outset of the investigation. The weight of explosive charge was varied by arithmetic intervals in the first series and by natural logarithmic intervals in the second. Figure 1, showing the sequence of data obtained for both series, was drawn to determine which of the two resulting distributions was better fitted by a normal curve. It was found that the arithmetic series (fig. 2) fitted the normal curve slightly better than the logarithmic series (fig. 3). However, a further study of the two methods revealed that both are acceptable with respect to distribution of data. This was demonstrated by four additional series of 60 shots each.
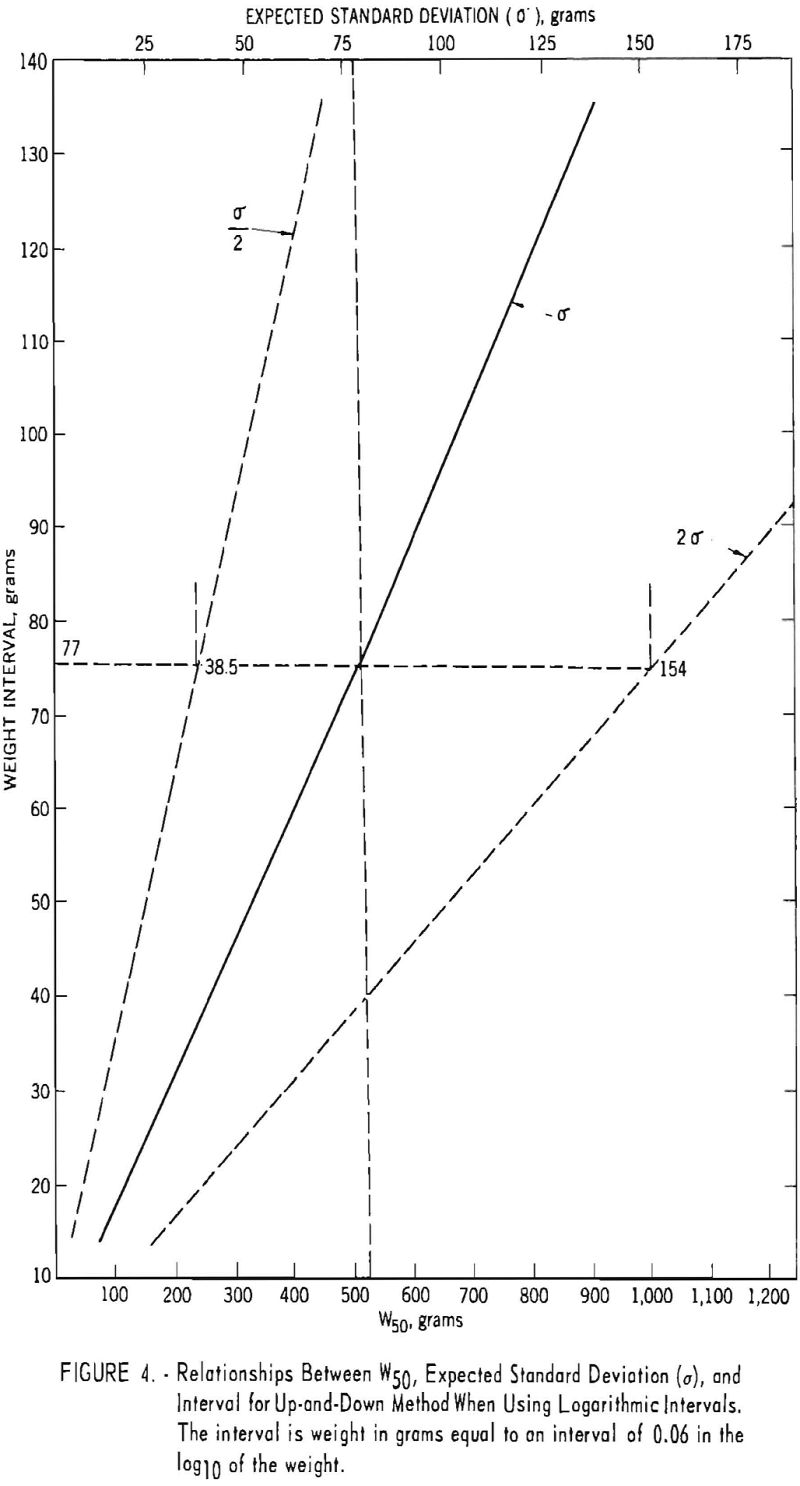
As the investigation progressed, the accumulated data furnished additional estimates of the standard deviations of the explosives, thus fulfilling requirement. Analysis of the data for 79 permissible-type explosives, including most of those in the present investigation, revealed that smaller standard deviations were associated with smaller W50 values; in fact, the average value of the standard deviation was approximately 15 percent of the W50 value. With intervals based on equal logarithmic units, this afforded a convenient means of automatically adjusting the interval size to the value of the standard deviation. Figure 4 shows the relationships between W50, the standard deviation, and the interval of weight based on the logarithmic units. This figure illustrates, for example, that if the W50 is 522 grams (lower scale) the corresponding expected standard deviation will be 77 grams (upper scale), and the numerical interval to use, corresponding to one standard deviation, will be 77 grams (left ordinate scale). But an interval within the range of one-half sigma and two sigma (38.5 to 154 grams) would meet the requirement of the up-and-down method. Accordingly, requirement was
fulfilled by the use of logarithmic intervals, and most of the up-and-down determinations of W50 were based on such units. It was also an economic advantage to use logarithmic intervals, because fewer shots were required at larger weights than with arithmetic intervals.
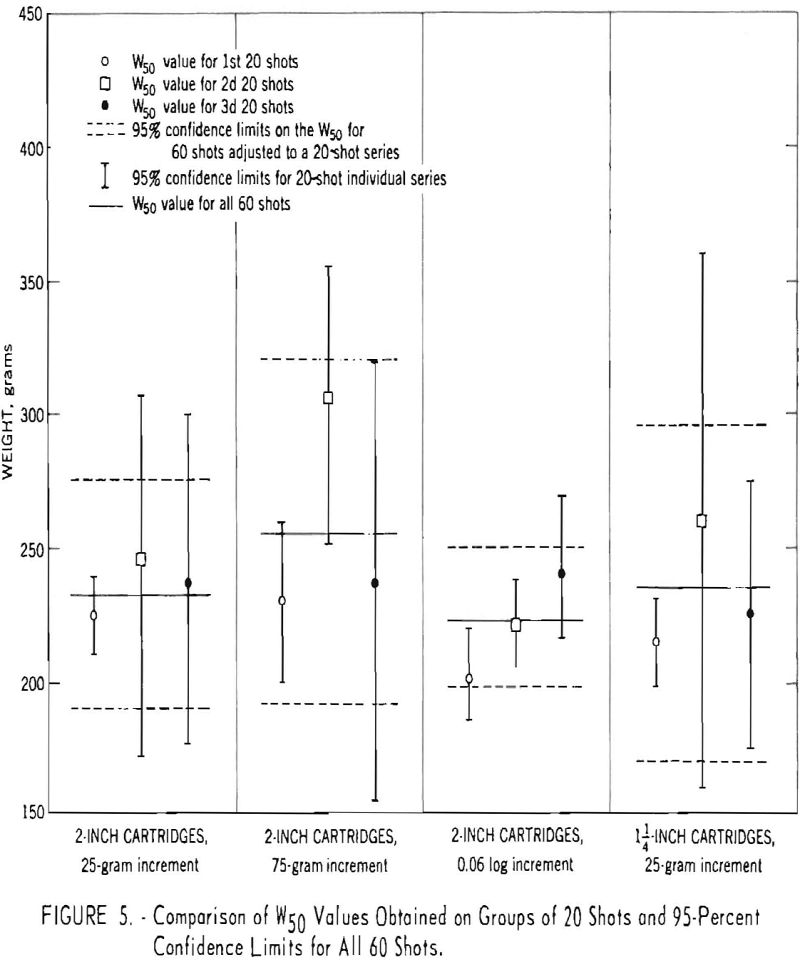
With reference to the number of shots, an experiment comprising 4 series of 60 shots each demonstrated that the up-and-down test gave reasonably good results for only 20 shots (fig. 5). Whether the increments were the arithmetic (25 or 75 grams) or the logarithmic interval, it was found that the W50 values based on 20 shots fall within the expected 95-percent confidence limits on the W50 values for 60 shots adjusted to a 20-shot series. Furthermore, each of the W50 values based on 60 shots, with 1 exception, falls within the adjusted 95-percent confidence limits of the other W50 values based on 60 shots. These confidence limits were obtained by multiplying the 95-percent confidence intervals based on 60 shots by √60/√20 or 1.7. The results of these 240 shots indicate that the W50, determined by the up-and-down method, can be reproduced satisfactorily and that 20 shots appeared adequate. The results in figure 5 also met the chi-square test requirements for normality of distribution.
Thus, it appeared that the requirements of the up-and-down method were being met and that the method could be validly applied for measuring the relative incendivity of permissible explosives.
Effect of Quantity of Sodium Chloride
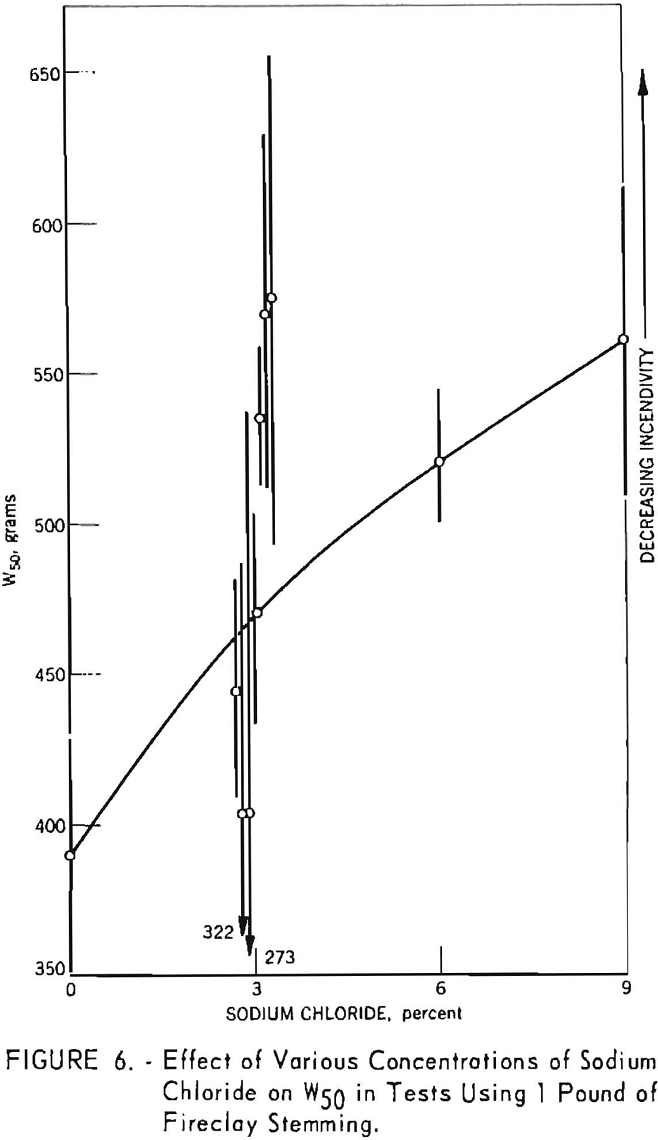
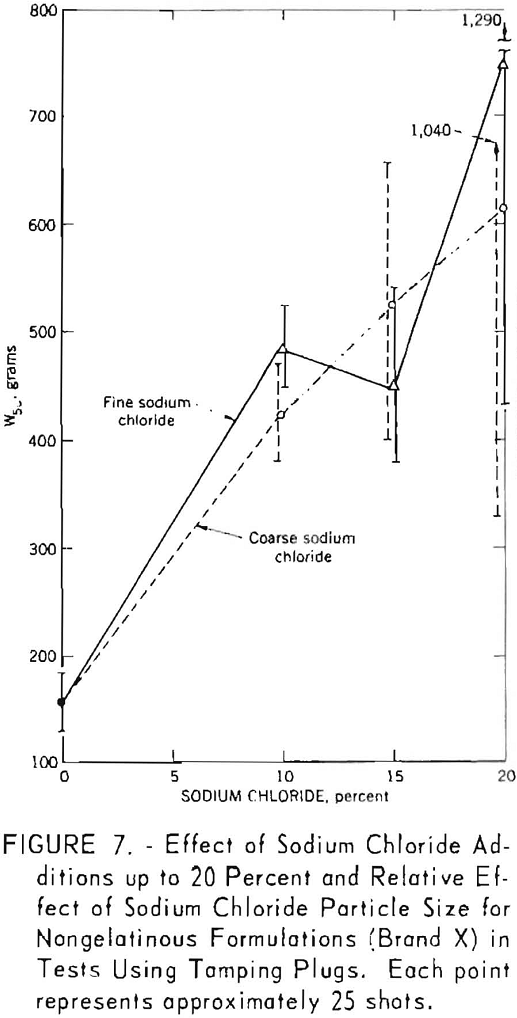
In the initial phase of the investigation a series of experiments was made with 10 modified formulations of a nongelatinous permissible explosive. The results, shown graphically in figure 6 have already been reported. The 3-percent-salt formulations were tested intermittently and therefore not randomized as a single experiment. Each W50 value was based on 28 shots; the complete experiment represents 280 shots, each stemmed with 1 pound of fire-clay. The vertical lines drawn through each point indicate the 95-percent confidence limits for the corresponding W50 value. These initial results indicated that a decrease in incendivity could be achieved by adding salt. However, inconsistencies and relatively wide scatter in the data led to a qualified conclusion and a decision to continue the study with higher salt concentrations. The results of all subsequent studies are shown graphically in figures 7 through 10. Each figure represents an individual randomized up-and-down experiment. The figures also include the results of subsequent experiments on the effect of sodium chloride particle size.
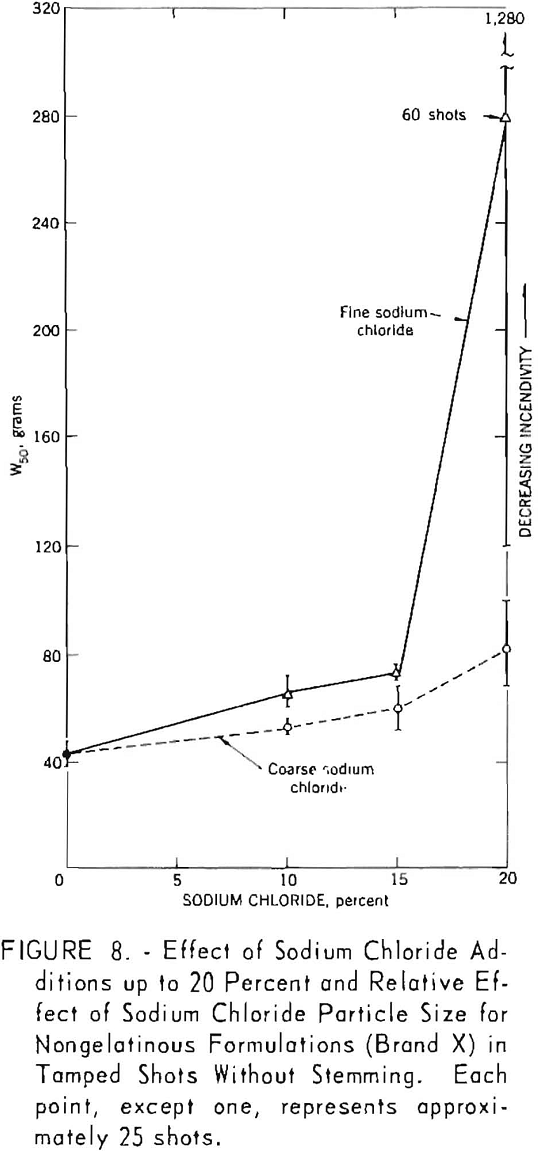
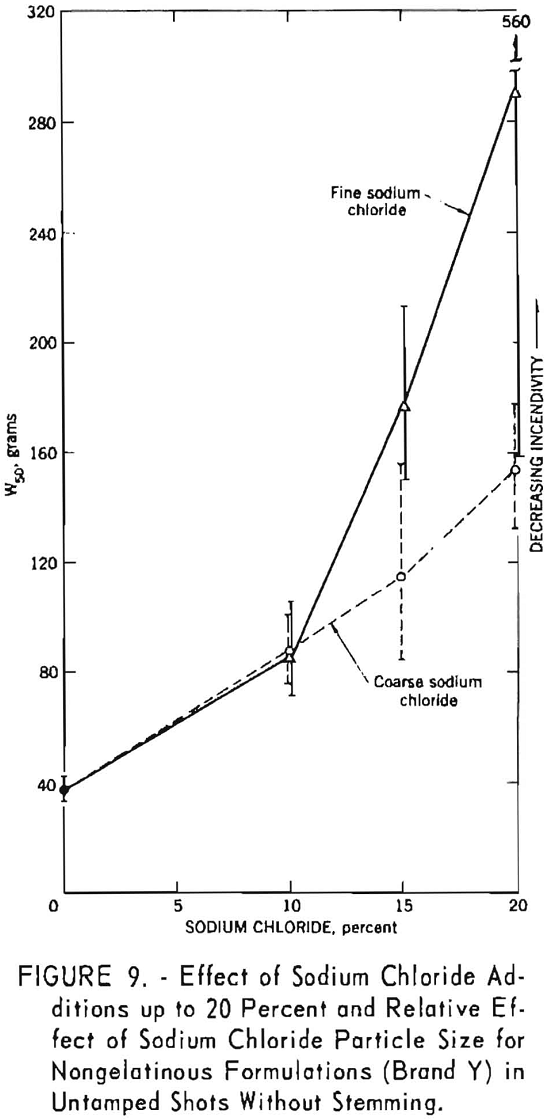
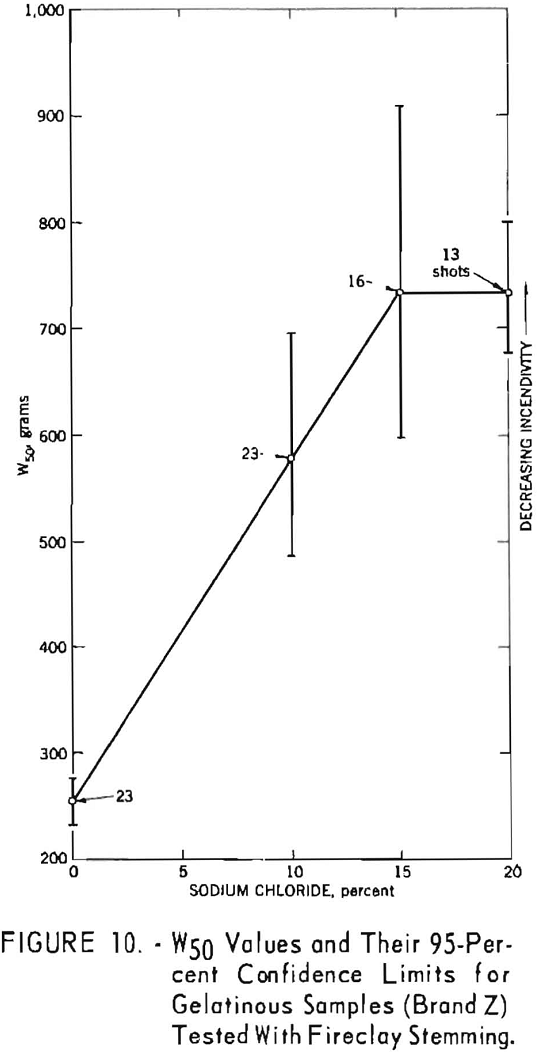
The data summarized in figure 7 were obtained from gallery tests conducted on the seven composition variations of a nongelatin (brand X). This experiment resulted in 175 shots, each stemmed with 2 asbestos tamping plugs. Figure 8 represents shots (about 175) with the same explosive but using tamped, unstemmed charges. The results of the next series, with seven composition variations of the lower density and velocity nongelatins (brand Y), are plotted in figure 9. These again represent a total of 175 shots with tamped, unstemmed charges. Finally, figure 10 gives the results of the last experiment, in which four gelatinous formulations (brand Z) were fired with fireclay stemming. Difficulties encountered because these relatively high performance explosives cracked the cannon explain the limited number of shots for the 15- and 20-percent-salt formulations.
The generally upward slope of the lines through the points in all figures indicates a definite effect of added sodium chloride in increasing the W50 value, that is, reducing the incendivity of the explosive. It is interesting to note that this effect is apparent for both stemmed and unstemmed shots despite the low values encountered under the latter condition. No attempt was made to draw a best fitting line through the points in the figures. The 95-percent confidence limits for each W50 value, indicated by vertical lines, permit comparison of any two values obtained in a test series. If these limits do not overlap, their corresponding W50 values must be significantly different. However, overlapping of confidence limits does not necessarily mean that the converse is true. In such instances, one must resort to statistical calculations.
All of the available data, listed in table 5, were analyzed statistically to determine the effect of added sodium chloride on the incendivity of permissible explosives. The table gives 87 W50 values obtained for formulations containing, respectively, 0, 3, 10, 15, and 20 percent added sodium chloride. These data represent approximately 2,400 shots. They include almost all shots made under the three- stemming conditions (1 pound of fireclay stemming, asbestos plugs, and no stemming), with both the nongelatinous and gelatinous formulations, in 2- or 1-¼-inch-diameter cartridges. To permit statistical analysis, the W50 values obtained under the different experimental conditions were
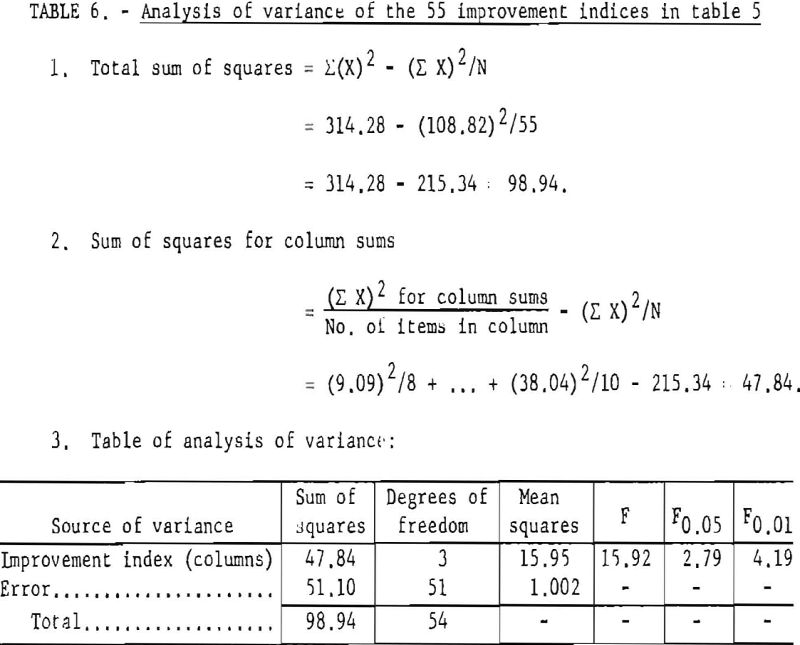
first transformed to ratios designated as improvement indices (table 5). For example, the W50 values for the 0- and 3-percent-salt formulations of explosive No. 1 are 390 and 470 grams, respectively; the corresponding ratio or improvement index is 470/390 or 1.21. Table 6 gives the analysis of variance for these ratios. The analysis shows that the addition of sodium chloride (up to 20 percent) has a highly significant effect in reducing the incendivity of permissible formulations.
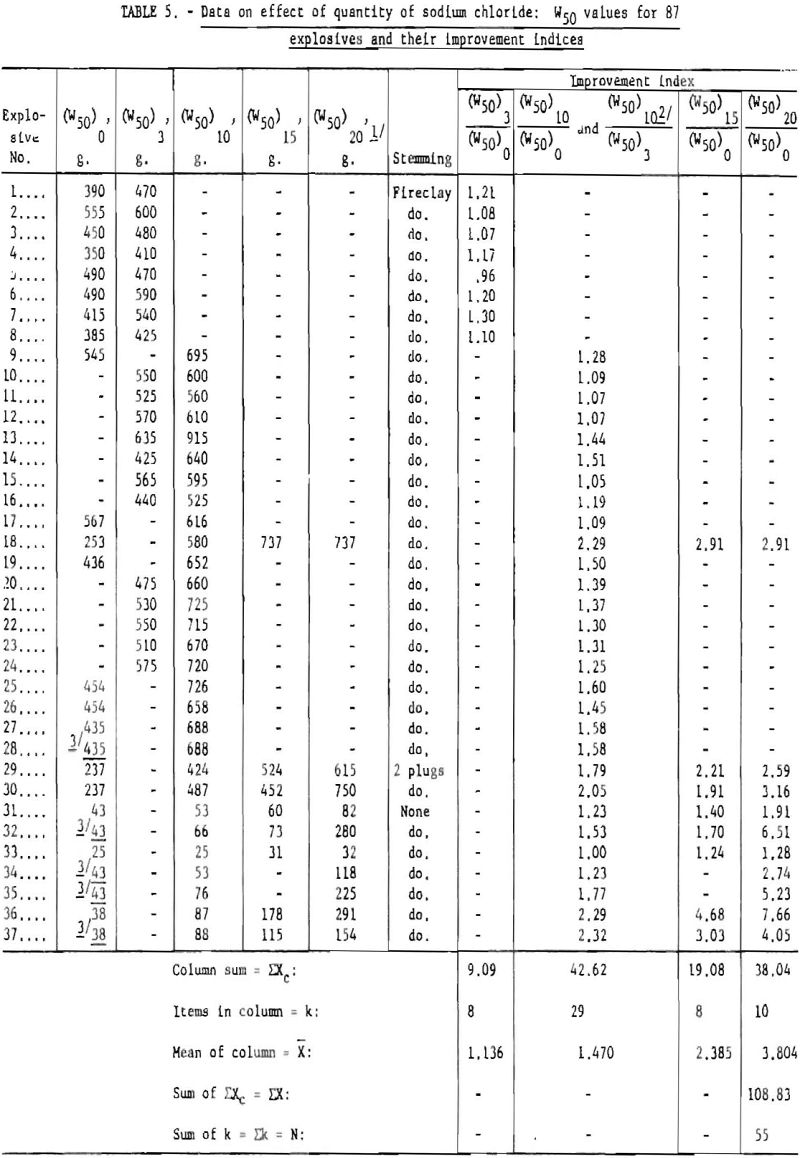
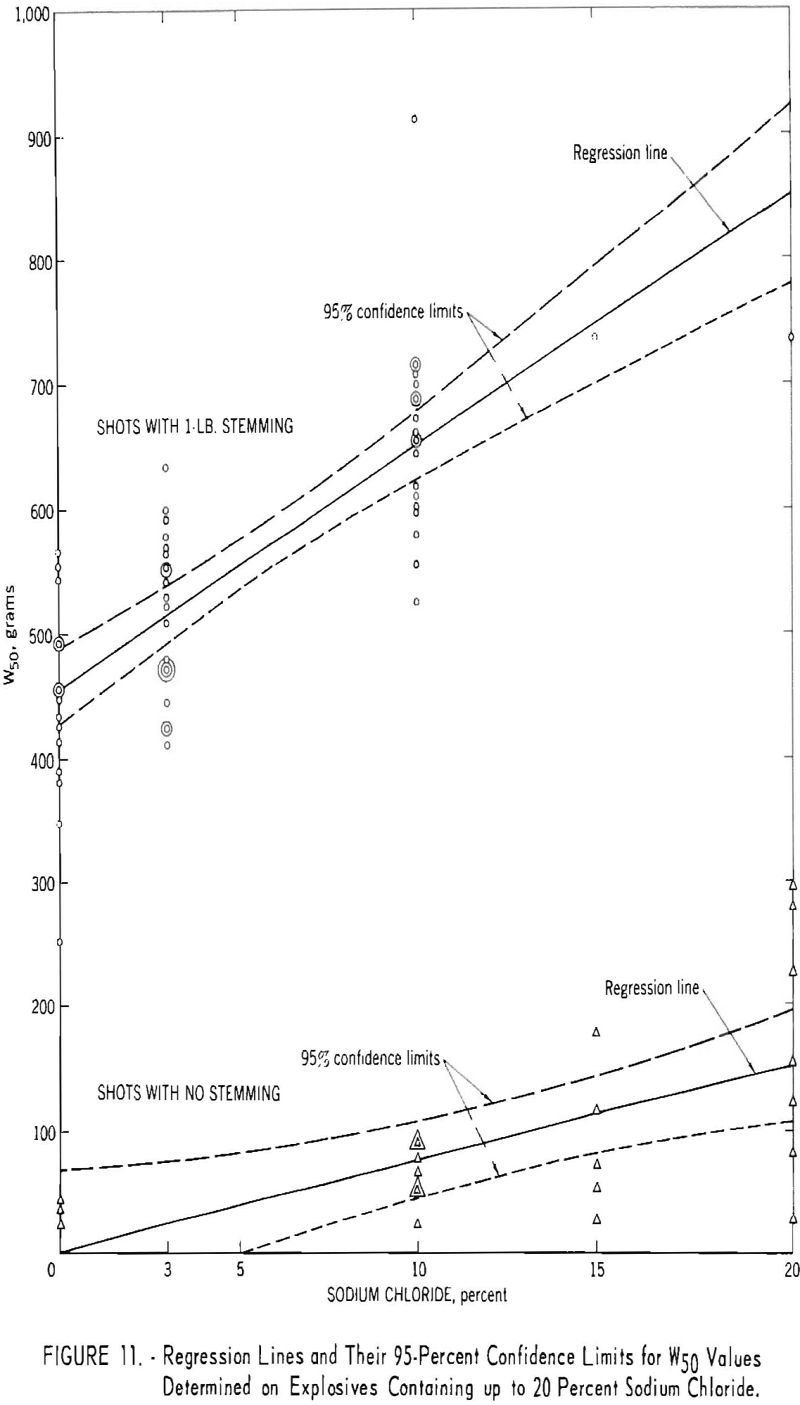
This conclusion is quite general and does not depend upon the type of stemming used or upon the gelatinous or nongelatinous character of the explosive. The results, based on the totality of the data, may be usefully portrayed as two regression lines (fig. 11). The top line represents the shots with 1 pound of fireclay stemming, and the bottom line represents the unstemmed shots. The regression lines show the W50 values expected for a given sodium chloride content, for both the stemmed and the unstemmed shots. Thus, for a formulation with 10 percent salt, similar to those already examined, the best estimate of the expected W50 in stemmed shots is 650 grams. The 95-percent confidence limits are given for each regression line.
The conclusion that added sodium chloride reduces the incendivity of permissible explosives agrees with reports of previous investigators. In particular, Imperial Chemical Industries and Loison and Sartorius have published incendivity versus sodium chloride curves supporting this conclusion
Effect of Particle Size of Sodium Chloride
The three series of experiments designed to evaluate the effect of the size of the sodium chloride particles were randomized separately and performed concurrently with the experiments for determining the effect of the quantity of salt (figs. 7-9). For this purpose, the sodium chloride was divided into two categories–coarse and line. The W50 values obtained in these experiments are generally higher for the fine-salt formulations than for the corresponding coarse-salt formulations, indicating that fine salt is more effective than coarse in reducing the incendivity. Of 13 comparisons made on the basis of the data in table 7, 1 showed no difference between W50 values (Nos. 4 and 5), and in 2 instances (Nos. 25 and 26 and Nos. 9 and 10) negative differences of 1 gram and 72 grams, respectively, were observed. It should be noted, however, that the negative difference of 72 grams was obtained with the X formulations; as mentioned earlier, the fine salt used in this sample had a somewhat larger particle size than that used in the Y formulations.
In preparation for a statistical analysis based on all pertinent data, improvement indices (I.I.) were calculated for the W50 values in table 7, as follows :
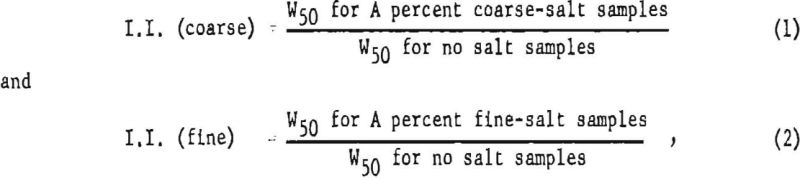
where A may be 10, 15, or 20 percent and represents the sodium chloride concentrations in the formulations studied. As some experiments were performed with stemming and others without stemming, these improvement indices were necessary so that the fine- and coarse-salt values could be compared on a common basis. All improvement indices for the fine- and coarse-salt formulations were grouped accordingly in the table, giving two groups of ratios that were considered as samples of two populations of interest.
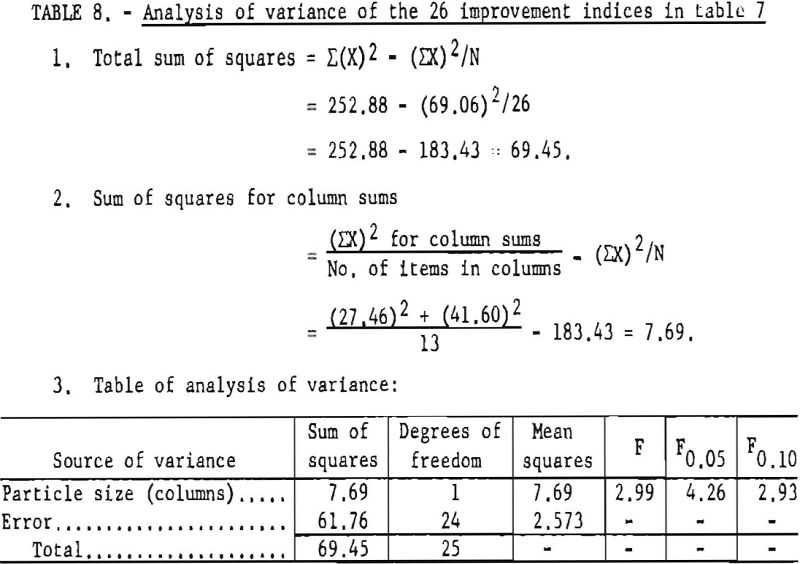
The analysis of variance for these data, presented in table 8, allows the conclusion that the fine salt reduced the incendivity of explosives significantly more than the coarse salt at the 90-percent confidence level. An analysis of the same data by means of the Student “t” test showed that the effect of particle size of sodium chloride is significant at the 95-percent confidence level. On the basis of a limited number of tests, J. E. Dolan has generalized that the particle-size effect of sodium chloride may be directly related to its surface area. If this conclusion is correct, the fine-salt variations should give significantly higher W50’s. The tentative nature of the conclusion derived from the analysis of variance may be explained by the extremely small number of tests considered. In such instances, slight variations in the data can alter the conclusions appreciably.
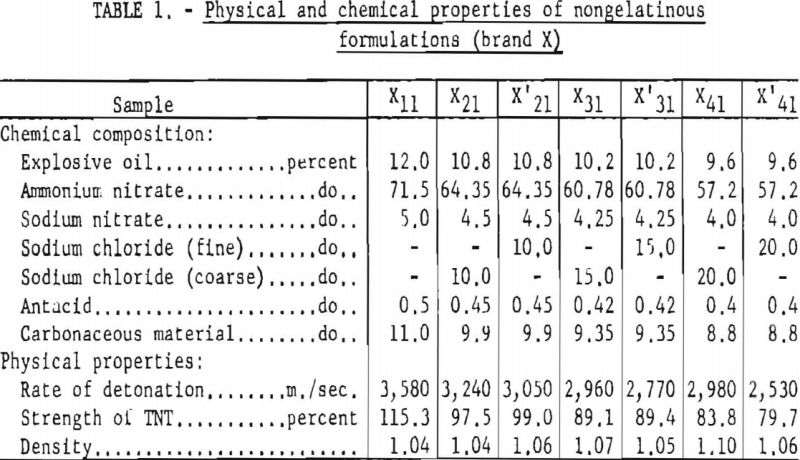
A further analysis of the data showed that the difference in the effects of coarse and fine salt was more marked for unstemmed than for stemmed shots. This is apparent from the ratios of W50 values obtained using the two types of salt granulation (table 9). The average ratios for the stemmed and unstemmed data were 1.07 and 1.70, respectively. A statistical analysis of the ratios in table 9, using the Student “t” test, showed that there is a significant difference between the stemmed and unstemmed tests at the 95-percent confidence level. Two plausible explanations for this difference are that in stemmed shots (1) the explosive is more confined, giving the salts a longer time interval in which to decompose more completely, regardless of their particle size, and (2) in addition to the average 70 grams of sodium chloride in each explosive charge, 1 pound of fireclay is present as stemming. As the salt represents about 15 percent of the total inerts, the other 85 percent being fire-clay, variations in the salt content of unstemmed shots should be more evident.