During the past year there have been a number of significant investigations that have furnished evidence on the driving forces governing grain growth and on the role played by boundary impurities.
Thus Burke has shown that in high-purity alpha brass the rate of grain growth in the earliest stages is approximately inversely proportional to the existing grain size. This relation agrees with one derived by Zener assuming that surface energy was the driving force in grain growth. Also in confirmation of the surface energy hypothesis, Burke found that growth virtually stopped when the grain boundaries or even only the grain corners were melted.
Transformations
Quite a number of contributions have been made concerning the nature of the martensite transformation, perhaps the least understood of all solid-state reactions.
Two sets of investigations have shown that certain transformations in pure metals exhibit the most important features of the martensite reaction, i.e., transformation only during cooling, and stabilization. Barrett and Trautz have observed such a transformation in lithium at low temperatures. Partial transformation to a close-packed structure, accompanied by audible clicks, was found to take place below dry-ice temperature. Low-temperature cold work caused additional transformation. Troiano and Tokich found the transformation of cobalt to occur during cooling, with the characteristics of a martensite transformation.
Generally the martensite reaction has been thought of as a shear-type transformation not involving a nucleation process. However, Fisher, Hollomon, and Turnbull have advanced the concept that martensite-type reactions are nucleated by embryos of the segregating phase retained in quenching from a high temperature. These embryos are promoted to nuclei (“athermal” nucleation) upon cooling, since the critical size of a nucleus decreases with decreasing temperature. Stabilization takes place because the concentration of subcritical embryos tends to decrease upon holding at a temperature far below the transformation temperature. They also suggested that martensite grows by rapid atom-by-atom diffusion across a coherent interface rather than by a semimacroscopic shear mechanism.
Kurdjumov has also made the suggestion that martensite forms by nucleation and growth, although he appears to believe that the nucleation takes place isothermally (“thermal” nucleation). Rather striking support for his growth mechanism has been obtained by Kurdjumov and Maksimova. They report that direct quenching of 6% Mn, 0.6% C steel in liquid nitrogen at —194C retains 100% austenite. More important, upon warming to —159C or above, a fraction of the austenite transforms isothermally to martensite (transformation continues for more than three hours at —159C). The limiting fraction transformed decreases with increasing temperature, falling to zero at Ms. The duration of the isothermal transformation period also decreases as the temperature rises. Similar effects were found for a 1.6% carbon steel. Although 20% martensite was formed during cooling to —194C, the 1.6% carbon steel continued to transform slowly at this temperature, and transformed more rapidly upon heating.
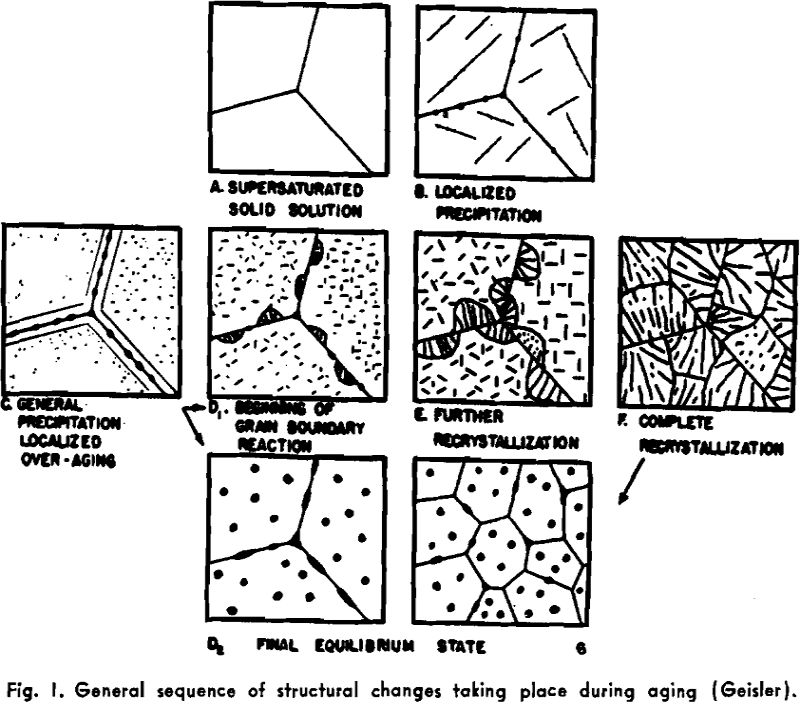
Age-Hardening
Geisler has presented an orderly correlation of the property changes and structural changes that take place during aging. It is shown that, qualitatively at least, it is possible to explain all the observed features of aging curves for various properties on the basis of simultaneous effects of the several structural changes that are known to occur. The general sequence of structural changes may take either of two courses, depending on whether or not recrystallization occurs at the grain boundaries (“discontinuous precipitation”).
Geisler and Newkirk have applied the type of analysis suggested above to an investigation of the sequence of structural and property changes during aging of a permanent-magnet alloy, Cunico. Changes in hardness, electrical resistance, and magnetic properties were interpreted in terms of attainment and loss of coherency, the grain-boundary recrystallization reaction, and depletion of the solute concentration in the matrix.
Diffusion
A number of investigations have been completed in which “uphill” diffusion was measured. One of the most interesting of these was carried out by V. Daniel on the alloy Cu4 Fe Ni3. Below 800C, this alloy breaks up into two face-centered cubic phases having the same proportions of iron and nickel but different copper contents. When the alloy is quenched into the two-phase region, the X-ray powder diagrams may be interpreted in terms of a periodic variation of copper concentration in one dimension. The amplitude of the wave (the maximum copper-concentration difference) changes rapidly with time relative to the change in wave length (the distance separating adjacent copper-concentration maxima) until the equilibrium or maximum amplitude is reached. Then the wave length continues to increase slowly with time. T
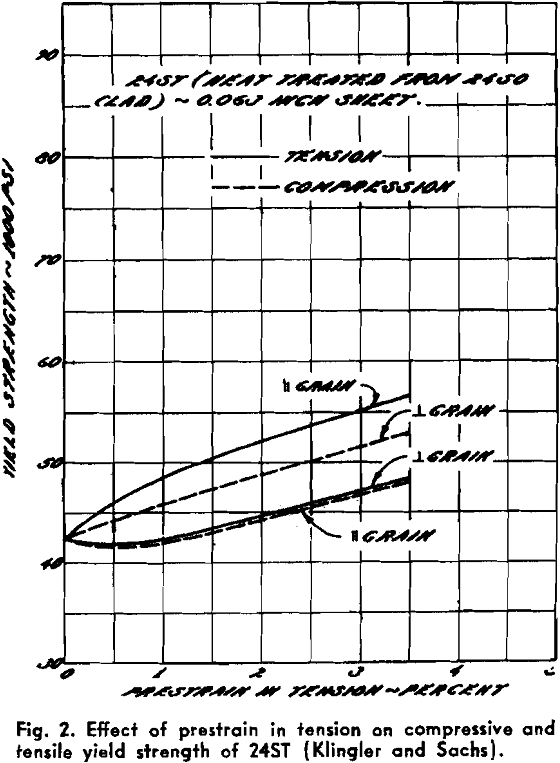
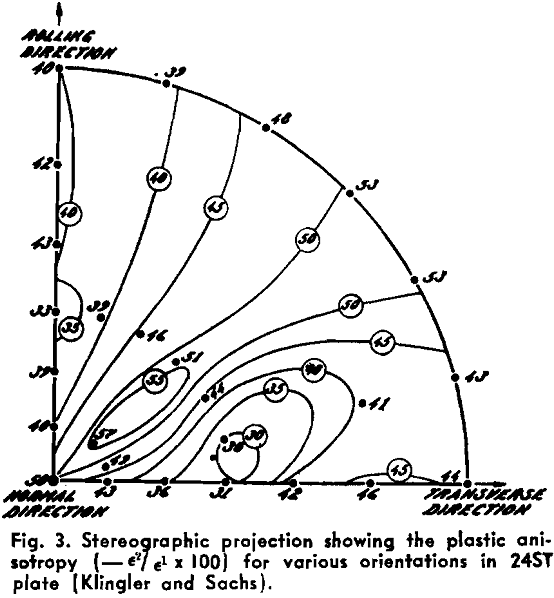
Powder Metallurgy
Shaler and Wulff calculated the rates of shrinkage of copper compacts, assuming a model in which sintering proceeds by the flow of metal under the influence of surface tension, modified by gas pressure. Fair agreement was found between calculated and measured shrinkage rates. The same authors in a note have suggested that the origin of the viscosity in their sintering mechanism arises from preferential self-diffusion of vacancies in the lattice, under the influence of stress.
Schwarzkopf discussed the mechanism of sintering and concluded that plastic flow plays an important role in all stages of the sintering mechanism. He also pointed out that surface diffusion and plastic flow do not take place independently of each other.
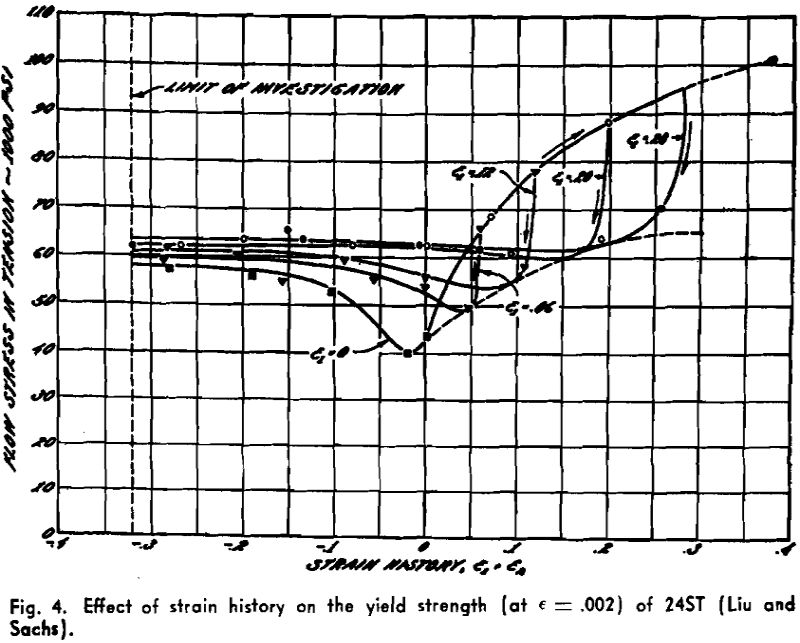
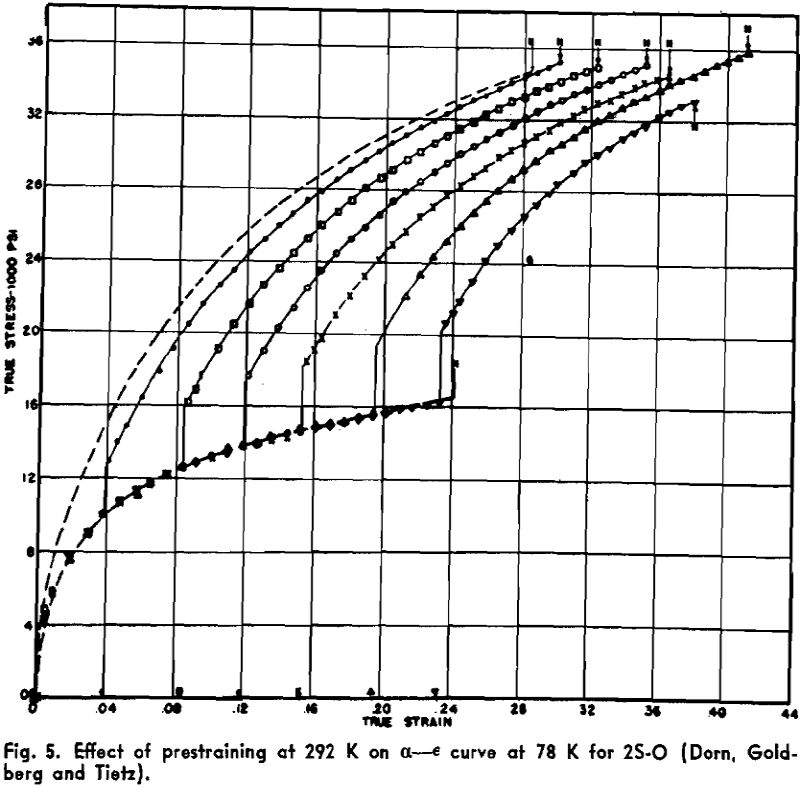
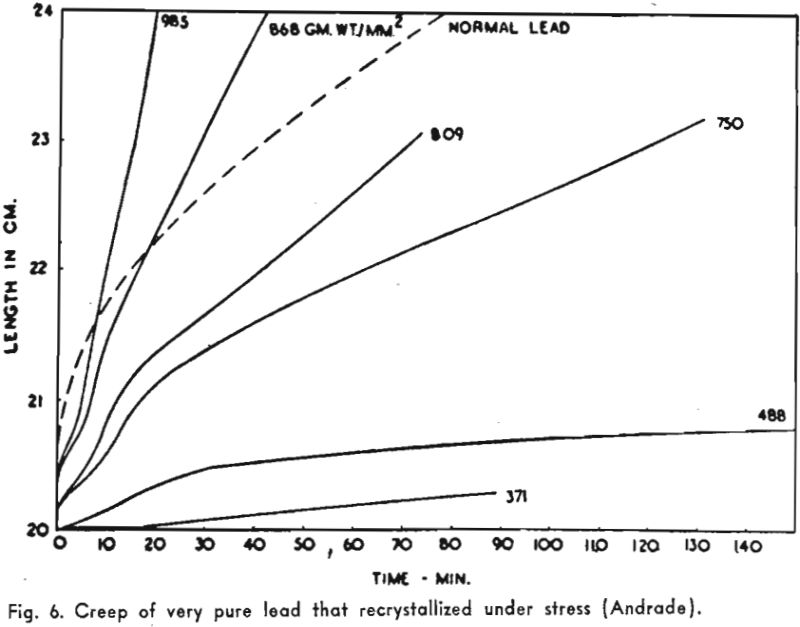
Twinning
W. P. Mason, H. J. McSkinnin, and W. Schockley found that during the formation of a twin there is an oscillation of the loaded surface having an amplitude of about ½ to 2 A and a period of 1 to 3 microseconds. This was interpreted as evidence that twinning proceeds by the passage of dislocations through the crystal at the speed of sound.
A. V. Stepanov has attempted a rationalization of the identity of twinning planes on the basis of elastic anisotropy. The results indicate that the criterion may be generally applicable. A. H. Cottrell has explained the yield point as being due to a cloud of solute atoms surrounding a dislocation and preventing its motion until a higher stress is reached than that required for propagation of a free dislocation. If this explanation is correct, discontinuous yielding should occur in single crystals of all materials for which it is observed in poly-crystals.
Discontinuous yielding has, in fact, been observed in cadmium crystals by Cottrell and Gibbons and by several previous workers. However, the lack of consistency in the conditions reported to be necessary for appearance of the phenomenon leads to some doubt about the interpretation.
Cross slip has been observed in brass crystals oriented for single slip according to the classical analysis. The cross slip bands were traces of octahedral planes containing the operating slip direction of the usual slip bands. We considered the cross slip to be compensatory to the tendency for rotation of the crystal under gripping conditions which prevented this rotation. D. C. Pack, W. M. Evans, and H. J. James have investigated theoretically the velocity of propagation of shock waves in steel and lead.
For iron, the calculated velocity of plane elastic waves is higher than that of shock waves, while in lead the reverse is true. The results were confirmed experimentally. In lead, the shock wave is damped, causing its velocity to decrease to less than that of plane elastic waves within a few cm of the source.
Fracture
Zener has presented evidence indicating that the stress concentration necessary to initiate fracture arises from slip bands stopped by grain boundaries, precipitate particles, and the like. Thus the locus of fracture stress has no meaning in the absence of deformation. In view of the large volume of literature based on both the statistical concept and the fracture-stress curve concept, such conclusions are significant and controversial.
The effect of stress state on the fracture of metals was discussed by J. E. Dorn in terms of a fracture-stress curve. He presented data on the fracture of aluminum and magnesium alloys under biaxial stresses. These results approximated the predictions of the maximum shear-stress law.
Corrosion
Polarography has been adapted to the study of corrosion by Van Ryssel-berghe and collaborators. A small sheet of metal is placed in the cell, with no contact with the electrodes. Corrosion by various substances, such as oxygen, hydrogen peroxide, acids, etc., is detected by measuring their reduction currents in solutions of electrolytes. As these reducible substances are consumed by the corroding metal, the dissolution of the metal may also be followed, to a certain extent, by the polarographic reduction current of the metallic ions.
Hickman and Gulbransen have continued their electron diffraction studies on the structure of oxide films formed on various alloys. They report that there is no unique structural change in the oxide film formed on copper-nickel alloys at the composition where they become passive to corrosion. This suggested that the mechanism of gas-phase oxidation and corrosion are basically different. They have also studied the rates of oxidation of a series of nickel-chromium alloys by means of the vacuum microbalance. They report that the 80% nickel-20% chromium series appears most promising from the standpoint of oxidation resistance. Silicon additions further improve this property.

