In reverberatory smelting, fuel is the chief item of expense, as it commonly is in processes using large percentages of it. Hence the most suitable supply is eagerly sought; that is, the supply which, in the end, yields the greatest net profit. The better grades are not infrequently bought, even at an advanced price, because they are apt to be decidedly more economical. High price, of itself, however, is no merit, for freight is generally the largest part of the cost of fuel, and the nearby supply may yield as high efficiency as that from a greater distance. If a poorer grade is reasonably good, and if its cost is low enough to more than offset the decreased technical efficiency, it is of course the more suitable.
Any smelting operation depends for both efficiency and economy on raising the furnace atmosphere considerably above the “critical” temperature; that is, above the temperature at which smelting is completed. If this temperature is not exceeded, smelting stops, no matter how much coal is burned; and the real value of a fuel depends on the number of degrees that the temperature is continuously kept above this point. For example, if one fuel maintains 2,500° F. and another only 2,100° F., while the “critical” temperature is 2,000° F., the first is worth at least five times as much as the second, although the increase above atmospheric temperature is only one-fifth more in one case than in the other. This “margin of temperature” above the smelting point is much more significant than the total number of heat units developed from the fuel. The case is parallel to that of water flowing from a pipe. The reservoir may contain a billion gallons of water; but, even though the fluid has come to the nozzle, not a drop will run out, where it can be of practical use, unless the level in the reservoir is higher than the nozzle; and the speed and volume of flow depend on how much higher the level is in the reservoir than at this point. If the temperature in a furnace is not above the critical level there will be no slag to flow over the skimming plate, even though there are enough heat units to bring an unlimited quantity of charge up to the point of slag formation; and just as the value of a water supply for the purpose of putting out a fire depends largely on the effective head, so the difficulties of furnace management vary inversely with the margin of temperature, provided other conditions are alike in the different cases. The most important of these other conditions is the presence of a good flame in the hearth, which is referred to in the discussion of several of the fuel tests and is taken up in some detail under “Character of the True Coal” .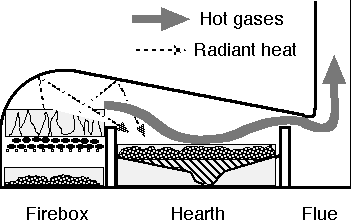
The heat must be applied at the right place, and the right place in a reverberatory furnace is the charge on the hearth. Some experiences are here presented showing the unfortunate condition, with some coals, of getting an excessive proportion of the heat into the flue or on the grate, and the high cost, in some cases, of getting the charge above the critical temperature.
Experiences With Various Fuels
At its old reduction works, the Anaconda Copper Mining Co. used wood for reverberatory smelting during a short period in its early history; but this fuel was by no means as efficient as fairly good coal; nor was it as economical, even though it was cut within 5 or 10 miles of the plant, while the coal had to stand the freight charge for a haul of 200 miles in one case and 500 in another.
Coal from Belt, Mont., was tried; but in a 20 by 12 ft. furnace with a 4 by 5 ft. fire box this could not be successfully used with natural draft, for four reasons: 1, there was a large loss of coal through the grate because the ash did not clinker, and the draft was only enough for a comparatively thin bed on the grate; 2, it contained too little of the heavy hydrocarbons to maintain a proper flame in the hearth; 3, its total heating value was too small to develop a very high temperature under any circumstances; and 4, the large amount of ash (25 to 30 per cent.), intimately mixed with the true coal, so interfered with efficient combustion as to still further increase the difficulty of getting a high temperature. With forced draft, however, and with an 8 by 5 ft. fire box, enough flame was driven into the hearth so that, moderate smelting conditions were obtained; and it was possible to carry such a depth of ash that the loss of fuel through the grate was much reduced. The intense forced-draft combustion at the bottom of the fuel bed, however, caused troublesome clinkering. The extent of this will be appreciated when it is stated that, although steam was introduced with the blast to lessen the clinker, so much of the latter built up that the entire grate had to be dumped and a new fire started every 12 hr. After using Belt coal for a year, a coal from Diamondville, Wyo., was adopted, and was successfully used with natural draft. The change resulted in a 40 per cent, decrease of the fuel cost per ton of material smelted; and it increased the capacity of the furnaces 66 per cent., thus leading to a decrease in the other items of cost per ton. This was a case where a very decided increase in the price of coal resulted in a remarkable net saving.
The size of furnaces was gradually increased at the old works till hearths 42 by 14 ft. were built, which resulted in striking economies of fuel, labor, and repairs. This increase continued at the new (Washoe) works with even greater advantage, as already recorded in the Transactions, and the benefits of this enlargement were so marked that the management determined to find whether; the low-priced Belt coal could be successfully used with strong natural draft (1.6 in. water column in the flue close to the furnace and 0.75 in. at the bridge) in the fire box 7 by 16 ft. by 24 in. deep attached to a 112 by 19 ft. hearth, which are the dimensions of most of the present furnaces. The trial was made in a furnace that was already in normal operation with Diamondville coal; but after the Diamondville coal was all burned out only one 15-ton charge could be smelted, and even this could not have been done except for the reserve heat in the immense pool of slag and matte (180 tons combined) that is kept in the hearth. The furnace froze an hour before the end of the test, though using Belt coal at the rate of 98 tons per 24 hr. After getting back to Diamondville coal, it took 7 hr. to get the charge hot enough, in front, to skim; though it was hot enough at the back to take a charge in 3 hr. The usual rate of smelting was regained in about 8 hr. Before the test began, the flue temperature averaged 2,200° F., as usual; for 4 hr., while the fire box contained both Diamondville and Belt coal, it was 2,000° F.; but for the last 2½ hr., with Belt coal alone, it was only 1,660° F. During the first 7 hr., with a good clinker bed, and with considerable heat from Diamondville coal, the boilers attached to the furnace developed 584 boiler horse power; but during the last 8 hr., with Belt alone, they developed only 389 h.p. This still left Belt coal with its bad reputation; but several years later, when on account of inherent losses in gas producers the management of the company’s Great Falls plant pretty definitely decided to abandon their producers, which were situated at a considerable distance from the furnaces, it was thought that this coal might be a commercial success at Great Falls, in a gas-producing fire box attached directly to the furnace, the freight being much less from Belt to Great Falls than to Anaconda, while the freight on Diamondville coal is higher to Great Falls than to Anaconda. They had always used, in their gas producers, coal from near Belt, and of the same general character as the Belt coal. Since their regenerative furnaces, designed like an open-hearth steel furnace, were not so well adapted to such an experiment as the furnaces at Anaconda, a 102 by 19 ft. hearth at the latter plant was equipped with a fire box that had the immense shaking-grate area of 19 by 21 ft. Under these conditions smelting succeeded even with natural draft, as shown in trials 1 and 3 of Table I. The surface of the fuel was kept 5½ ft. above the grate, this being the best depth found by experience. In the first trial nothing but roasted concentrate (“calcine”), containing a small amount of lime rock flux, was smelted; but in No. 3 there was added a portion of flue dust, which is harder to smelt than calcine. In parallel runs with Diamondville coal in one of the regular furnaces (trials 2 and 4 in the table) the results were normal when smelting calcine alone, but 8 per cent, better than normal when flue dust was added to the charge. The comparison of coals was still decidedly in favor of Diamondville normal results, the tons smelted per ton of coal being a good deal more than double, the cost for coal being 36 per cent, less per ton of material smelted, and the tonnage smelted per furnace being about 20 per cent, greater. Moreover, Diamondville requires only three men per furnace on each 8-hr. shift, while the Belt required six men at best. Most of the extra labor was due to the troublesome ash and clinker.
With forced draft in the gas-producing fire box, and carrying the surface of the fuel bed 7 ft. above the grate, the consumption of Belt coal was 19 per cent, less than with natural draft, while the smelting capacity was increased 12 per cent., thus increasing the ratio between tons of material smelted and tons of coal used from 1.99 to 2.74 (compare trial 5 with trial 1 in Table I). However, even this was much short of Diamondville results, which yielded 9 per cent, less cost for fuel, per ton smelted, and 8 per cent, more smelting capacity for the furnace (compare trials 2 and 5); and there was, with Belt coal, the decidedly increased labor cost, a much larger capital outlay for equipment, the power to supply blast, and a large consumption of steam under the grate to prevent clinkering. The power used for forced draft was 23.1 h.p., and 11.5 h.p. for the secondary air introduced over the bridge. The steam used under the grate was 4,600 lb. per hour (143 boiler horse power). Even with natural draft there was a steam consumption of 2,220 lb. per hour (69 boiler horsepower). The steam generated in the boilers attached to the Belt furnace was 532 boiler horse power with forced draft, and 619 with natural draft. In trial No. 4, with Diamondville coal and natural draft, the horse power developed was 469.
The total cost under these best conditions for Belt coal would be at least 15 per cent, more than with Diamondville at Anaconda. Possibly some other style of gas producer would be better; but it is not likely that Belt coal can ever compete with good Diamondville here, unless by some very radical change in conditions. At Great Falls, however, with the very different freights from the two coal camps, Belt may be the more economical coal.
Rock Springs (Wyoming) coal was used at the old works for several years; but was not as good as Diamondville, because it flashed too much

in the fire; that is, it gave off its volatile matter so rapidly that there was a good flame in the furnace for a few minutes, but for most of the time there was practically no flame. To minimize this effect, the fire-box temperature was kept as low as possible, by letting a thick bed of ash accumulate. This procedure also prevented much loss of fuel through the grate; but, as the ash was fine and did not clinker, the grating had to be done cautiously. A furnace would use about 25 per cent, more of Rock Springs than of Diamondville coal, but would not smelt more than 90 per cent, as much material.
A number of trials have been made at the new works with mine-run coal from Bear Creek, Mont. The first shipment was received before the mines were much developed; and the natural-draft results were like those with Belt. (The Belt mines, however, were well developed). With the coal 24 in. deep in the 7 by 16 ft. fire box the furnace froze in 6 or 8 hr., despite the best efforts with different methods of firing, and while using coal at the rate of 72 tons in 24 hr. The cause of this failure was lack of flame. This same shipment of coal succeeded, however, under forced draft, even though in one case it was used at a slower rate than during the natural-draft trial; and this was due to the intense combustion on the grate distilling some heavy volatile matter into the hearth, and developing flame there. This is parallel to the experience with Belt coal. Having the Bear Creek coal 38 in. deep, with an average air pressure of 2.6 in. under the grate, 118 tons of calcine were smelted per 24 hr., with 65 tons of coal (ratio 1.8); but with a fuel bed only 24 in. deep, and a 1.7 in. under-grate pressure, the decidedly better result was obtained of smelting 180 tons of calcine with 80 tons of coal (ratio 2.2). (See trials 6 and 7 in Table I.) A parallel run with 61 tons of Diamondville coal smelted 267 tons of calcine and flue dust (ratio 4.4), a normal result with this coal. (See trial 8.) The fuel cost for even the better of the Bear Creek results was 75 per cent, higher, per ton smelted, than for the Diamondville result, and would be nearly 100 per cent, higher if flue dust had been included in the charge, as it was with Diamondville, because of the slower smelting of dust.
Even with forced draft a really good flame could not be obtained from this lot of Bear Creek coal; but after 15 months further development of the mines the coal was of a quality to give fairly good flame without forced draft. The efficiency, however, was 25 per cent, better with forced than with natural draft, as indicated by the ratio and “efficiency” columns for trials 9 and 10 in Table I. Trial 10 was the most efficient and economical of all the tests of Bear Creek coal, but its fuel cost per ton smelted was 14 per cent, higher than for Diamondville in a parallel test (trial 11), and 30 per cent, higher than for normal Diamondville results when the charge, as in trial 10, contains no flue dust. Another large shipment, six months later, was not as good as the one just mentioned (see trials 12, 14, 16, and 18 of the table.)
Another Montana mine-run coal, from Roundup, gave results which, as a whole, were about the same as with the Bear Creek (see trials 20 to 26), but the Roundup netted 5 to 10 per cent, less cost per ton smelted, because of a lower freight rate. The cost, however, averaged nearly 70 per cent, more with Roundup than with Diamondville coal, although the price of a ton of Roundup was one-sixth less than for Diamondville.
What Constitutes Good Fuel
So far as the coal is concerned, there are four factors that affect smelting efficiency: 1, the character of the true coal substance; 2, the percentage of fines; 3, the percentage of moisture; and 4, the character and percentage of ash; and these call for some discussion.
Character of the True Coal
As to the character of the real coal substance, something can be judged from proximate analyses; but these must be used with a good deal of caution, for they may be very misleading in regard to the ratio of fixed carbon to the volatile portion of the real coal, and they tell nothing as to the composition and qualities of the volatile matter. Frank Haas has shown that the two coals whose analyses are quoted below are undoubtedly identical in the character of the coal substance, though the proximate analyses suggest quite otherwise when considered in the usual way. They are from different parts of the same bed. His conclusion is based on intimate practical familiarity with the coals; on a study of the ultimate analyses; on consideration of the sulphur included as part of the volatile matter; and on changes in the ash which affect the apparent percentages of volatile matter and fixed carbon.

Mr. Haas’s proximate analyses appear to have been made with a good deal of care; but with less care the results are still more misleading. The analyses in Table II for our trials 9, 10, and 11 illustrate what happens when the conditions, as to time and temperature, are not according to the standard directions for determining volatile matter. The ratio of volatile matter to fixed carbon, in these three cases, is respectively 1.01, 1.06, and 1.01; but in no other case in the table is the ratio higher than 0.89 for either of these coals. These three analyses were made together; and the fact that the ratio is equally high for both Bear Creek and Diamondville is practically conclusive evidence that the samples were heated too much in the determination of volatile matter. The errors were not appreciated till it was too late to make checks.
Even the moisture determination may lead to errors in the apparent percentage of volatile matter; P. L. Teed shows that in the standard method for moisture the loss from some coals amounts to 2 per cent, more than the true moisture.
We know that while the volatile matter contains hydrocarbons, H and CO (combustibles), it also contains N, CO2 and H2O (non-combustibles), this H2O having evidently been combined as part of the coal; and that the relative quantities of these different substances differ greatly from coal to coal. Porter and Ovitz found the non-combustible gases from several coals (not including mechanically absorbed moisture), all treated in a similar way, varied from 1 per cent, to at least 15 per cent, of the total coal. The ultimate analyses made by the government bureaus show oxygen content of unweathered bituminous coals, after reducing to an ash and moisture free basis, ranging from 5 to 17 per cent. ; and in samples taken near the outcrop, where weathering has occurred, this figure runs as high as 27 percent. This oxygen not only displaces, so to speak, an equal amount of carbon and hydrogen, but also renders useless, for heat production, a considerable part of the carbon and hydrogen that are present, by being already combined with them. David White has shown, by examination of a very large amount of data; that each per cent, of oxygen has the same effect as 1 per cent, of ash in reducing the heating value shown by a calorimeter. The commercial value is reduced in still greater measure, because the gas resulting from this oxygen has to be heated to the furnace temperature at the expense of the heat-producing portion of the coal, thus still further reducing the available heat and the margin above the critical temperature.

In most bituminous coal there is about 5 per cent, of hydrogen (on the moisture-free basis), which of course burns to water, and this water must be considered when interpreting reports on the heating value. Laboratory determinations of heating value are made with this water condensed to liquid; but in actual use it passes away as gas, and the error in the reported heat units amounts to about 500 B.t.u. per pound of dry coal.
Manufacturers of illuminating gas find that the relative quantities of the different hydrocarbon gases and of the different tar-like substances volatilized from coal vary greatly at different temperatures with any particular coal, as well as from one coal to another: moderate temperatures yielding large quantities of the heavier products, while very high temperatures yield more of the lighter products. The lighter gases burn with a clear blue flame, while the heavier products burn with a more or less opaque yellow or white flame due to the incandescence of solid particles. All experience shows that this latter kind of flame is one of the chief essentials for successful reverberatory work, a very large part of the heat utilized by the charge being received by direct radiation from these highly incandescent particles. The trouble with a clear flame is that comparatively little of the hot gas comes in contact with the charge, to permit the direct absorption of heat; and not much of it comes in contact with the roof and walls, and so they receive comparatively little heat to radiate on to the charge; but the suspended incandescent particles have an immense surface per pound, from which they radiate a good deal of their heat to the charge. They also undoubtedly absorb some of the heat developed from the lighter gases and then radiate that to the charge also. With a good flame, the total surface of the incandescent particles in the furnace at any moment is far in excess of the entire surface of the roof and walls. The effect of this is like increasing the flow from a water reservoir, when a fire is to be extinguished, by multiplying the number of streams, and thereby greatly increasing the efficiency of action. This statement supplements the argument in regards to the “margin of temperature” on pp. 1847 and 1848. Two cases have already been cited in which a lack of incandescent flame was largely responsible for complete failure to do any smelting (Belt and Bear Creek coals). Another striking experience with this principle was had with the Bruckner roasters at the old works, in which Belt coal gave very little flame at the fire-box end, and none at all at the other end even with forced draft. In order to get results it was necessary to drive the fire hard, which produced such a high temperature at the fire-box end that there was more or less trouble from sintering, while the charge never got hot enough at the other end to roast satisfactorily. With Diamondville coal this trouble did not exist. It should be noted that an excessively dense (smoky) flame is to be avoided; for two reasons: first, there is increased difficulty in supplying and mixing enough air for combustion; and second, it is almost certain that, in such a dense cloud, the heat radiated from most of the incandescent particles is obstructed and the charge not efficiently heated.
In order to get this luminous flame, even with good coal, a fairly thick fuel bed must be maintained on the grate; so that the condition is approximated in which just enough air comes through the grate to burn the fixed carbon to CO, while the rest of the air is admitted through adjustable checker holes above the bridge wall. The large hearth forms nearly the ideal combustion chamber demanded by these conditions. In boiler practice it is rather unusual to have more than an 8-in. fuel bed; but in large reverberatories with 1½-in. draft in the flue a 24-in. bed is more or less standard for good coal containing only a moderate percentage of fines. With coals that do not flame well under natural draft there is often a marked improvement by using forced draft. The latter so increases the temperature in the fire box as to drive off more volatile matter. This is illustrated by the experiences with Belt, Bear Creek, and Roundup coals, all of which did better smelting with forced than with natural draft.
Percentage of Fines
In ordinary boiler practice, the equipment can be so designed and used as to get reasonably good efficiency with any coal that is fit to be so called, although good coal is much more satisfactory than poor, and is likely to be more economical; but in reverberatory smelting there are much narrower limitations. The chemical properties have been indicated as the most important of these; but experience proves that the range of size is also very important. On account of conditions that the Anaconda Co. could not control, the quality of coal received from Diamondville became worse several years ago, the chief difficulty being that it was necessary to mine a soft coal, which crumbles badly and yields a large percentage of fines. Our standard furnace results, with only a moderate proportion of fines, consist in burning 60 tons of coal per 24 hr. and smelting 270 tons of charge (ratio 4.5); 80 to 85 per cent, of the charge being hot calcine, and the rest flue dust, which is harder to smelt than calcine. The poor coal dropped the results to 240 tons smelted with 60 tons of coal (ratio 4.0). The coal at this time contained only about 40 per cent, coarser than ¾ in. as against 60 to 75 per cent, coarser than this size when getting normal results. A test of Diamondville lump, which probably contained 80 per cent, or more coarser than ¾ in., smelted 328 tons of calcine and flue dust with 63 tons of coal (ratio 5.2). (Trial 30 in Table I). Comparing ratios, this lump coal yielded 30 per cent, better results than the poor mine run, and 15 per cent. better than good mine run. Another test, with one-third Bear Creek lump and two-thirds of the poor Diamondville mine run, smelted 248 tons with 62 tons of the mixed coal (ratio 4.0) ; but a parallel test with the Diamondville alone smelted 254 tons with 59 tons of coal (ratio 4.3). (See trials 28 and 29.) The best result in three days’ test of Roundup lump was 263 tons of calcine (no flue dust) smelted with 79 tons of coal (ratio 3.3). (Trial 31 C.) Comparing the ratio and “efficiency” columns of trials 20 and 31 C, the Roundup lump gave 40 per cent, better results than Roundup run of mine under similar conditions.
The three sets of results under trial 31 in the table show the need for the furnace men to learn by experience how best to handle each coal: there was a marked improvement each succeeding day, but we considered that the third day’s result was the best that could be had with this coal. The shipment was not large enough for more than the three days’ run; but in the other trials here recorded the preliminary experimenting was done before official testing was undertaken.
Neither the Bear Creek lump nor the Roundup lump is as economical as Diamondville run of mine. The Diamondville lump, however, is more economical; but, unfortunately, it cannot be supplied in sufficient quantity.
It may be stated that no coal larger than 7 or 8 in. ever goes into the fire box.
A coal sized anything like as closely as the regular commercial sizes of anthracite would probably give very poor results, because there would be such a free passage of air through the mass that the volatile matter would largely burn before it reached the hearth.
The reduced efficiency and increased cost resulting from a large percentage of fine coal are due to three considerations. First, the fresh supply of fine coal temporarily smothers the fire, thereby lessening the amount of air that can be drawn through; while the greater surface exposed per pound of fine than per pound of lump coal permits a sudden evolution of more volatile matter than can be burned by the available air supply, so that considerable fuel passes into the flue unconsumed; and to adjust the air supply through the checker work above the bridge is quite impracticable. Second, it is next to impossible to prevent a large loss of the fine coal through the grate. This loss at Anaconda is 25 to 30 per cent, of the coal when there is the excessive percentage of fines or an insufficient clinker bed, but only 12 or 15 per cent, under normally favorable conditions. Third, this dropping of fines is likely to leave large holes in the fuel bed for some time; but, as this condition does not occur till later than the excessive distillation of volatile matter, a good deal of unnecessary air enters and lowers the furnace temperature.
We are satisfied that the best solution of this question of fines is to pulverize all the coal and burn it by blowing it directly into the hearth of the furnace. Used in this way, all of the fixed carbon, and even the ash, as well as the heavy volatile hydrocarbons, supply incandescent surfaces for radiation of heat to the charge; there is absolutely no ash-pit loss; and the conditions are so uniform that the air supply can be adjusted to a nicety, with the assurance that there will be no decided shortage of air at one time and a considerable excess soon after. Moreover, there will be used only the minimum excess of air, so that a much greater “ margin of temperature ” will be attained. The extremely successful results of this method at the Canadian Copper Co.’s plant, together with the results at several steel plants, are so encouraging that the Anaconda Co. is preparing to give it a thorough trial. The conditions necessary to success are much better understood than they were a few years ago, and we expect that there will be no commercially insurmountable difficulties.
Moisture
This is an impurity, which nobody wishes to pay for, and which requires heat to evaporate. Six per cent, of moisture in a pound of coal absorbs 120 B.t.u. in evaporating and being raised to the temperature of the reverberatory hearth; and in the case of Diamondville coal, burned in our large furnaces, reduces the temperature to 2,800° F. when it would otherwise be 2,825° F.; a reduction of the margin above the critical temperature that probably reduces the capacity and economy by at least 3 per cent.
Ash and Clinker
Ash, like moisture, reduces the heat units per pound of coal, and yet it has a distinct value: with a great many coals the loss through the grate would be excessive if there was not a bed of clinker. In some cases, however, the ash clinkers so badly as to seriously clog the grate and interfere with combustion. This was the case with Diamondville coal when it was used under forced draft in 50-ft. furnaces at Anaconda. The average treatment of calcine, flue dust, and added lime rock, with forced draft, was 104 tons per 24 hr., while using 44 tons of coal (ratio 2.4); but with natural draft it was 144 tons of similar charge, using 46 tons of coal (ratio 3.1); an increase of 30 per cent, in efficiency. A little of this increase was due to certain changes in furnace design; but, as intimated, most of it resulted from the adoption of natural draft. The most important change in design was, in fact, a 58 per cent, increase in the grate area (from 53.3 sq. ft. to 84 sq. ft.) to suit the requirements of the coal under natural draft. With forced draft there was a tremendous volume of dense flame for some time after the grate was cleaned; so much, in fact, that a good deal of fuel passed away unburned. This excessive flaming was due to the high temperature in the fuel bed, caused by the intense forced-draft combustion; but this high temperature gradually covered the grate with a dense clinker, so that the passage of air was much reduced and there was practically no flame in the furnace. The blast had to be shut off every 4 hr. to clean the grate. An attempt to improve matters by introducing steam with the blast failed to help much, though with Belt coal steam was of marked benefit in reducing clinker troubles. A large percentage of ash prevents efficient contact of air with the fuel in the fire box, unless there is a very deep bed. When using Belt coal (25 to 30 per cent. ash) with natural draft and only a 24-in. bed, it is altogether probable that there was a large excess of oxygen in the hearth (though we have no gas analyses to confirm it), for a good deal of the air passing through the bed would come in contact with very little except ash. There was not enough volatile combustible to utilize this extra air (the ratio of volatile matter to fixed carbon is low in this coal), which therefore acted to cool the hearth. With the deep-bed gas-producer conditions this excess air could not pass through, We have gas analyses to prove this.
The essence of the clinkering problem is, like that of the slag problem, the question of fusibility; but it is seldom possible to predict from analysis of the ash what will be the qualities or the clinker, or even whether it will or will not clinker. For example, there are decided variations in the four following analyses of Diamondville ash, yet the clinkering qualities are practically constant.

It often happens that there are great variations in clinkering properties of different portions of the ash; yet if one portion is easily fusible, at the temperature to which it is subjected after the fuel is largely burned away from it, it is quite certain to surround other portions, so that they— contribute to the volume of clinker though they have not fused. The most satisfactory investigations of the clinker problem seem to be those made by E. G. Bailey. He shows the present hopelessness of judging anything from analyses; but has adopted a laboratory fusion test of the average ash, and says:
“ Even if the ash is made of a mixture of high and low fusing temperature material and if only that having a fusing temperature below that of the fuel bed is melted into a clinker, there will be a greater percentage of such ash fused into a clinker from a coal having a low average fusing temperature than from a coal having a high average fusing temperature. In actual practical tests it has been found that the percentage of ash which is formed into clinker as well as the obstructed grate area per pound of clinker hold a close relation to the fusing temperature of the ash from different coals when burned under similar conditions.”
He shows plots of some results which indicate that, with good average conditions on steam-boiler grates; less than 25 per cent, of the total ash clinkers if its average fusion temperature is 2,500° F. or higher; but the proportion of clinker rapidly increases to 50 per cent, as the average fusion temperature falls to 2,300° F.
Description and Discussion of Coals Used
The Diamondville run of mine is decidedly the most economical of all the coals we have used, though it costs, delivered to the furnaces, from one-eighth to one-half more than any of the others except Rock Springs. This is because it is much the most efficient. It is a long-flame bituminous coal from which, in a properly designed furnace, with natural draft, the volatile combustible matter is distilled in just about the same length of time that the fixed carbon is burning in the fire box; keeping the entire furnace nicely filled with actively burning flame about half the time, and a very considerable amount of flame all the time. The distillation is rapid enough to produce a high temperature near the bridge, where the ore is charged, yet slow enough for the gases to give up a reasonable proportion of their heat before passing into the flue. The normal furnace results consist in smelting 270 tons of hot calcine and flue dust per 24 hr., while using 60 tons of this coal. The temperature of the gases averages 2,800° F. just beyond the bridge, while the formation temperature of the slag is about 2,000° F. The freezing temperature of the slag, after it is formed, is possibly not over 1,800° F., but it has an actual temperature of nearly 2,100° F. as it runs out of the flue end of the furnace. The gases enter the flue at an average of 2,200° F. These conditions guarantee good settlement of the matte, and easy skimming of the slag.
With good natural draft the Diamondville ash forms a clinker of moderate hardness, which is barred out without much trouble, and at the same time maintains an excellent fuel bed and keeps the total loss of fuel, through the grate, down to 12 or 15 per cent, when there is not an excessive amount of fines in the coal received. Most of this loss occurs at the time of grating. With forced draft, however, the clinker is so hard and tough, and forms in such large masses, as to require excessive labor in cleaning the grates. It is thus seen that, with natural draft, Diamondville approaches the ideal coal for smelting; and in fact it has the reputation of being one of the best coals available in the Rocky Mountain region for this purpose.
Belt is a bituminous coal, containing a good deal of carbonaceous shale and considerable iron pyrite, the 25 and 30 per cent, of ash in the mine run being a serious source of difficulty and inefficiency in its use. With moderate fire-box temperatures the ash does not clinker, but remains to a large extent in lumps; yet with the intense fire-box combustion accompanying forced draft, and sometimes with strong natural draft, it clinkers seriously unless steam is used beneath the grate. It ordinarily gives but very little good flame unless the temperature in the fuel bed is greatly increased, by means of forced draft, and even then the flame is far from ideal. Not only is the percentage of volatile matter low, but its ratio to fixed carbon is also low. When used in our regular fire box with natural draft it will do no smelting; and even with the immense gas-producing fire box the temperature a little beyond the bridge was only 2,500° to 2,600° as compared to 2,800° from Diamondville coal used in the regular fire box; but there were very large volumes of this lower-temperature gas, because of the great quantity of coal consumed.
Bear Creek is a sub-bituminous coal (between good bituminous and lignite). It flames sufficiently to smelt with natural draft on a normal-sized grate; but gives a better flame and more efficient smelting with forced draft. Under any conditions it flames, after fresh firing, only a quarter to a half as long a time as Diamondville, and so requires more frequent firing. With natural draft it produces a temperature just beyond the bridge 150° to 200° F. lower than Diamondville; and with forced draft it is 100° F. lower than Diamondville. It appears to deteriorate in heating value rather fast after mining, but does not slack seriously. It does not clinker much under either natural or forced draft; and, for that reason, is subject to a 25 per cent, fuel loss through the grate.
Roundup is also a sub-bituminous coal. It flames similarly to Bear Creek and gives about the same furnace temperatures. It slacks somewhat rapidly on exposure. Its ash clinkers somewhat more than Bear Creek, but not as much as Diamondville; and consequently suffers an intermediate fuel loss into the ash pit.
It seems reasonable to suppose that, after removing moisture, the higher the ratio of hydrogen to carbon the larger will be the proportion of total combustible that will be given off in the volatile form and serve to produce flame in the hearth; other conditions, of course, being the same. On the other hand, flaming is in all probability reduced the higher the ratio of oxygen to total combustible. If these two statements are true, then the relation between the first of these ratios and the second is one indicator as to the value of coals for smelting. The following figures are derived from average ultimate analyses of certain of the coals discussed in this paper. These analyses are taken from Bulletin No. 22 of the U. S. Bureau of Mines (samples from near the surface of mine workings being omitted); and the relation just mentioned is shown in the last column.

For these four coals the numbers in the last column stand in the same order as the normal smelting efficiencies that can be obtained with any ordinary provision short of the elaborate and expensive gas-producing fire box that was used for Belt.
Other Important Factors
Of the factors other than fuel, the one that requires most careful attention is proper fluxing of the charges: and this means, not merely that a day’s average sample shall have a suitable analysis, but that each individual charge shall have such composition as to yield a readily fusible and fluid slag, of low specific gravity; it also means that the speed of smelting depends on how intimately the various constituents of each charge are mixed. From the following facts it is clear that these conditions are not always attained. In December, 1903, Mr. Mathewson changed the practice of adding a small proportion of lime rock flux directly to the reverberatories, without any special mixing with the rest of the charge, to mixing it, with some regularity, with the concentrates and fine ore fed to the roasters. The result was to increase the reverberatory performance from 144 tons smelted with 46 tons of coal (ratio 3.1) to 163 tons smelted with 48 tons (ratio 3.4). The most striking example we have had of the effect of poor fluxing came when a charge of calcine failed to smelt in three times the usual 1 hr. and 20 min., though the entire contents of the hearth got so hot that the matte pool “boiled” (gave off sulphur) at the end of the 4 hr., and frothed considerable of the accumulated slag out of the furnace. It is, of course, understood that any approach to such poor results is not at all common.
Preheating the Charge
It is well known that the capacity and economy of a furnace are very much decreased if the hot calcine from the roasters is allowed to cool before smelting. At Argo, Colo., a 23 per cent, increase in capacity was obtained, with no increase of fuel consumption, by charging the hot calcine directly to the reverberatory instead of allowing an intermediate cooling. The calcine made up only one-half of the charge, the other half continuing to be cold ore. At Anaconda the calcine enters the reverberatories at a temperature of 900° F.; and most of the flue dust is thoroughly hot.
Preheating the Air
Various attempts to save money by preheating the air for copper blast furnaces have been abandoned, though the fuel efficiency of the furnaces themselves was much increased when the air was raised to 700° or 800° F. There have been two reasons for the commercial failure: first, the cost of the extraneous fuel used to do the heating; second, the cost for much extra power to force the blast through the small and tortuous passages of the heating devices. Both of these troubles can be overcome for reverberatory work. The first step is to use the waste heat from the furnaces, instead of extra fuel. The gas comes away from these furnaces at a much higher temperature than from blast furnaces, and there is not the frequent inrush of large volumes of cold air such as occurs through the charge doors of the blast furnaces. In the few cases where preheated air has been used in reverberatories for copper smelting, natural draft and efficient heating devices have been utilized. The gas-fired furnaces at Great Falls have always had the checker work of the open-hearth steel furnace. The Peyton Chemical Works, near Martinez, Cal., heated the air for an oil-fired furnace in open flues, without checker work but with several turns purposely introduced. With hot air (varying from 750° to 1,830° F.) their furnace capacity was 50 per cent, greater than with cold air. It is easier and cheaper to remove dust from these open flues than from the checker work.
Producing Steam with Waste Heat
Since the practice of utilizing the flue gases to generate steam was adopted at Anaconda it has also been put into effect at several other large plants. At Anaconda about 25 per cent, of the heating value of the total coal is thus made effective; while only 15 per cent, of this value is actually utilized for smelting. This steam from the eight furnaces generates 30 per cent, of the 16,700 mechanical horse power used in the entire concentrating and smelting plant.
Boilers for this service must be arranged for the gases to pass through in a straight line, and with plenty of passage area, in order not to affect the draft. With the several changes of direction found in ordinary boiler practice the smelting capacity is seriously reduced; so much, in fact, as to affect the profits much more than the money value of all the steam.
For a good deal of information and suggestion, the writer is indebted to W. M. Kelly, general smelter foreman; F. W. C. Whyte, manager of coal mines; and especially to Edward O’Brien, general reverberatory foreman.

